Yonsei Med J.
2017 Jul;58(4):823-828. 10.3349/ymj.2017.58.4.823.
A Combination of Short and Simple Surfactant Protein B and C Analogues as a New Synthetic Surfactant: In Vitro and Animal Experiments
- Affiliations
-
- 1Department of Pediatrics, Kyung Hee University School of Medicine, Seoul, Korea. baecw@khnmc.or.kr
- KMID: 2419090
- DOI: http://doi.org/10.3349/ymj.2017.58.4.823
Abstract
- PURPOSE
Pulmonary surfactants for preterm infants contain mostly animal-derived surfactant proteins (SPs), which are essential for lowering surface tension. We prepared artificial pulmonary surfactants using synthetic human SP analogs and performed in vitro and in vivo experiments.
MATERIALS AND METHODS
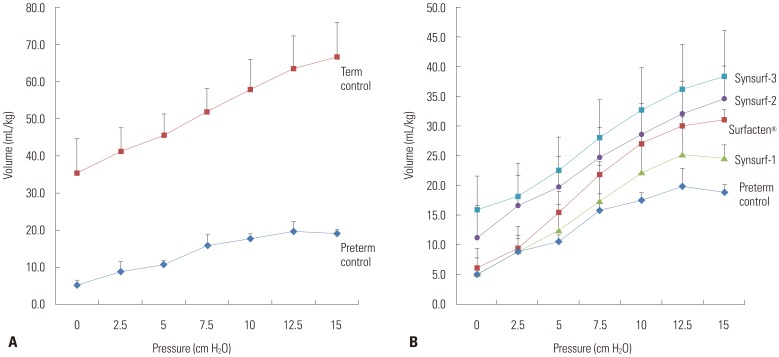
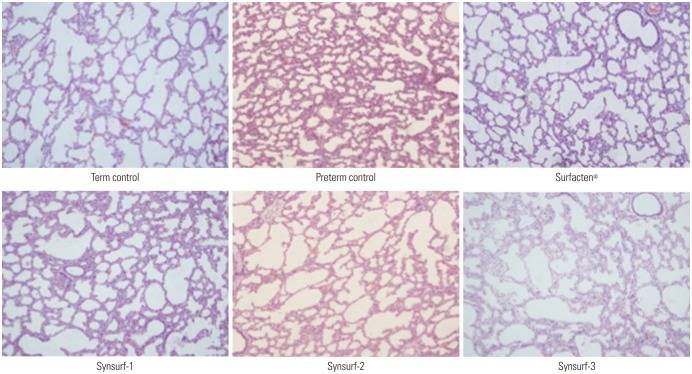
We synthesized peptide analogues that resemble human SP-B (RMLPQLVCRLVLRCSMD) and SP-C (CPVHLKRLLLLLLLLLLLLLLLL). Dipalmitoylphosphatidylcholine (DPPC), phosphatidylglycerol (PG), and palmitic acid (PA) were added and mixed in lyophilized to render powdered surfactant. Synsurf-1 was composed of DPPC:PG:PA:SP-B (75:25:10:3, w/w); Synsurf-2 was composed of DPPC:PG:PA:SP-C (75:25:10:3, w/w); and Synsurf-3 was composed of DPPC:PG:PA:SP-B:SP-C (75:25:10:3:3, w/w). We performed in vitro study to compare the physical characteristics using pulsating bubble surfactometer and modified Wilhelmy balance test. Surface spreading and adsorption test of the surfactant preparations were measured. In vivo test was performed using term and preterm rabbit pups. Pressure-volume curves were generated during the deflation phase. Histologic findings were examined.
RESULTS
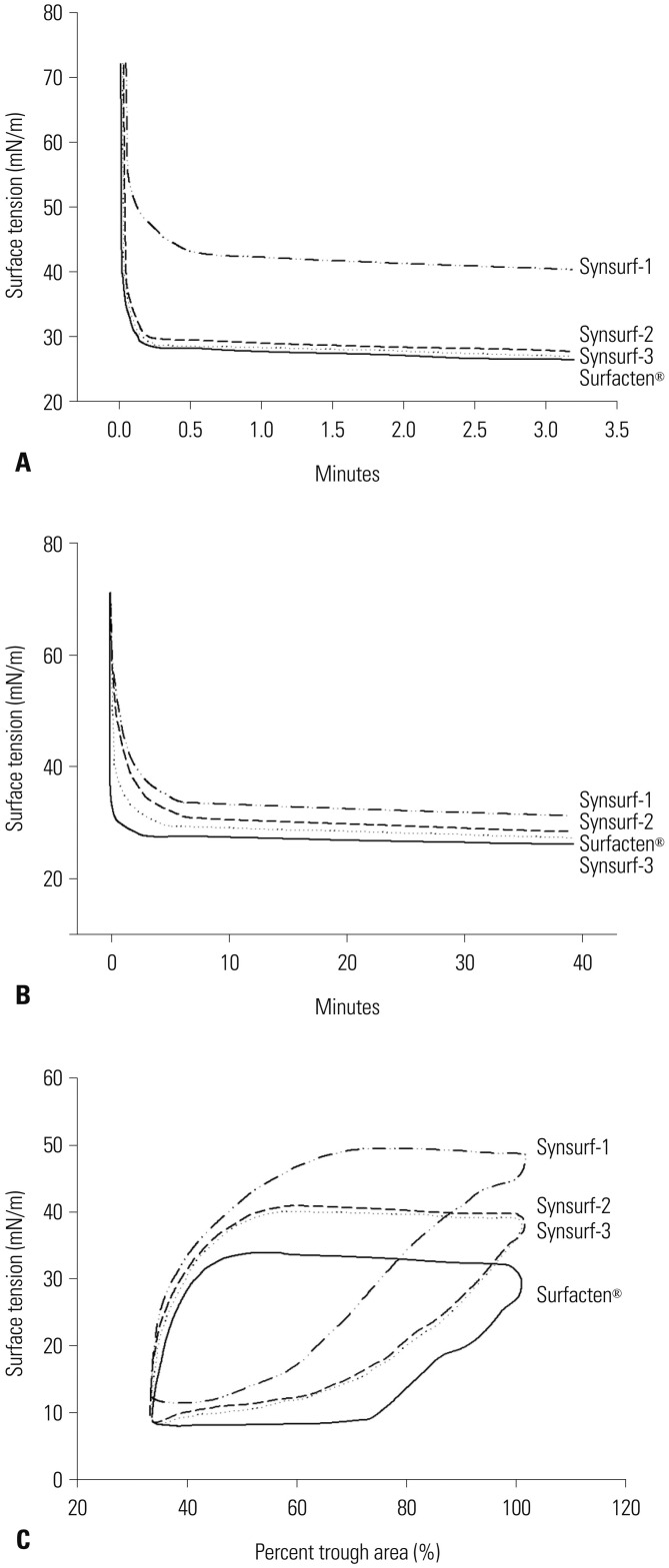
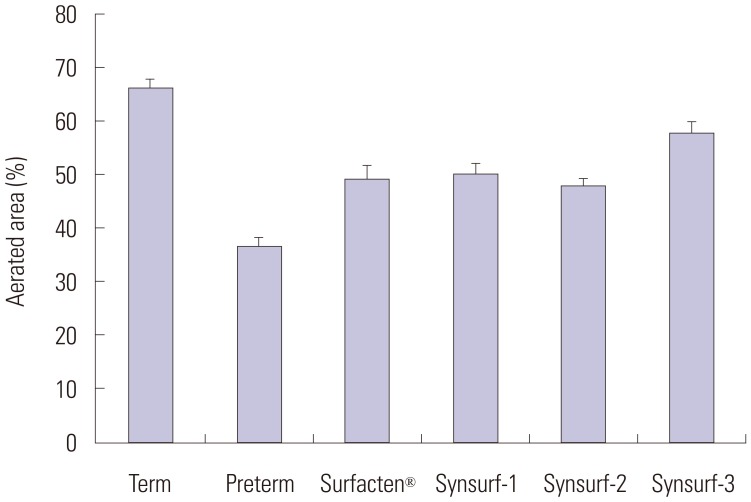
Pulsating bubble surfactometer readings revealed following minimum and maximum surface tension (mN/m) at 5 minutes: Surfacten® (5.5±0.4, 32.8±1.6), Synsurf-1 (16.7±0.6, 28.7±1.5), Synsurf-2 (7.9±1.0, 33.1±1.6), and Synsurf-3 (7.1±0.8, 34.5±1.0). Surface spreading rates were as follows: Surfacten® (27 mN/m), Synsurf-1 (43 mN/m), Synsurf-2 (27 mN/m), and Synsurf-3 (27 mN/m). Surface adsorption rate results were as follows: Surfacten® (28 mN/m), Synsurf-1 (35 mN/m), Synsurf-2 (29 mN/m), and Synsurf-3 (27 mN/m). The deflation curves were best for Synsurf-3; those for Synsurf-2 were better than those for Surfacten®. Synsurf-1 was the worst surfactant preparation. Microscopic examination showed the largest aerated area of the alveoli in the Synsurf-3 group, followed by Synsurf-1 and Surfacten®; Synsurf-2 was the smallest.
CONCLUSION
Synsurf-3 containing both SP-B and SP-C synthetic analogs showed comparable and better efficacy than commercially used Surfacten® in lowering surface tension, pressure-volume curves, and tissue aerated area of the alveoli.
Keyword
MeSH Terms
-
Animals
Animals, Newborn
Birth Weight
Emulsions
Humans
Lung/drug effects
Pressure
Pulmonary Surfactant-Associated Protein B/*pharmacology
Pulmonary Surfactant-Associated Protein C/*pharmacology
Rabbits
Surface Properties
Emulsions
Pulmonary Surfactant-Associated Protein B
Pulmonary Surfactant-Associated Protein C
Figure
Cited by 1 articles
-
History of Pulmonary Surfactant Replacement Therapy for Neonatal Respiratory Distress Syndrome in Korea
Chong-Woo Bae, Chae Young Kim, Sung-Hoon Chung, Yong-Sung Choi
J Korean Med Sci. 2019;34(25):. doi: 10.3346/jkms.2019.34.e175.
Reference
-
1. Jobe AH, Hillman N, Polglase G, Kramer BW, Kallapur S, Pillow J. Injury and inflammation from resuscitation of the preterm infant. Neonatology. 2008; 94:190–196. PMID: 18832854.
Article2. Miedema M, de Jongh FH, Frerichs I, van Veenendaal MB, van Kaam AH. Changes in lung volume and ventilation during surfactant treatment in ventilated preterm infants. Am J Respir Crit Care Med. 2011; 184:100–105. PMID: 21493733.
Article3. Fujiwara T, Maeta H, Chida S, Morita T, Watabe Y, Abe T. Artificial surfactant therapy in hyaline-membrane disease. Lancet. 1980; 1:55–59. PMID: 6101413.
Article4. Soll R, Ozek E. Multiple versus single doses of exogenous surfactant for the prevention or treatment of neonatal respiratory distress syndrome. Cochrane Database Syst Rev. 2009; (1):CD000141. PMID: 19160177.
Article5. Kim SM, Park YJ, Chung SH, Choi YS, Kim CH, Bae CW. Early prophylactic versus late selective use of surfactant for respiratory distress syndrome in very preterm infants: a collaborative study of 53 multi-center trials in Korea. J Korean Med Sci. 2014; 29:1126–1131. PMID: 25120324.
Article6. Zhu Y, Miller TL, Chidekel A, Shaffer TH. KL4-surfactant (Lucinactant) protects human airway epithelium from hyperoxia. Pediatr Res. 2008; 64:154–158. PMID: 18391844.
Article7. Moya F, Sinha S, Gadzinowski J, D'Agostino R, Segal R, Guardia C, et al. One-year follow-up of very preterm infants who received lucinactant for prevention of respiratory distress syndrome: results from 2 multicenter randomized, controlled trials. Pediatrics. 2007; 119:e1361–e1370. PMID: 17533176.
Article8. Lu KW, Taeusch HW. Combined effects of polymers and KL(4) peptide on surface activity of pulmonary surfactant lipids. Biochim Biophys Acta. 2010; 1798:1129–1134. PMID: 20188696.
Article9. Soll RF, Blanco F. Natural surfactant extract versus synthetic surfactant for neonatal respiratory distress syndrome. Cochrane Database Syst Rev. 2001; (2):CD000144. PMID: 11405951.
Article10. Kallberg Y, Gustafsson M, Persson B, Thyberg J, Johansson J. Prediction of amyloid fibril-forming proteins. J Biol Chem. 2001; 276:12945–12950. PMID: 11134035.
Article11. Robertson B, van Golde LMG. Pulmonary surfactant: from molecular biology to clinical practice. Amsterdam: Elsevier Science Publishers;1992. p. 561–592.12. Moya F. Synthetic surfactants: where are we? Evidence from randomized, controlled clinical trials. J Perinatol. 2009; 29(Suppl 2):S23–S28. PMID: 19399006.
Article13. Seger N, Soll R. Animal derived surfactant extract for treatment of respiratory distress syndrome. Cochrane Database Syst Rev. 2009; (2):CD007836. PMID: 19370695.
Article14. Walther FJ, Waring AJ, Sherman MA, Zasadzinski JA, Gordon LM. Hydrophobic surfactant proteins and their analogues. Neonatology. 2007; 91:303–310. PMID: 17575474.
Article15. Otsubo E, Takei T, Nomura M. Synthesis, purification and surface activities of the human pulmonary surfactant protein-C (SP-C) analogue, SP-CL16 (6-28). Biol Pharm Bull. 2001; 24:1362–1365. PMID: 11767102.16. Bae CW, Chung SH, Choi YS. Development of a synthetic surfactant using a surfactant protein-C peptide analog: in vitro studies of surface physical properties. Yonsei Med J. 2016; 57:203–208. PMID: 26632402.
Article17. Otsubo E, Takei T. Effects of the human pulmonary surfactant protein-C (SP-C), SP-CL16 (6-28) on surface activities of surfactants with various phospholipids. Biol Pharm Bull. 2002; 25:1303–1306. PMID: 12392083.18. Johansson J. Membrane properties and amyloid fibril formation of lung surfactant protein C. Biochem Soc Trans. 2001; 29(Pt 4):601–606. PMID: 11498036.
Article19. Almlén A, Walther FJ, Waring AJ, Robertson B, Johansson J, Curstedt T. Synthetic surfactant based on analogues of SP-B and SP-C is superior to single-peptide surfactants in ventilated premature rabbits. Neonatology. 2010; 98:91–99. PMID: 20110733.
Article20. Seehase M, Collins JJ, Kuypers E, Jellema RK, Ophelders DR, Ospina OL, et al. New surfactant with SP-B and C analogs gives survival benefit after inactivation in preterm lambs. PLoS One. 2012; 7:e47631. PMID: 23091635.
Article21. Bae CW, Choi YM, Kang JH, Lee SC, Kim KL, Hahm KS. A study of surface physical properties of new surfactant using synthetic peptides of surfactant protein-B. Korean J Pediatr. 2000; 43:1059–1067.22. Nogee LM, de Mello DE, Dehner LP, Colten HR. Brief report: deficiency of pulmonary surfactant protein B in congenital alveolar proteinosis. N Engl J Med. 1993; 328:406–410. PMID: 8421459.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The use of artificial pulmonary surfactant in neonatal respiratory distress
- New Synthetic Surfactants for Neonates
- Erratum to “Development of a Synthetic Surfactant Using a Surfactant Protein-C Peptide Analog: In Vitro Studies of Surface Physical Properties†by Bae CW, et al. (Yonsei Med J 2016;57(1):203–208.)
- Surfactant preparations for preterm infants with respiratory distress syndrome: past, present, and future
- Preparation and in vitro Physical Activities of Crude Natural Surfactant and Artificial Pulmonary Surfactant Containing Synthetic Peptide and Phospholipid Mixtures