Lab Anim Res.
2018 Sep;34(3):126-131. 10.5625/lar.2018.34.3.126.
Neither polyphenol-rich red wine nor fenofibrate affects the onset of type-1 diabetes mellitus in the BB rat
- Affiliations
-
- 1The Rolf Luft Research Center for Diabetes and Endocrinology, Karolinska Institutet, Karolinska University Hospital L1, Stockholm, Sweden. Lisa.juntti-berggren@ki.se
- KMID: 2420825
- DOI: http://doi.org/10.5625/lar.2018.34.3.126
Abstract
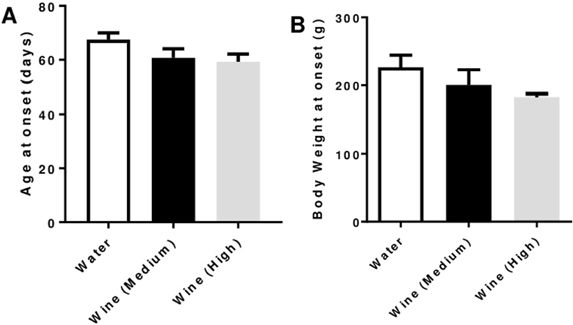
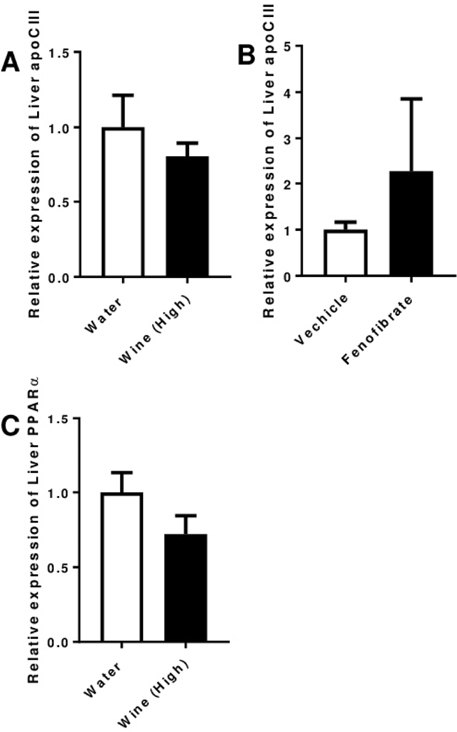
- Serum levels of the pro-inflammatory apolipoprotein CIII (apoCIII) are increased in type-1 diabetic (T1D) patients and when β-cells are exposed to apoCIII they undergo apoptosis, which can be prevented by an antibody against apoCIII. We have previously investigated the BB rat, an animal model that develops a human-like T1D at the age of around 60 days, and found that apoCIII was also increased in sera from pre-diabetic rats and this promoted β-cell death. Lowering apoCIII with an oligonucleotide antisense during a phase of the pre-diabetic period prolonged the time to onset of T1D. In order to find other ways to lower apoCIII we in this study tested non-alcoholic red wine with medium and high concentrations of polyphenols and the lipid-lowering drug, fenofibrate, both reported to decrease the expression of apoCIII by activating peroxisome proliferator-activated receptors. Pre-diabetic BB-rats were treated orally for one month prior to the expected onset of diabetes with the two different wines or fenofibrate. None of the treatments prevented or prolonged the time to onset of diabetes and the expression of apoCIII was unaffected in this animal model for T1D. However, it must be emphasized that this does not exclude that other species can show a response to these substances.
Keyword
MeSH Terms
Figure
Reference
-
1. Juntti-Berggren L, Larsson O, Rorsman P, Ammälä C, Bokvist K, Wåhlander K, Nicotera P, Dypbukt J, Orrenius S, Hallberg A, Berggren PO. Increased activity of L-type Ca2+ channels exposed to serum from patients with type I diabetes. Science. 1993; 261(5117):86–90.
Article2. Shi Y, Yang G, Yu J, Yu L, Westenbroek R, Catterall WA, Juntti-Berggren L, Berggren PO, Yang SN. Apolipoprotein CIII hyperactivates β cell CaV1 channels through SR-BI/β1 integrin-dependent coactivation of PKA and Src. Cell Mol Life Sci. 2014; 71(7):1289–1303.
Article3. Holmberg R, Refai E, Höög A, Crooke RM, Graham M, Olivecrona G, Berggren PO, Juntti-Berggren L. Lowering apolipoprotein CIII delays onset of type 1 diabetes. Proc Natl Acad Sci U S A. 2011; 108(26):10685–10689.
Article4. Chen M, Breslow JL, Li W, Leff T. Transcriptional regulation of the apoC-III gene by insulin in diabetic mice: correlation with changes in plasma triglyceride levels. J Lipid Res. 1994; 35(11):1918–1924.
Article5. Altomonte J, Cong L, Harbaran S, Richter A, Xu J, Meseck M, Dong HH. Foxo1 mediates insulin action on apoC-III and triglyceride metabolism. J Clin Invest. 2004; 114(10):1493–1503.
Article6. Vu-Dac N, Gervois P, Torra IP, Fruchart JC, Kosykh V, Kooistra T, Princen HM, Dallongeville J, Staels B. Retinoids increase human apo C-III expression at the transcriptional level via the retinoid X receptor. Contribution to the hypertriglyceridemic action of retinoids. J Clin Invest. 1998; 102(3):625–632.
Article7. Palsamy P, Subramanian S. Ameliorative potential of resveratrol on proinflammatory cytokines, hyperglycemia mediated oxidative stress, and pancreatic beta-cell dysfunction in streptozotocin-nicotinamide-induced diabetic rats. J Cell Physiol. 2010; 224(2):423–432.8. Chuang CC, McIntosh MK. Potential mechanisms by which polyphenol-rich grapes prevent obesity-mediated inflammation and metabolic diseases. Annu Rev Nutr. 2011; 31:155–176.
Article9. Del Bas JM, Fernández-Larrea J, Blay M, Ardèvol A, Salvadó MJ, Arola L, Bladé C. Grape seed procyanidins improve atherosclerotic risk index and induce liver CYP7A1 and SHP expression in healthy rats. FASEB J. 2005; 19(3):479–481.
Article10. Landrault N, Poucheret P, Azay J, Krosniak M, Gasc F, Jenin C, Cros G, Teissedre PL. Effect of a polyphenols-enriched chardonnay white wine in diabetic rats. J Agric Food Chem. 2003; 51(1):311–318.
Article11. Noonan JE, Jenkins AJ, Ma JX, Keech AC, Wang JJ, Lamoureux EL. An update on the molecular actions of fenofibrate and its clinical effects on diabetic retinopathy and other microvascular end points in patients with diabetes. Diabetes. 2013; 62(12):3968–3975.
Article12. Chen Y, Hu Y, Lin M, Jenkins AJ, Keech AC, Mott R, Lyons TJ, Ma JX. Therapeutic effects of PPARα agonists on diabetic retinopathy in type 1 diabetes models. Diabetes. 2013; 62(1):261–272.
Article13. Juntti-Berggren L, Refai E, Appelskog I, Andersson M, Imreh G, Dekki N, Uhles S, Yu L, Griffiths WJ, Zaitsev S, Leibiger I, Yang SN, Olivecrona G, Jörnvall H, Berggren PO. Apolipoprotein CIII promotes Ca2+-dependent beta cell death in type 1 diabetes. Proc Natl Acad Sci U S A. 2004; 101(27):10090–10094.14. Åvall K, Ali Y, Leibiger IB, Leibiger B, Moede T, Paschen M, Dicker A, Daré E, Köhler M, Ilegems E, Abdulreda MH, Graham M, Crooke RM, Tay VS, Refai E, Nilsson SK, Jacob S, Selander L, Berggren PO, Juntti-Berggren L. Apolipoprotein CIII links islet insulin resistance to β-cell failure in diabetes. Proc Natl Acad Sci U S A. 2015; 112(20):E2611–E2619.
Article15. Hiukka A, Ståhlman M, Pettersson C, Levin M, Adiels M, Teneberg S, Leinonen ES, Hultén LM, Wiklund O, Oresic M, Olofsson SO, Taskinen MR, Ekroos K, Borén J. ApoCIII-enriched LDL in type 2 diabetes displays altered lipid composition, increased susceptibility for sphingomyelinase, and increased binding to biglycan. Diabetes. 2009; 58(9):2018–2026.
Article16. Kohan AB. Apolipoprotein C-III: a potent modulator of hypertriglyceridemia and cardiovascular disease. Curr Opin Endocrinol Diabetes Obes. 2015; 22(2):119–125.17. Norata GD, Tsimikas S, Pirillo A, Catapano AL. Apolipoprotein C-III: From Pathophysiology to Pharmacology. Trends Pharmacol Sci. 2015; 36(10):675–687.
Article18. Ooi EM, Barrett PH, Chan DC, Watts GF. Apolipoprotein C-III: understanding an emerging cardiovascular risk factor. Clin Sci (Lond). 2008; 114(10):611–624.
Article19. Hokanson JE, Kinney GL, Cheng S, Erlich HA, Kretowski A, Rewers M. Susceptibility to type 1 diabetes is associated with ApoCIII gene haplotypes. Diabetes. 2006; 55(3):834–838.
Article20. Petersen KF, Dufour S, Hariri A, Nelson-Williams C, Foo JN, Zhang XM, Dziura J, Lifton RP, Shulman GI. Apolipoprotein C3 gene variants in nonalcoholic fatty liver disease. N Engl J Med. 2010; 362(12):1082–1089.
Article21. Perry RJ, Samuel VT, Petersen KF, Shulman GI. The role of hepatic lipids in hepatic insulin resistance and type 2 diabetes. Nature. 2014; 510(7503):84–91.
Article22. Pollin TI, Damcott CM, Shen H, Ott SH, Shelton J, Horenstein RB, Post W, McLenithan JC, Bielak LF, Peyser PA, Mitchell BD, Miller M, O'Connell JR, Shuldiner AR. A null mutation in human APOC3 confers a favorable plasma lipid profile and apparent cardioprotection. Science. 2008; 322(5908):1702–1705.
Article23. Atzmon G, Rincon M, Schechter CB, Shuldiner AR, Lipton RB, Bergman A, Barzilai N. Lipoprotein genotype and conserved pathway for exceptional longevity in humans. PLoS Biol. 2006; 4(4):e113.
Article24. Jørgensen AB, Frikke-Schmidt R, Nordestgaard BG, Tybjærg-Hansen A. Loss-of-function mutations in APOC3 and risk of ischemic vascular disease. N Engl J Med. 2014; 371(1):32–41.25. Alaupovic P, Bard JM, Tavella M, Shafer D. Identification of apoB-containing lipoprotein families in NIDDM. Diabetes. 1992; 41:Suppl 2. 18–25.
Article26. Florez H, Mendez A, Casanova-Romero P, Larreal-Urdaneta C, Castillo-Florez S, Lee D, Goldberg R. Increased apolipoprotein C-III levels associated with insulin resistance contribute to dyslipidemia in normoglycemic and diabetic subjects from a triethnic population. Atherosclerosis. 2006; 188(1):134–141.
Article27. Chiva-Blanch G, Urpi-Sarda M, Ros E, Valderas-Martinez P, Casas R, Arranz S, Guillén M, Lamuela-Raventós RM, Llorach R, Andres-Lacueva C, Estruch R. Effects of red wine polyphenols and alcohol on glucose metabolism and the lipid profile: a randomized clinical trial. Clin Nutr. 2013; 32(2):200–206.
Article28. Markoski MM, Garavaglia J, Oliveira A, Olivaes J, Marcadenti A. Molecular Properties of Red Wine Compounds and Cardiometabolic Benefits. Nutr Metab Insights. 2016; 9:51–57.
Article29. Muhlestein JB, May HT, Jensen JR, Horne BD, Lanman RB, Lavasani F, Wolfert RL, Pearson RR, Yannicelli HD, Anderson JL. The reduction of inflammatory biomarkers by statin, fibrate, and combination therapy among diabetic patients with mixed dyslipidemia: the DIACOR (Diabetes and Combined Lipid Therapy Regimen) study. J Am Coll Cardiol. 2006; 48(2):396–401.30. Vadillo M, Ardévol A, Fernández-Larrea J, Pujadas G, Bladé C, Salvadó MJ, Arola L, Blay M. Moderate red-wine consumption partially prevents body weight gain in rats fed a hyperlipidic diet. J Nutr Biochem. 2006; 17(2):139–142.31. Milat AM, Mudniæ I, Grkoviæ I, Kljuèeviæ N, Grga M, Jerèiæ I, Juriæ D, Ivankoviæ D, Benzon B, Boban M. Effects of White Wine Consumption on Weight in Rats: Do Polyphenols Matter? Oxid Med Cell Longev. 2017; 2017:8315803.
Article32. Ye P, Wang ZJ, Zhang XJ, Zhao YL. Age-related decrease in expression of peroxisome proliferator-activated receptor alpha and its effects on development of dyslipidemia. Chin Med J (Engl). 2005; 118(13):1093–1098.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Spontaneous diabetes mellitus in the BB rat: anti-islet cellular cytotoxicity and natural killer (NK) cell activity
- Spontaneous diabetes mellitus in the BB rat: effect of depletion ofT lymphocytes and natural killer cells on anti-islet cellular cytotoxicity
- Management of Type 2 Diabetes Mellitus in Adolescents and Young Adults
- HDL Cholesterol Reduction during Rosiglitazone and Fenofibrate Treatment in a Type 2 Diabetes Mellitus Patient with Dyslipidemia
- Role of Fenofibrate Use in Dyslipidemia and Related Comorbidities in the Asian Population: A Narrative Review