Cancer Res Treat.
2018 Oct;50(4):1130-1139. 10.4143/crt.2017.171.
Prognostic Factors and Decision Tree for Long-Term Survival in Metastatic Uveal Melanoma
- Affiliations
-
- 1Department of Ophthalmology, Bellvitge University Hospital, Barcelona, Spain. dlorenzo@bellvitgehospital.cat
- 2Department of Oncology, Vall d'Hebron University Hospital, Barcelona, Spain.
- 3Department of Oncology, Catalan Institute of Oncology, Barcelona, Spain.
- 4Department of Brachytherapy, Catalan Institute of Oncology, Barcelona, Spain.
- 5Medical Research Division, IMIM–Hospital del Mar Medical Research Institute, Barcelona, Spain.
- KMID: 2424786
- DOI: http://doi.org/10.4143/crt.2017.171
Abstract
- PURPOSE
The purpose of this study was to demonstrate the existence of a bimodal survival pattern in metastatic uveal melanoma. Secondary aims were to identify the characteristics and prognostic factors associated with long-term survival and to develop a clinical decision tree.
MATERIALS AND METHODS
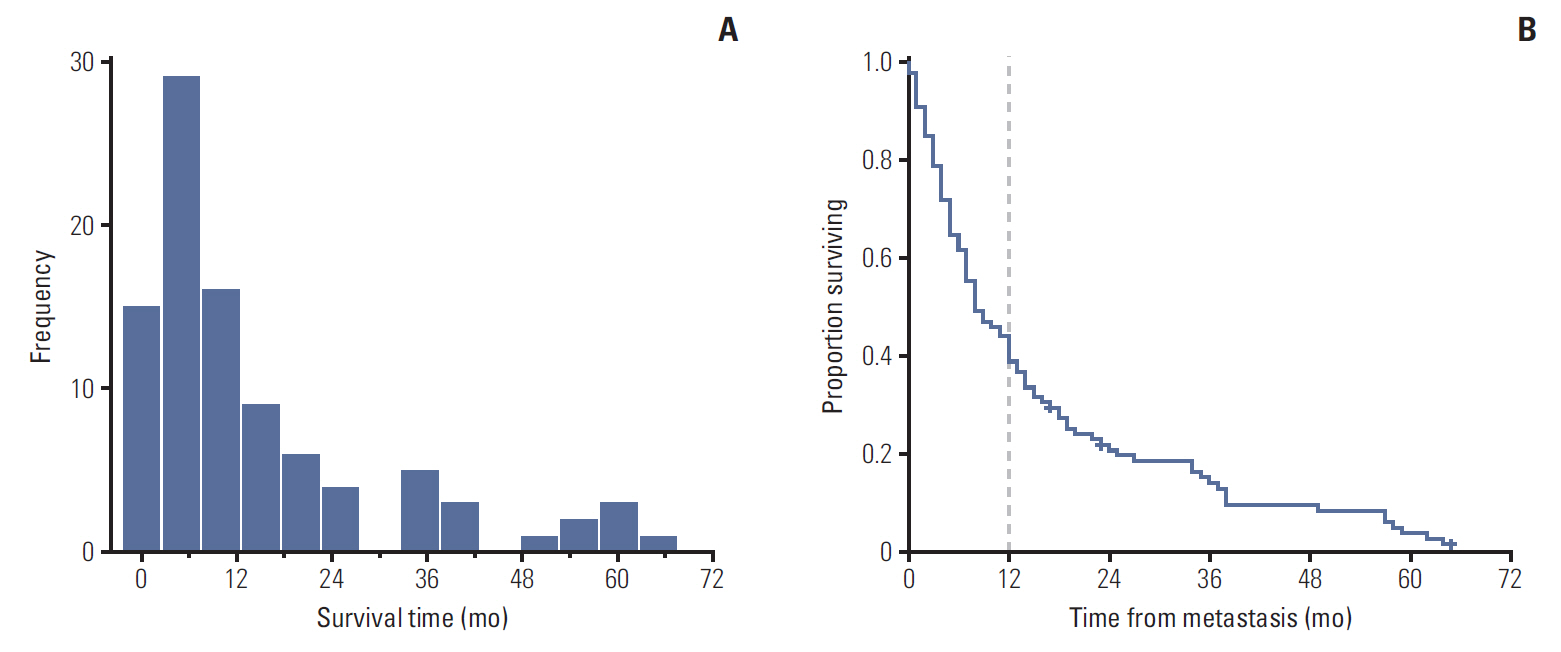
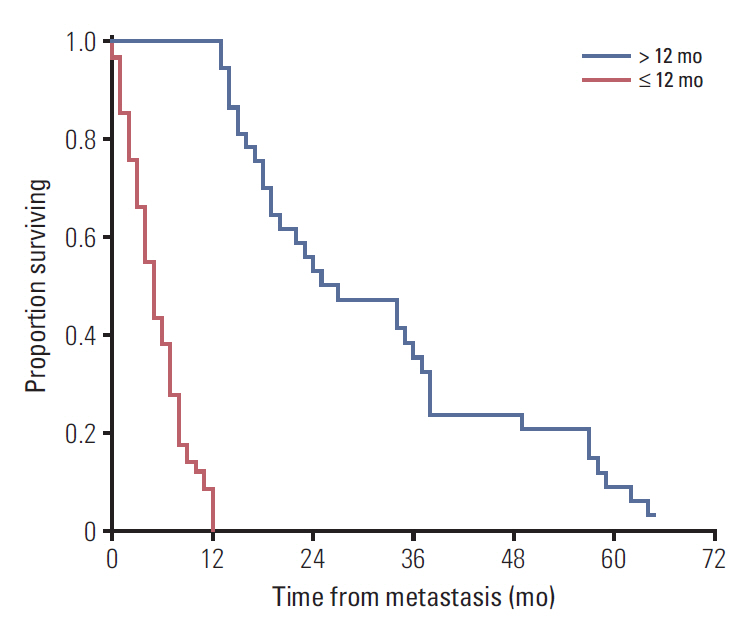
The medical records of 99 metastatic uveal melanoma patients were retrospectively reviewed. Patients were classified as either short (≤ 12 months) or long-term survivors (> 12 months) based on a graphical interpretation of the survival curve after diagnosis of the first metastatic lesion. Ophthalmic and oncological characteristicswere assessed in both groups.
RESULTS
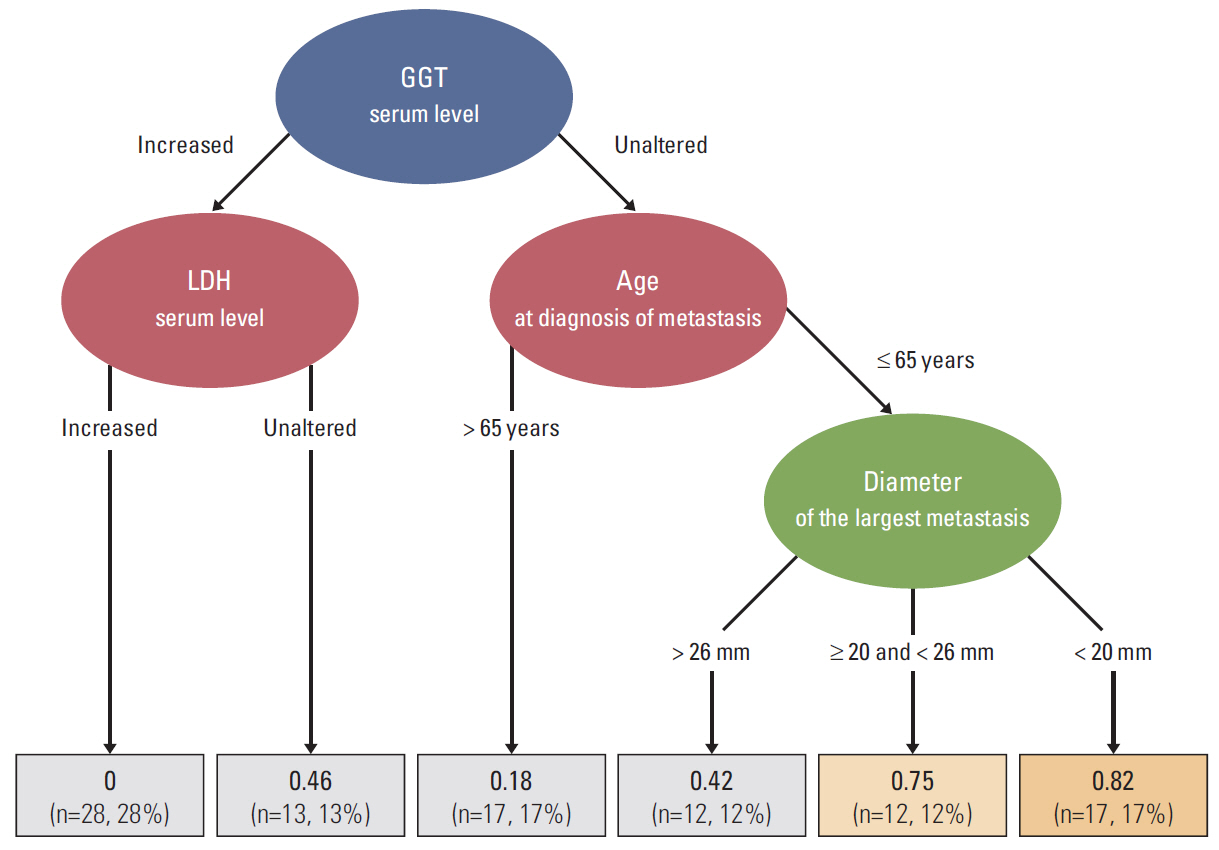
Of the 99 patients, 62 (62.6%) were classified as short-term survivors, and 37 (37.4%) as long-term survivors. The multivariate analysis identified the following predictors of long-term survival: age ≤ 65 years (p=0.012) and unaltered serum lactate dehydrogenase levels (p=0.018); additionally, the size (smaller vs. larger) of the largest liver metastasis showed a trend towards significance (p=0.063). Based on the variables significantly associated with long-term survival, we developed a decision tree to facilitate clinical decision-making.
CONCLUSION
The findings of this study demonstrate the existence of a bimodal survival pattern in patients with metastatic uveal melanoma. The presence of certain clinical characteristics at diagnosis of distant disease is associated with long-term survival. A decision tree was developed to facilitate clinical decision-making and to counsel patients about the expected course of disease.
MeSH Terms
Figure
Reference
-
References
1. Bishop KD, Olszewski AJ. Epidemiology and survival outcomes of ocular and mucosal melanomas: a population-based analysis. Int J Cancer. 2014; 134:2961–71.
Article2. Collaborative Ocular Melanoma Study Group. The COMS randomized trial of iodine 125 brachytherapy for choroidal melanoma: V. Twelve-year mortality rates and prognostic factors: COMS report No. 28. Arch Ophthalmol. 2006; 124:1684–93.3. Cho Y, Chang JS, Yoon JS, Lee SC, Kim YB, Kim JH, et al. Ruthenium-106 brachytherapy with or without additional local therapy shows favorable outcome for variable-sized choroidal melanomas in Korean patients. Cancer Res Treat. 2018; 50:138–47.
Article4. Agarwala SS, Eggermont AM, O'Day S, Zager JS. Metastatic melanoma to the liver: a contemporary and comprehensive review of surgical, systemic, and regional therapeutic options. Cancer. 2014; 120:781–9.
Article5. Gragoudas ES, Egan KM, Seddon JM, Glynn RJ, Walsh SM, Finn SM, et al. Survival of patients with metastases from uveal melanoma. Ophthalmology. 1991; 98:383–9.6. Singh AD, Kalyani P, Topham A. Estimating the risk of malignant transformation of a choroidal nevus. Ophthalmology. 2005; 112:1784–9.
Article7. Singh AD, Turell ME, Topham AK. Uveal melanoma: trends in incidence, treatment, and survival. Ophthalmology. 2011; 118:1881–5.
Article8. Woodman SE. Metastatic uveal melanoma: biology and emerging treatments. Cancer J. 2012; 18:148–52.9. Buder K, Gesierich A, Gelbrich G, Goebeler M. Systemic treatment of metastatic uveal melanoma: review of literature and future perspectives. Cancer Med. 2013; 2:674–86.
Article10. Duh EJ, Schachat AP, Albert DM, Patel SM. Long-term survival in a patient with uveal melanoma and liver metastasis. Arch Ophthalmol. 2004; 122:285–7.
Article11. Diener-West M, Reynolds SM, Agugliaro DJ, Caldwell R, Cumming K, Earle JD, et al. Development of metastatic disease after enrollment in the COMS trials for treatment of choroidal melanoma: Collaborative Ocular Melanoma Study Group Report No. 26. Arch Ophthalmol. 2005; 123:1639–43.12. Rietschel P, Panageas KS, Hanlon C, Patel A, Abramson DH, Chapman PB. Variates of survival in metastatic uveal melanoma. J Clin Oncol. 2005; 23:8076–80.
Article13. Buzzacco DM, Abdel-Rahman MH, Park S, Davidorf F, Olencki T, Cebulla CM. Long-term survivors with metastatic uveal melanoma. Open Ophthalmol J. 2012; 6:49–53.14. Edge SB, Byrd SR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC cancer staging manual. New York: Springer;2010. p. 547–59.15. Hothorn T, Hornik K, Zeileis A. Unbiased recursive partitioning: a conditional inference framework. J Comput Graph Stat. 2006; 15:651–74.
Article16. Kath R, Hayungs J, Bornfeld N, Sauerwein W, Hoffken K, Seeber S. Prognosis and treatment of disseminated uveal melanoma. Cancer. 1993; 72:2219–23.
Article17. Eskelin S, Pyrhonen S, Hahka-Kemppinen M, Tuomaala S, Kivela T. A prognostic model and staging for metastatic uveal melanoma. Cancer. 2003; 97:465–75.
Article18. Harbour JW, Chao DL. A molecular revolution in uveal melanoma: implications for patient care and targeted therapy. Ophthalmology. 2014; 121:1281–8.19. Segura MF, Belitskaya-Levy I, Rose AE, Zakrzewski J, Gaziel A, Hanniford D, et al. Melanoma MicroRNA signature predicts post-recurrence survival. Clin Cancer Res. 2010; 16:1577–86.
Article20. Damato B. Developments in the management of uveal melanoma. Clin Exp Ophthalmol. 2004; 32:639–47.
Article21. Singh AD, Shields CL, Shields JA. Prognostic factors in uveal melanoma. Melanoma Res. 2001; 11:255–63.
Article22. Hsueh EC, Essner R, Foshag LJ, Ye X, Wang HJ, Morton DL. Prolonged survival after complete resection of metastases from intraocular melanoma. Cancer. 2004; 100:122–9.
Article23. Frenkel S, Nir I, Hendler K, Lotem M, Eid A, Jurim O, et al. Long-term survival of uveal melanoma patients after surgery for liver metastases. Br J Ophthalmol. 2009; 93:1042–6.
Article24. Algazi AP, Tsai KK, Shoushtari AN, Munhoz RR, Eroglu Z, Piulats JM, et al. Clinical outcomes in metastatic uveal melanoma treated with PD-1 and PD-L1 antibodies. Cancer. 2016; 122:3344–53.
Article25. Carvajal RD, Schwartz GK, Tezel T, Marr B, Francis JH, Nathan PD. Metastatic disease from uveal melanoma: treatment options and future prospects. Br J Ophthalmol. 2017; 101:38–44.
Article26. Piulats JM, De La Cruz-Merino L, Curiel García MT, Berrocal A, Alonso-Carrión L, Espinosa E, et al. Phase II multicenter, single arm, open label study of nivolumab (NIVO) in combination with ipilimumab (IPI) as first line in adult patients (pts) with metastatic uveal melanoma (MUM): GEM1402 NCT-02626962. J Clin Oncol. 2017; 35(Suppl):Abstr 9533.
Article27. Oliva M, Rullan AJ, Piulats JM. Uveal melanoma as a target for immune-therapy. Ann Transl Med. 2016; 4:172.
Article28. Doherty JR, Cleveland JL. Targeting lactate metabolism for cancer therapeutics. J Clin Invest. 2013; 123:3685–92.
Article29. Bedikian AY, Legha SS, Mavligit G, Carrasco CH, Khorana S, Plager C, et al. Treatment of uveal melanoma metastatic to the liver: a review of the M. D. Anderson Cancer Center experience and prognostic factors. Cancer. 1995; 76:1665–70.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinicopathologic features and survival outcomes of ocular melanoma: a series of 31 cases from a tertiary university hospital
- Uveal Melanoma: A Review of the Literature and Personal Experiences
- Prognoses and Clinical Outcomes of Primary and Recurrent Uveal Melanoma
- Cytologic Features of Metastatic Melanoma
- Growth and Characterization of the Uveal Melanoma in Vitro