Nat Prod Sci.
2018 Sep;24(3):159-163. 10.20307/nps.2018.24.3.159.
Chinoketides A and B, Two New Antimicrobial Polyketides from the Endophytes of Distylium chinense with the “Black-Box†Co-culture Method
- Affiliations
-
- 1Hubei Key Laboratory of Natural Product Research and Development, College of Biological and Pharmaceutical Sciences, China Three Gorges University, Yichang, 443002, People's of Republic China. zhyguoctgu@foxmail.com
- 2Institute of Pharmaceutical Biology and Biotechnology, Heinrich-Heine-Universitat Dusseldorf, Universitatsstrasse 1, 40225 Dusseldorf, Germany.
- KMID: 2422007
- DOI: http://doi.org/10.20307/nps.2018.24.3.159
Abstract
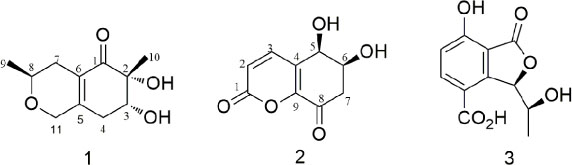
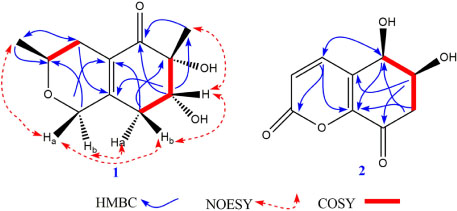
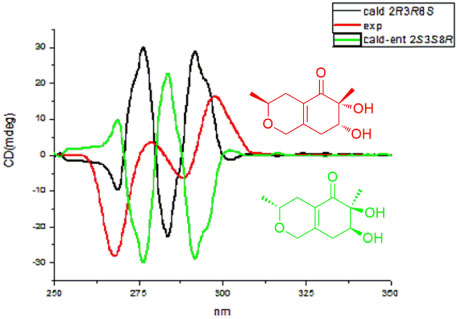
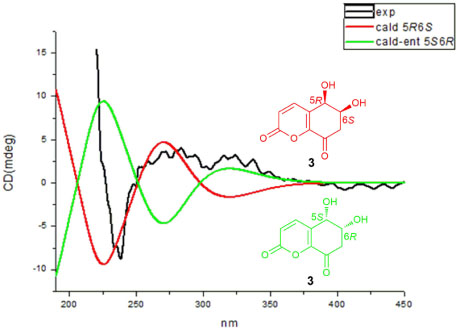
- Two new polyketides, chinoketides A and B (1 - 2) with a known compound xylarphthalide A (3), were isolated from the solid medium of the endophytes from the leaves of the relic plant Distylium chinense with the "black-box" co-culture method, and the structures of two new compounds were elucidated by NMR, MS and CD spectra. And the absolute configurations of chinoketides A (1) and B (2) were determined as 2R,3R,8S and 5R,6S by calculating their ECD spectra to compare with the experimental CD spectra. Finally, the antimicrobial activities were evaluated to Erwinia carotovora sub sp. Carotovora (Jones) Bersey et al, and the results showed that compounds 1 - 3 displayed the antimicrobial activities with MIC value at 20.5, 30.4 and 10.2 µg/mL.
Keyword
MeSH Terms
Figure
Reference
-
1. Stierle A, Strobel G, Strierle D. Science. 1993; 260:214–216.2. Brady SF, Singh MP, Janso JE, Clardy J. Org Lett. 2000; 2:4047–4049.3. Kontnik R, Clardy J. Org Lett. 2008; 10:4149–4151.4. Li J, Li L, Si Y, Jiang X, Guo L, Che Y. Org Lett. 2011; 13:2670–2673.5. Huang X, Huang H, Li H, Sun X, Huang H, Lu Y, Lin Y, Long Y, She Z. Org Lett. 2013; 15:721–723.6. Xiao Z, Huang H, Shao C, Xia X, Ma L, Huang X, Lu Y, Lin Y, Long Y, She Z. Org Lett. 2013; 15:2522–2525.7. Zhou M, Miao MM, Du G, Li XN, Shang SZ, Zhao W, Liu ZH, Yang GY, Che CT, Hu QF, Gao XM. Org Lett. 2014; 16:5016–5019.8. Zhang P, Mándi A, Li XM, Du FY, Wang JN, Li X, Kurtán T, Wang BG. Org Lett. 2014; 16:4834–4837.9. Marmann A, Aly AH, Lin W, Wang B, Proksch P. Mar Drugs. 2014; 12:1043–1065.10. Arnold AE, Carson WP, Schnitzer SA. West Sussex. UK: Wiley-Blackwell;2008. p. 254–255.11. Guo Z, Li X, Zhang L, Feng Z, Deng Z, He H, Zou K. Nat Prod Res. 2016; 30:2582–2589.12. Feng ZW, Lv MM, Li XS, Zhang L, Liu CX, Guo ZY, Deng ZS, Zou K, Proksch P. Molecules. 2016; 21:1438–1446.13. Wan Q, Feng Z, Li X, Lv M, Guo Z, Deng Z, Zou K. Z Naturforsch B. 2016; 71:283–286.14. Zhang L, Liu Y, Deng Z, Guo Z, Chen J, Tu X, Zou K. Nat Prod Commun. 2013; 8:83–84.15. Bruhn T, Schaumlöffel A, Hemberger Y, Bringmann G. Chirality. 2013; 25:243–249.16. Sheldrick GM. Shelxl-97, Program for the Refinements of Crystal Structure. Germany: University of Göttingen;1997.17. Yakushiji F, Miyamoto Y, Kunoh Y, Okamoto R, Nakaminami H, Yamazaki Y, Noguchi N, Hayashi Y. ACS Med Chem Lett. 2013; 4:220–224.18. Zheng N, Yao F, Liang X, Liu Q, Xu W, Liang Y, Liu X, Li J, Yang R. Nat Prod Res. 2018; 32:755–760.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Endophytic Fungi Inhabiting Medicinal Plants and Their Bioactive Secondary Metabolites
- Computational Approach for the Analysis of Post-PKS Glycosylation Step
- Bioprospecting Endophytic Fungi and Their Metabolites from Medicinal Tree Aegle marmelos in Western Ghats, India
- Diversity of Endophytic Fungi Isolated from Korean Ginseng Leaves
- Antimicrobials and Antimicrobial Resistant Superbacteria