J Pathol Transl Med.
2018 Jul;52(4):257-261. 10.4132/jptm.2018.05.08.
Malignant Pleural Effusion from Metastatic Prostate Cancer: A Case Report with Unusual Cytologic Findings
- Affiliations
-
- 1Department of Hospital Pathology, Seoul St. Mary’s Hospital, Seoul, Korea. leekyoyo@catholic.ac.kr
- 2Department of Hospital Pathology, Yeouido St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 2417812
- DOI: http://doi.org/10.4132/jptm.2018.05.08
Abstract
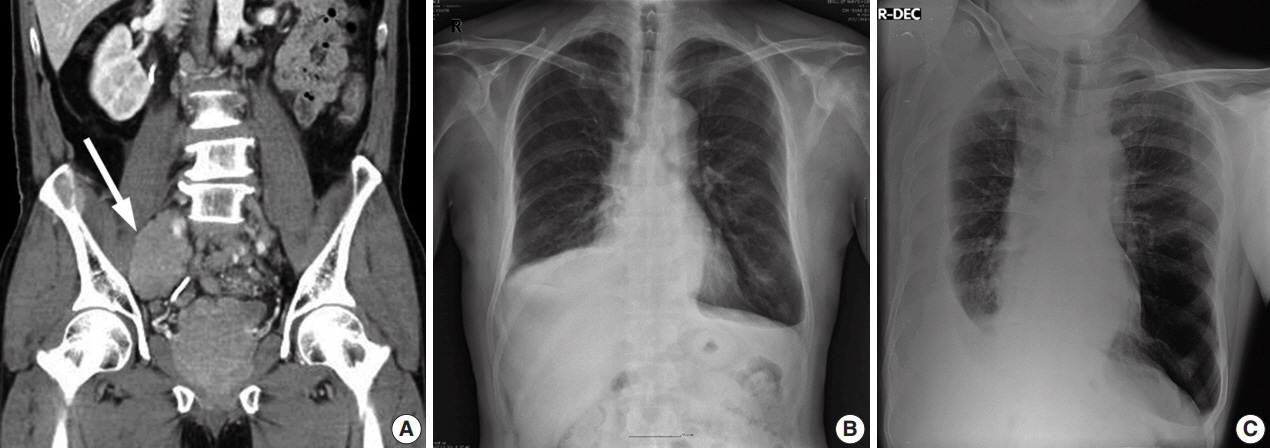
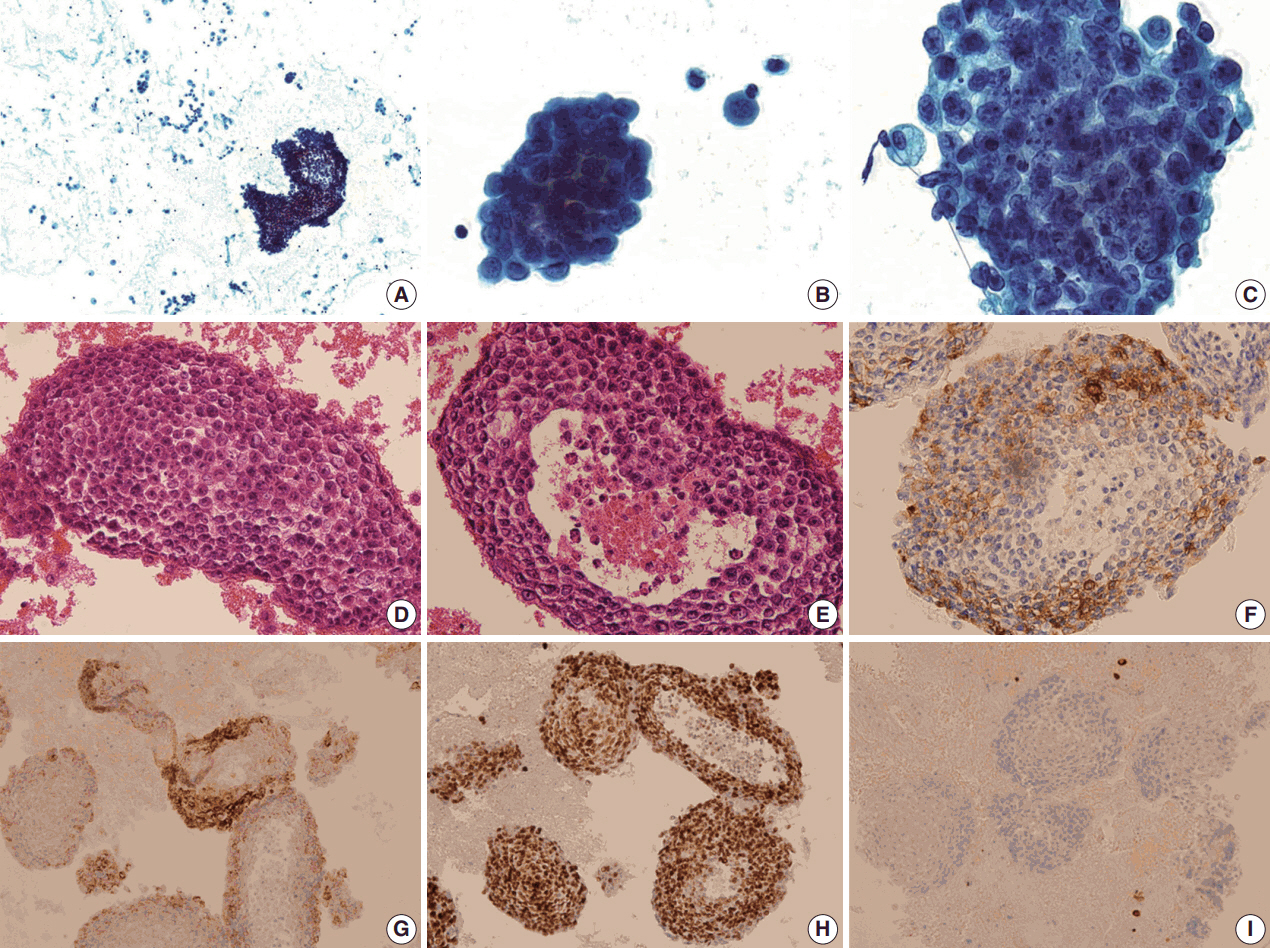
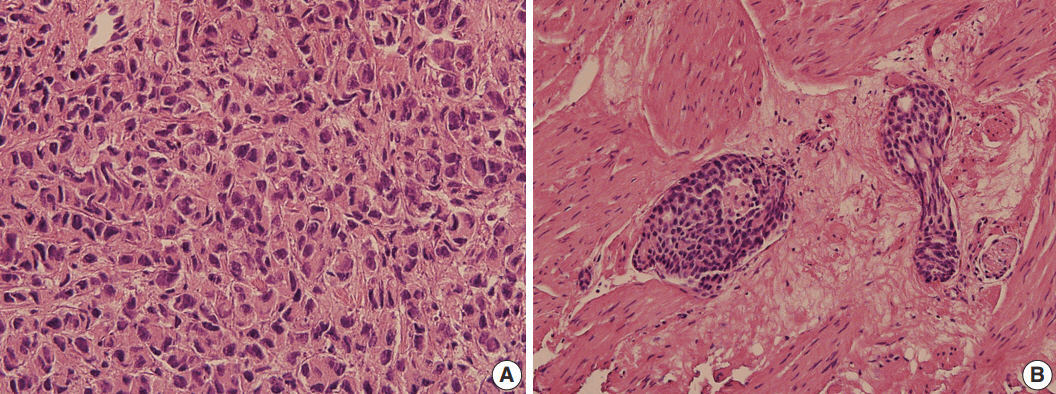
- We present a case of 55-year-old man who complained of dyspnea and sputum for a month. He was an ex-smoker with a history of prostate cancer and pulmonary tuberculosis. Chest radiographs revealed bilateral pleural effusions of a small to moderate amount. Pigtail catheters were inserted for drainage. The pleural fluid consisted of large clusters and tightly cohesive groups of malignant cells, which however could not be ascribed to prostate cancer with certainty. We performed immunocytochemical panel studies to determine the origin of cancer metastasis. The immunostaining results were positive for prostate-specific antigen, alpha-methylacyl-coenzyme A racemase, and Nkx 3.1, consistent with prostate cancer. Pleural effusion associated with prostate cancer is rare. To our knowledge, this is the first case report in Korea to describe cytologic features of malignant pleural effusion associated with prostate cancer.
MeSH Terms
Figure
Reference
-
1. Egan AM, McPhillips D, Sarkar S, Breen DP. Malignant pleural effusion. QJM. 2014; 107:179–84.
Article2. Thomas JM, Musani AI. Malignant pleural effusions: a review. Clin Chest Med. 2013; 34:459–71.3. Lim MH, Garrettc J, Mowlem L, Yap E. Diagnosing malignant pleural effusions: how do we compare? N Z Med J. 2013; 126:42–8.4. Bubendorf L, Schopfer A, Wagner U, et al. Metastatic patterns of prostate cancer: an autopsy study of 1,589 patients. Hum Pathol. 2000; 31:578–83.
Article5. Vinjamoori AH, Jagannathan JP, Shinagare AB, et al. Atypical metastases from prostate cancer: 10-year experience at a single institution. AJR Am J Roentgenol. 2012; 199:367–72.
Article6. Ani I, Costaldi M, Abouassaly R. Metastatic prostate cancer with malignant ascites: a case report and literature review. Can Urol Assoc J. 2013; 7:E248–50.
Article7. Bajpai PA, Joshi P, Joseph D, Bajpai A. A case of recurrent massive right sided pleural effusion-an unusual presentation of carcinoma prostate. Lung India. 2014; 31:423–4.
Article8. dos Santos VM, Zembrzuski MM, Gouvea IP, Nery NS, dos Santos LA. Massive pleural effusion due to metastasis of prostate cancer. West Indian Med J. 2011; 60:690–3.9. Knight JC, Ray MA, Benzaquen S. Malignant pleural effusion from prostate adenocarcinoma. Respir Med Case Rep. 2014; 13:24–5.
Article10. Saif MW. Malignant ascites associated with carcinoma of the prostate. J Appl Res. 2005; 5:305–11.11. Saif MW, Figg WD, Hewitt S, Brosky K, Reed E, Dahut W. Malignant ascites as only manifestation of metastatic prostate cancer. Prostate Cancer Prostatic Dis. 1999; 2:290–3.
Article12. Mai KT, Roustan Delatour NL, Assiri A, Al-Maghrabi H. Secondary prostatic adenocarcinoma: a cytopathological study of 50 cases. Diagn Cytopathol. 2007; 35:91–5.
Article13. Renshaw AA, Nappi D, Cibas ES. Cytology of metastatic adenocarcinoma of the prostate in pleural effusions. Diagn Cytopathol. 1996; 15:103–7.
Article14. Carrascosa M, Perez-Castrillon JL, Mendez MA, Cillero L, Valle R. Malignant pleural effusion from prostatic adenocarcinoma resolved with hormonal therapy. Chest. 1994; 105:1577–8.
Article15. Shimizu T, Shibata Y, Uchida T, Satoh J. Severe flare-up in a prostate cancer patient treated with luteinizing hormone-releasing hormone analogue depot. Hinyokika Kiyo. 1993; 39:953–5.16. Mestitz H, Pierce RJ, Holmes PW. Intrathoracic manifestations of disseminated prostatic adenocarcinoma. Respir Med. 1989; 83:161–6.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Malignant Pleural Effusion Treated by Chemical Pleurodesis in Ovarian Carcinoma
- Composite Epithelioid Hemangioendothelioma in Pleural Effusion Mimicking Metastatic Adenocarcinoma: Cytologic and Immunocytochemical Findings
- Cytologic Diagnosis of Malignant Pleural Effusion in Multiple Myeloma: Two Case Reports
- Four Cases of Malignant Pleural Effusion in Patients with Papillary Thyroid Carcinoma
- Clinical Evaluation of Exudative Pleural Effusion