Cancer Res Treat.
2018 Jul;50(3):861-871. 10.4143/crt.2017.237.
Combination of Tumor Volume and Epstein-Barr Virus DNA Improved Prognostic Stratification of Stage II Nasopharyngeal Carcinoma in the Intensity Modulated Radiotherapy Era: A Large-Scale Cohort Study
- Affiliations
-
- 1Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, China. maihq@sysucc.org.cn
- 2Department of Nasopharyngeal Carcinoma, Sun Yat-sen University Cancer Center, Guangzhou, China.
- 3ZhongShan School of Medicine, Sun Yat-sen University, Guangzhou, China.
- KMID: 2417875
- DOI: http://doi.org/10.4143/crt.2017.237
Abstract
- PURPOSE
Little is known about combination of the circulating Epstein-Barr viral (EBV) DNA and tumor volume in prognosis of stage II nasopharyngeal carcinoma (NPC) patients in the intensity modulated radiotherapy (IMRT) era. We conducted this cohort study to evaluate the prognostic values of combining these two factors.
MATERIALS AND METHODS
By Kaplan-Meier, we compare the differences of survival curves between 385 patients with different EBV DNA or tumor volume levels, or with the combination of two biomarkers mentioned above.
RESULTS
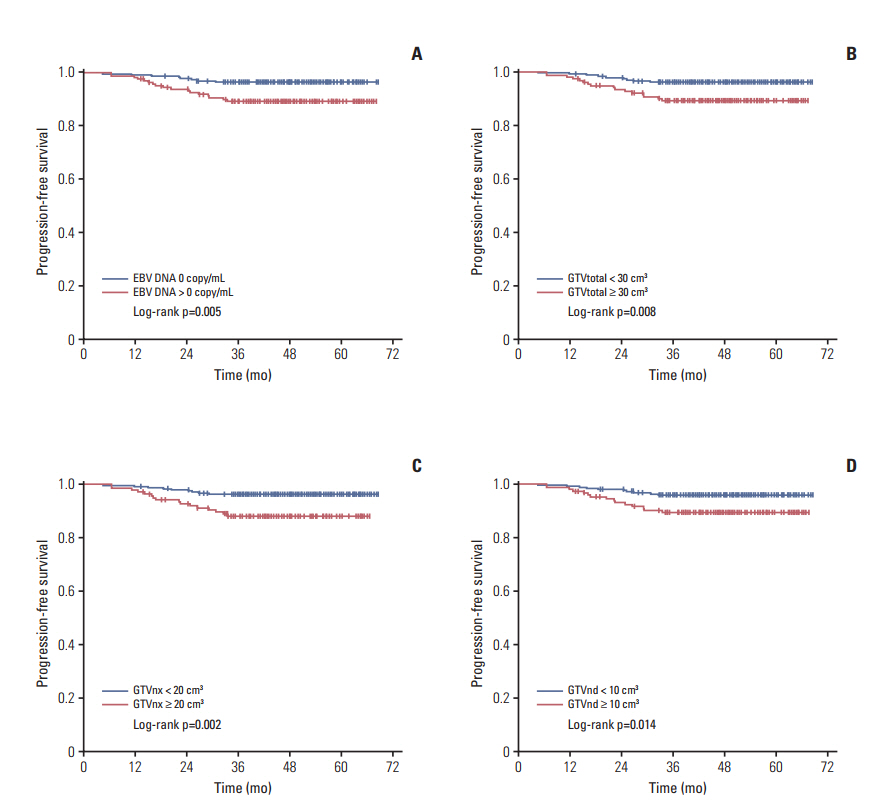
Gross tumor volume of cervical lymph nodes (GTVnd, p < 0.001) and total tumor volume (GTVtotal, p < 0.001) were both closely related to pretreatment EBV DNA, while gross tumor volume of nasopharynx (GTVnx, p=0.047) was weakly related to EBV DNA. EBV DNA was significantly correlated with progress-free survival (PFS, p=0.005), locoregional-free survival (LRFS, p=0.039), and distant metastasis-free survival (DMFS, p=0.017), while GTVtotal, regardless of GTVnx and GTVnd, had a significant correlation with PFS and LRFS. The p-values of GTVtotal for PFS and LRFS were 0.008 and 0.001, respectively. According to GTVtotal and pretreatment EBV DNA level, patients were divided into a low-risk group (EBV DNA 0 copy/mL, GTVtotal < 30 cm³; EBV DNA 0 copy/mL, GTVtotal ≥ 30 cm³; or EBV DNA > 0 copy/mL, GTVtotal < 30 cm³) and a high-risk group (EBV DNA > 0 copy/mL, GTVtotal ≥ 30 cm³). When patients in the low-risk group were compared with those in the high-risk group, 3-year PFS (p=0.003), LRFS (p=0.010), and DMFS (p=0.031) rates were statistically significant.
CONCLUSION
Pretreatment plasma EBV DNA and tumor volume were both closely correlated with prognosis of stage II NPC patients in the IMRT era. Combination of EBV DNA and tumor volume can refine prognosis and indicate for clinical therapy.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Choa G. Nasopharyngeal carcinoma. Some observations on the clinical features and technique of examination. Pac Med Surg. 1967; 75:172–4.2. Cao SM, Simons MJ, Qian CN. The prevalence and prevention of nasopharyngeal carcinoma in China. Chin J Cancer. 2011; 30:114–9.
Article3. Lee AW, Ng WT, Chan YH, Sze H, Chan C, Lam TH. The battle against nasopharyngeal cancer. Radiother Oncol. 2012; 104:272–8.
Article4. Wee JT, Ha TC, Loong SL, Qian CN. Is nasopharyngeal cancer really a "Cantonese cancer"? Chin J Cancer. 2010; 29:517–26.
Article5. Chen QY, Wen YF, Guo L, Liu H, Huang PY, Mo HY, et al. Concurrent chemoradiotherapy vs radiotherapy alone in stage II nasopharyngeal carcinoma: phase III randomized trial. J Natl Cancer Inst. 2011; 103:1761–70.
Article6. Lai SZ, Li WF, Chen L, Luo W, Chen YY, Liu LZ, et al. How does intensity-modulated radiotherapy versus conventional two-dimensional radiotherapy influence the treatment results in nasopharyngeal carcinoma patients? Int J Radiat Oncol Biol Phys. 2011; 80:661–8.7. Peng G, Wang T, Yang KY, Zhang S, Zhang T, Li Q, et al. A prospective, randomized study comparing outcomes and toxicities of intensity-modulated radiotherapy vs. conventional two-dimensional radiotherapy for the treatment of nasopharyngeal carcinoma. Radiother Oncol. 2012; 104:286–93.
Article8. Su Z, Mao YP, Tang J, Lan XW, OuYang PY, Xie FY. Long-term outcomes of concurrent chemoradiotherapy versus radiotherapy alone in stage II nasopharyngeal carcinoma treated with IMRT: a retrospective study. Tumour Biol. 2016; 37:4429–38.
Article9. Zhang F, Zhang Y, Li WF, Liu X, Guo R, Sun Y, et al. Efficacy of concurrent chemotherapy for intermediate risk NPC in the intensity-modulated radiotherapy era: a propensity-matched analysis. Sci Rep. 2016; 5:17378.
Article10. Ji MF, Huang QH, Yu X, Liu Z, Li X, Zhang LF, et al. Evaluation of plasma Epstein-Barr virus DNA load to distinguish nasopharyngeal carcinoma patients from healthy high-risk populations in Southern China. Cancer. 2014; 120:1353–60.
Article11. Wei WI, Sham JS. Nasopharyngeal carcinoma. Lancet. 2005; 365:2041–54.
Article12. Mutirangura A, Pornthanakasem W, Theamboonlers A, Sriuranpong V, Lertsanguansinchi P, Yenrudi S, et al. Epstein-Barr viral DNA in serum of patients with nasopharyngeal carcinoma. Clin Cancer Res. 1998; 4:665–9.13. Chan AT, Lo YM, Zee B, Chan LY, Ma BB, Leung SF, et al. Plasma Epstein-Barr virus DNA and residual disease after radiotherapy for undifferentiated nasopharyngeal carcinoma. J Natl Cancer Inst. 2002; 94:1614–9.
Article14. Lo YM, Chan LY, Lo KW, Leung SF, Zhang J, Chan AT, et al. Quantitative analysis of cell-free Epstein-Barr virus DNA in plasma of patients with nasopharyngeal carcinoma. Cancer Res. 1999; 59:1188–91.15. Lin JC, Wang WY, Chen KY, Wei YH, Liang WM, Jan JS, et al. Quantification of plasma Epstein-Barr virus DNA in patients with advanced nasopharyngeal carcinoma. N Engl J Med. 2004; 350:2461–70.
Article16. Chen MK, Chen TH, Liu JP, Chang CC, Chie WC. Better prediction of prognosis for patients with nasopharyngeal carcinoma using primary tumor volume. Cancer. 2004; 100:2160–6.
Article17. Chua DT, Sham JS, Kwong DL, Tai KS, Wu PM, Lo M, et al. Volumetric analysis of tumor extent in nasopharyngeal carcinoma and correlation with treatment outcome. Int J Radiat Oncol Biol Phys. 1997; 39:711–9.
Article18. Ma J, Liu L, Tang L, Zong J, Lin A, Lu T, et al. Retropharyngeal lymph node metastasis in nasopharyngeal carcinoma: prognostic value and staging categories. Clin Cancer Res. 2007; 13:1445–52.
Article19. Tang LQ, Chen QY, Fan W, Liu H, Zhang L, Guo L, et al. Prospective study of tailoring whole-body dual-modality[18F]fluorodeoxyglucose positron emission tomography/computed tomography with plasma Epstein-Barr virus DNA for detecting distant metastasis in endemic nasopharyngeal carcinoma at initial staging. J Clin Oncol. 2013; 31:2861–9.20. Zhao C, Han F, Lu LX, Huang SM, Lin CG, Deng XW, et al. Intensity modulated radiotherapy for local-regional advanced nasopharyngeal carcinoma. Ai Zheng. 2004; 23(11 Suppl):1532–7.21. Chen WS, Lu JX, Ye SX. Application of combined detection of EBV VCA-IgA and EA-IgA antibodies in the diagnosis of NPC. J Trop Med. 2010; 10:434–6.22. Cho W. Molecular biomarker discovery and progress in nasopharyngeal carcinoma. Cancer Res. 2007; 67(9 Suppl):Abstr LB-49.23. Chen M, Yin L, Wu J, Gu JJ, Jiang XS, Wang DJ, et al. Impact of plasma Epstein-Barr virus-DNA and tumor volume on prognosis of locally advanced nasopharyngeal carcinoma. Biomed Res Int. 2015; 2015:617949.
Article24. Chen WH, Tang LQ, Guo SS, Chen QY, Zhang L, Liu LT, et al. Prognostic value of plasma Epstein-Barr virus DNA for local and regionally advanced nasopharyngeal carcinoma treated with cisplatin-based concurrent chemoradiotherapy in intensity-modulated radiotherapy era. Medicine (Baltimore). 2016; 95:e2642.
Article25. Lo YM, Leung SF, Chan LY, Chan AT, Lo KW, Johnson PJ, et al. Kinetics of plasma Epstein-Barr virus DNA during radiation therapy for nasopharyngeal carcinoma. Cancer Res. 2000; 60:2351–5.26. Guo Q, Lu T, Lin S, Zong J, Chen Z, Cui X, et al. Long-term survival of nasopharyngeal carcinoma patients with Stage II in intensity-modulated radiation therapy era. Jpn J Clin Oncol. 2016; 46:241–7.
Article27. Ma BB, King A, Lo YM, Yau YY, Zee B, Hui EP, et al. Relationship between pretreatment level of plasma Epstein-Barr virus DNA, tumor burden, and metabolic activity in advanced nasopharyngeal carcinoma. Int J Radiat Oncol Biol Phys. 2006; 66:714–20.
Article28. Hou X, Zhao C, Guo Y, Han F, Lu LX, Wu SX, et al. Different clinical significance of pre- and post-treatment plasma Epstein-Barr virus DNA load in nasopharyngeal carcinoma treated with radiotherapy. Clin Oncol (R Coll Radiol). 2011; 23:128–33.
Article29. Ma BB, Mo FK, Chan AT, Hui EP, Leung SF, Lo YM, et al. The prognostic significance of tumor vascular invasion and its association with plasma Epstein-Barr virus DNA, tumor volume and metabolic activity in locoregionally advanced nasopharyngeal carcinoma. Oral Oncol. 2008; 44:1067–72.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Prognostic Value of Serum Epstein-Barr Virus Antibodies and Their Correlation with TNM Classification in Patients with Locoregionally Advanced Nasopharyngeal Carcinoma
- Current Management Strategy of Nasopharyngeal Carcinoma
- A Case of Epstein-Barr Virus-Positive Diffuse Large B-Cell Lymphoma Occurring in Thyroid Gland
- Individualized Concurrent Chemotherapy for Patients with Stage III-IVa Nasopharyngeal Carcinoma Receiving Neoadjuvant Chemotherapy Combined with Definitive Intensity-Modulated Radiotherapy
- Tumor volume/metabolic information can improve the prognostication of anatomy based staging system for nasopharyngeal cancer? Evaluation of the 8th edition of the AJCC/UICC staging system for nasopharyngeal cancer