Yonsei Med J.
2017 May;58(3):613-618. 10.3349/ymj.2017.58.3.613.
Multiplex Ligation-Dependent Probe Amplification in X-linked Recessive Muscular Dystrophy in Korean Subjects
- Affiliations
-
- 1Department of Rehabilitation Medicine, Gangnam Severance Hospital and Rehabilitation Institute of Neuromuscular Disease, Yonsei University College of Medicine, Seoul, Korea. kswoong@yuhs.ac
- 2Department of Laboratory Medicine, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2419120
- DOI: http://doi.org/10.3349/ymj.2017.58.3.613
Abstract
- PURPOSE
Duchenne muscular dystrophy (DMD) and Becker muscular dystrophy (BMD) are similar genetic disorders whose patterns of mutation and disease phenotypes might be expected to show differences among different countries. We analyzed multiplex ligation-dependent probe amplification (MLPA) data in a large number of Korean patients with DMD/BMD.
MATERIALS AND METHODS
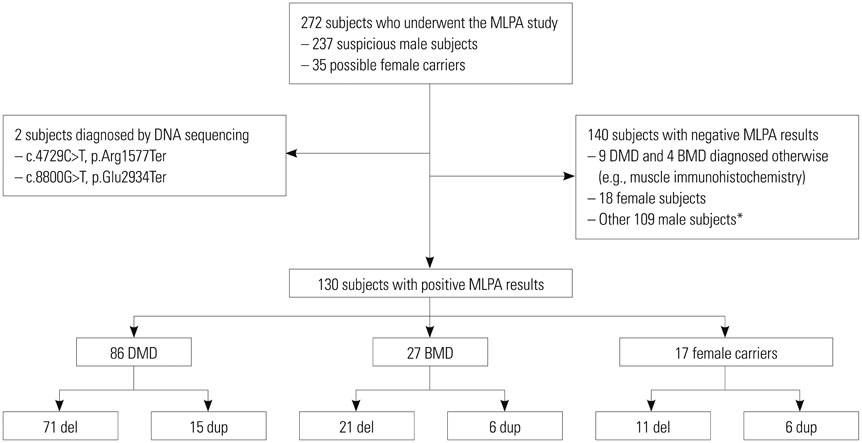
We obtained 130 positive MLPA results (86 DMD, 27 BMD, and 17 female carriers) from 272 candidates (237 clinically suspected patients and 35 possible female carriers) who took part in this study. We analyzed the mutation patterns among 113 patients diagnosed by MLPA and calculated deletion/duplication percentages from a total of 128 patients, including 15 patients who were diagnosed using methods other than MLPA. We also analyzed hot spot locations among the 130 MLPA-positive results.
RESULTS
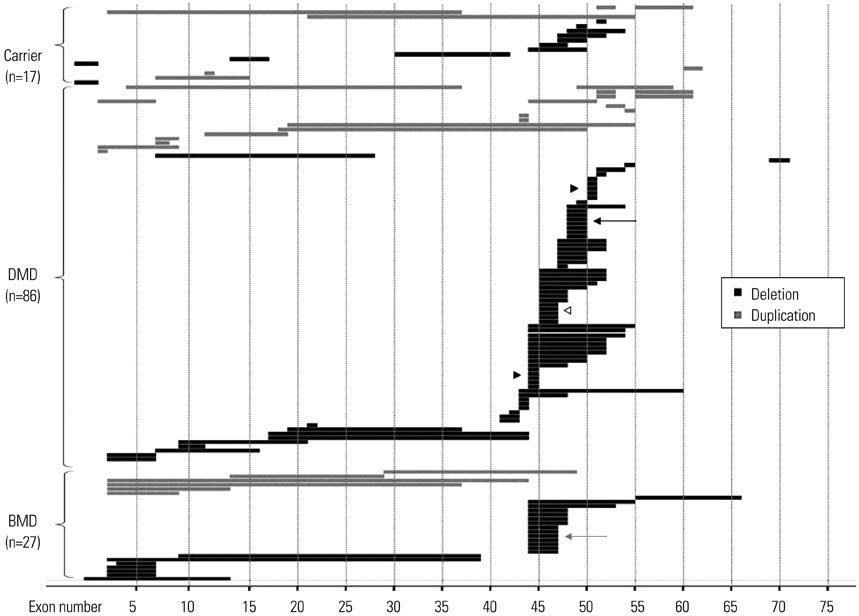
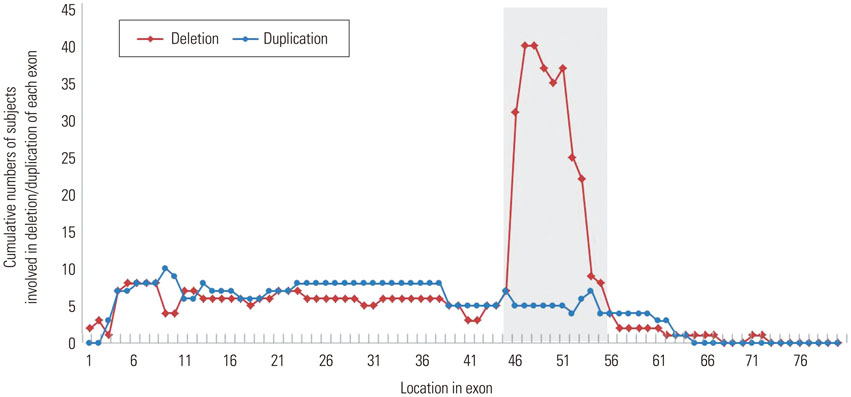
Most mutations were detected in a central hot spot region between exons 44 and 55 (80 samples, 60.6%). Unlike previous reports, a second frequently observed hot spot near the 5'-end was not distinctive. MLPA detected deletions in specific exons in 92 patients with DMD/BMD (71.8%) and duplications in 21 patients (16.4%).
CONCLUSION
Our MLPA study of a large number of Korean patients with DMD/BMD identified the most frequent mutation hot spot, as well as a unique hot spot pattern. DMD gene mutation patterns do not appear to show significant ethnic differences.
Keyword
MeSH Terms
-
Adolescent
Adult
Asian Continental Ancestry Group/genetics
Child
DNA Mutational Analysis/*methods
Dystrophin/*genetics
Exons/genetics
Female
Gene Deletion
Heterozygote
Humans
Male
Mass Screening
Multiplex Polymerase Chain Reaction/*methods
Muscular Dystrophy, Duchenne/*diagnosis/ethnology/*genetics
Mutation/*genetics
Republic of Korea
Retrospective Studies
Sequence Analysis, DNA
Sequence Deletion
Young Adult
Dystrophin
Figure
Reference
-
1. Mendell JR, Shilling C, Leslie ND, Flanigan KM, al-Dahhak R, Gastier-Foster J, et al. Evidence-based path to newborn screening for Duchenne muscular dystrophy. Ann Neurol. 2012; 71:304–313.
Article2. Deconinck N, Dan B. Pathophysiology of duchenne muscular dystrophy: current hypotheses. Pediatr Neurol. 2007; 36:1–7.
Article3. American Academy of Pediatrics Section on Cardiology and Cardiac Surgery. Cardiovascular health supervision for individuals affected by Duchenne or Becker muscular dystrophy. Pediatrics. 2005; 116:1569–1573.4. Moxley RT 3rd, Pandya S, Ciafaloni E, Fox DJ, Campbell K. Change in natural history of Duchenne muscular dystrophy with long-term corticosteroid treatment: implications for management. J Child Neurol. 2010; 25:1116–1129.
Article5. Mandel JL. Dystrophin. The gene and its product. Nature. 1989; 339:584–586.6. Nishio H, Takeshima Y, Narita N, Yanagawa H, Suzuki Y, Ishikawa Y, et al. Identification of a novel first exon in the human dystrophin gene and of a new promoter located more than 500 kb upstream of the nearest known promoter. J Clin Invest. 1994; 94:1037–1042.
Article7. Matsuo M. Duchenne and Becker muscular dystrophy: from gene diagnosis to molecular therapy. IUBMB Life. 2002; 53:147–152.
Article8. Muntoni F, Torelli S, Ferlini A. Dystrophin and mutations: one gene, several proteins, multiple phenotypes. Lancet Neurol. 2003; 2:731–740.
Article9. Koenig M, Hoffman EP, Bertelson CJ, Monaco AP, Feener C, Kunkel LM. Complete cloning of the Duchenne muscular dystrophy (DMD) cDNA and preliminary genomic organization of the DMD gene in normal and affected individuals. Cell. 1987; 50:509–517.
Article10. Forrest SM, Cross GS, Flint T, Speer A, Robson KJ, Davies KE. Further studies of gene deletions that cause Duchenne and Becker muscular dystrophies. Genomics. 1988; 2:109–114.
Article11. Worton RG, Thompson MW. Genetics of Duchenne muscular dystrophy. Annu Rev Genet. 1988; 22:601–629.
Article12. Den Dunnen JT, Grootscholten PM, Bakker E, Blonden LA, Ginjaar HB, Wapenaar MC, et al. Topography of the Duchenne muscular dystrophy (DMD) gene: FIGE and cDNA analysis of 194 cases reveals 115 deletions and 13 duplications. Am J Hum Genet. 1989; 45:835–847.13. Hu XY, Ray PN, Murphy EG, Thompson MW, Worton RG. Duplicational mutation at the Duchenne muscular dystrophy locus: its frequency, distribution, origin, and phenotypegenotype correlation. Am J Hum Genet. 1990; 46:682–695.14. White S, Kalf M, Liu Q, Villerius M, Engelsma D, Kriek M, et al. Comprehensive detection of genomic duplications and deletions in the DMD gene, by use of multiplex amplifiable probe hybridization. Am J Hum Genet. 2002; 71:365–374.
Article15. Gatta V, Scarciolla O, Gaspari AR, Palka C, De Angelis MV, Di Muzio A, et al. Identification of deletions and duplications of the DMD gene in affected males and carrier females by multiple ligation probe amplification (MLPA). Hum Genet. 2005; 117:92–98.
Article16. Lalic T, Vossen RH, Coffa J, Schouten JP, Guc-Scekic M, Radivojevic D, et al. Deletion and duplication screening in the DMD gene using MLPA. Eur J Hum Genet. 2005; 13:1231–1234.
Article17. Hwa HL, Chang YY, Chen CH, Kao YS, Jong YJ, Chao MC, et al. Multiplex ligation-dependent probe amplification identification of deletions and duplications of the Duchenne muscular dystrophy gene in Taiwanese subjects. J Formos Med Assoc. 2007; 106:339–346.
Article18. Uwineza A, Hitayezu J, Murorunkwere S, Ndinkabandi J, Kalala Malu CK, Caberg JH, et al. Genetic diagnosis of Duchenne and Becker muscular dystrophy using multiplex ligation-dependent probe amplification in Rwandan patients. J Trop Pediatr. 2014; 60:112–117.
Article19. Chen C, Ma H, Zhang F, Chen L, Xing X, Wang S, et al. Screening of Duchenne muscular dystrophy (DMD) mutations and investigating its mutational mechanism in Chinese patients. PLoS One. 2014; 9:e108038.
Article20. Lee BL, Nam SH, Lee JH, Ki CS, Lee M, Lee J. Genetic analysis of dystrophin gene for affected male and female carriers with Duchenne/Becker muscular dystrophy in Korea. J Korean Med Sci. 2012; 27:274–280.
Article21. Schouten JP, McElgunn CJ, Waaijer R, Zwijnenburg D, Diepvens F, Pals G. Relative quantification of 40 nucleic acid sequences by multiplex ligation-dependent probe amplification. Nucleic Acids Res. 2002; 30:e57.
Article22. Manjunath M, Kiran P, Preethish-Kumar V, Nalini A, Singh RJ, Gayathri N. A comparative study of mPCR, MLPA, and muscle biopsy results in a cohort of children with Duchenne muscular dystrophy: a first study. Neurol India. 2015; 63:58–62.
Article23. Prior TW, Bridgeman SJ. Experience and strategy for the molecular testing of Duchenne muscular dystrophy. J Mol Diagn. 2005; 7:317–326.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Novel Non-contiguous Duplications in the DMD Gene in Five Patients with Duchenne Muscular Dystrophy
- Female Carriers of Duchenne Muscular Dystrophy
- Evaluation of Multiplex PCR Assay Using Dual Priming Oligonucleotide System for Detection Mutation in the Duchenne Muscular Dystrophy Gene
- Three Cases of Manifesting Female Carriers in Patients with Duchenne Muscular Dystrophy
- Clinical Usefulness of Molecular Diagnosis in Dystrophin Gene Mutations Using the Multiplex Ligation-dependent Probe Amplification (MLPA) Method