Korean J Ophthalmol.
2018 Apr;32(2):147-153. 10.3341/kjo.2017.0068.
Distribution of Interleukin-22–secreting Immune Cells in Conjunctival Associated Lymphoid Tissue
- Affiliations
-
- 1Department of Ophthalmology, Seoul National University College of Medicine, Seoul, Korea. kmk9@snu.ac.kr
- 2Laboratory of Ocular Regenerative Medicine and Immunology, Seoul Artificial Eye Center, Seoul National University Hospital Biomedical Research Institute, Seoul, Korea.
- KMID: 2408955
- DOI: http://doi.org/10.3341/kjo.2017.0068
Abstract
- PURPOSE
Interleukin (IL)-22 is a cytokine involved in epithelial cell regeneration. Currently, no research studies have analyzed the distribution of the three distinct IL-22-secreting cell populations in human or mouse conjunctiva. This study investigated the distribution of the three main populations of IL-22-secreting immune cells, αβ Th cells, γδ T cells, or innate cells (innate lymphoid cells [ILCs] or natural killer cells), in conjunctival associated lymphoid tissues (CALTs) in human and mouse models.
METHODS
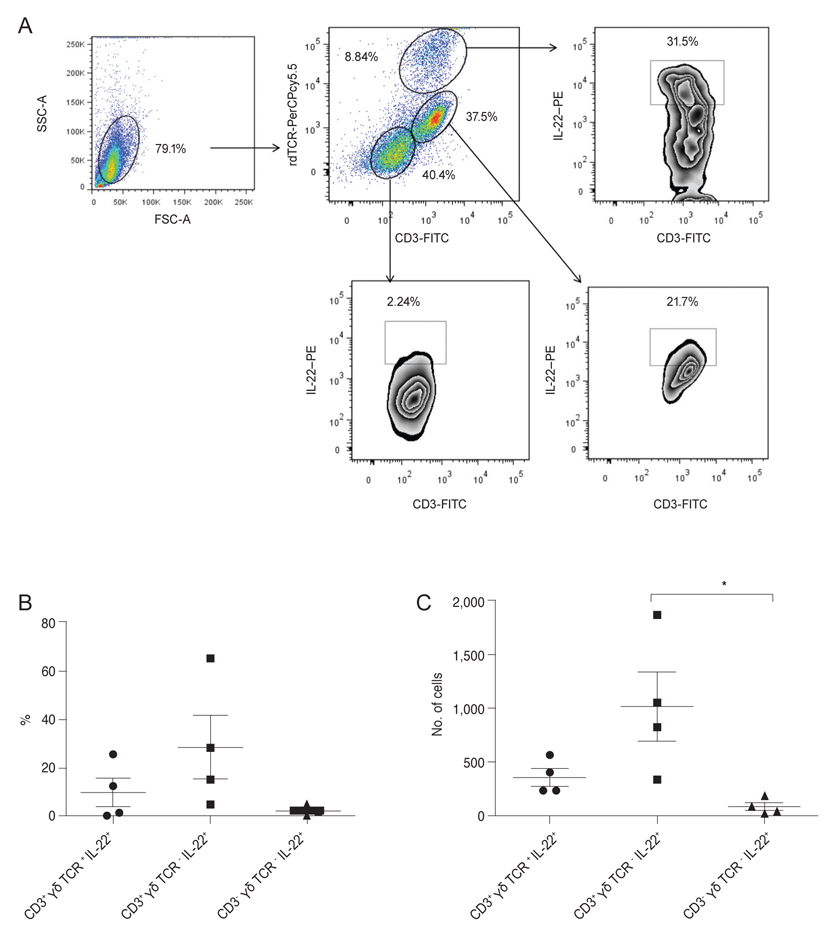
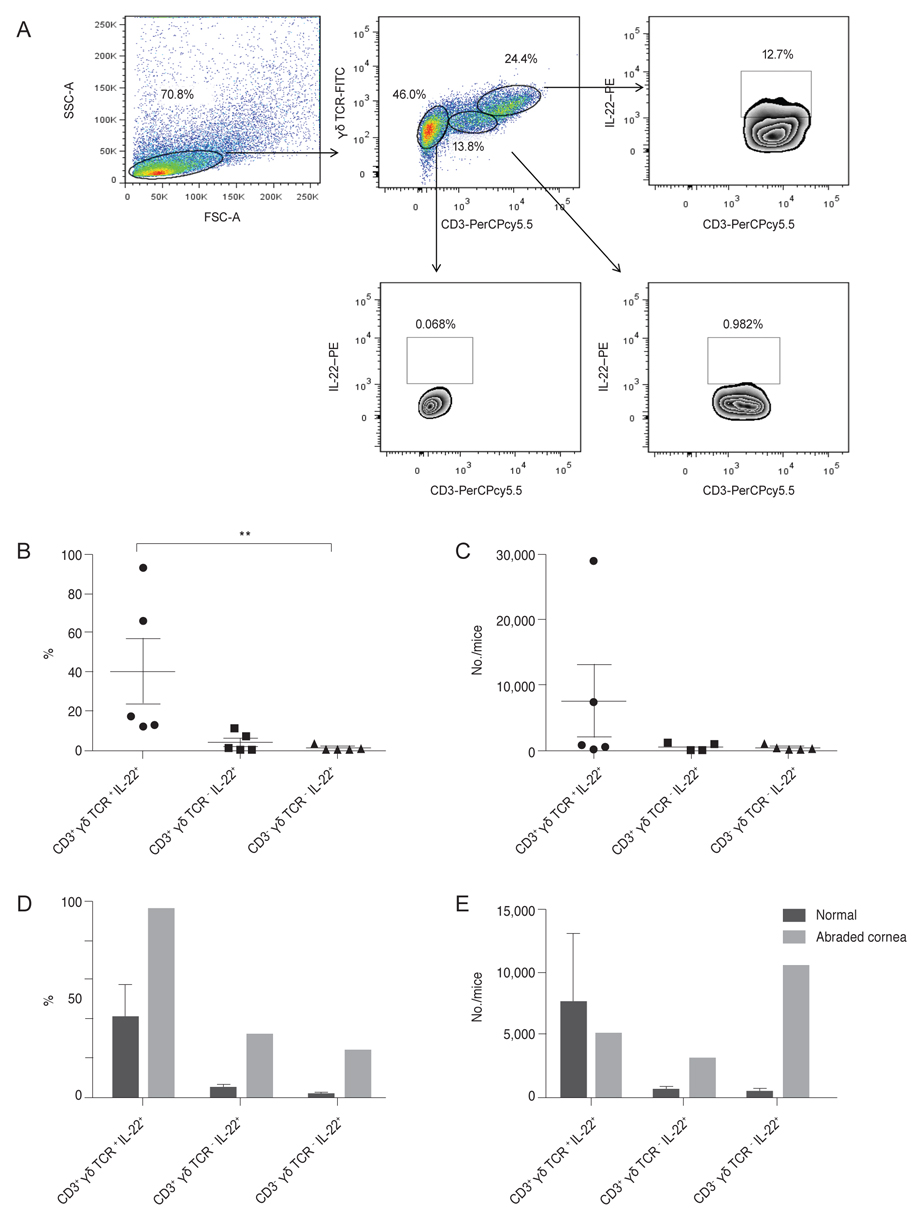
We collected discarded cadaveric bulbar conjunctival tissue specimens after preservation of the corneo-limbal tissue for keratoplasty from four enucleated eyes of the domestic donor. The bulbar conjunctiva tissue, including the cornea from normal (n = 27) or abraded (n = 4) B6 mice, were excised and pooled in RPMI 1640 media. After the lymphoid cells were gated in forward and side scattering, the αβ Th cells, γδ T cells, or innate lymphoid cells were positively or negatively gated using anti-CD3, anti-γδ TCR, and anti-IL-22 antibodies, with a FACSCanto flow cytometer.
RESULTS
In normal human conjunctiva, the percentage and number of cells were highest in αβ Th cells, followed by γδ T cells and CD3-γδ TCR - IL-22+ innate cells (presumed ILCs, pILCs) (Kruskal-Wallis test, p = 0.012). In normal mice keratoconjunctiva, the percentage and total number were highest in γδ T cells, followed by αβ Th cells and pILCs (Kruskal-Wallis test, p = 0.0004); in corneal abraded mice, the population of αβ Th cells and pILCs tended to increase.
CONCLUSIONS
This study suggests that three distinctive populations of IL-22-secreting immune cells are present in CALTs of both humans and mice, and the proportions of IL-22+αβ Th cells, γδ T cells, and pILCs in CALTs in humans might be differently distributed from those in normal mice.
Keyword
MeSH Terms
Figure
Reference
-
1. Nikoopour E, Bellemore SM, Singh B. IL-22, cell regeneration and autoimmunity. Cytokine. 2015; 74:35–42.
Article2. Zenewicz LA, Flavell RA. Recent advances in IL-22 biology. Int Immunol. 2011; 23:159–163.
Article3. Huang Y, Yang Z, Huang C, et al. γδ T cell-dependent regulatory T cells prevent the development of autoimmune keratitis. J Immunol. 2015; 195:5572–5581.
Article4. Li Z, Burns AR, Miller SB, Smith CW. CCL20, γδ T cells, and IL-22 in corneal epithelial healing. FASEB J. 2011; 25:2659–2668.
Article5. Li Z, Burns AR, Rumbaut RE, Smith CW. Gamma delta T cells are necessary for platelet and neutrophil accumulation in limbal vessels and efficient epithelial repair after corneal abrasion. Am J Pathol. 2007; 171:838–845.6. Liu Q, Smith CW, Zhang W, et al. NK cells modulate the inflammatory response to corneal epithelial abrasion and thereby support wound healing. Am J Pathol. 2012; 181:452–462.
Article7. Sedda S, Marafini I, Figliuzzi MM, et al. An overview of the role of innate lymphoid cells in gut infections and inflammation. Mediators Inflamm. 2014; 2014:235460.
Article8. Goto Y, Obata T, Kunisawa J, et al. Innate lymphoid cells regulate intestinal epithelial cell glycosylation. Science. 2014; 345:1254009.
Article9. Dudakov JA, Hanash AM, van den Brink MR. Interleukin-22: immunobiology and pathology. Annu Rev Immunol. 2015; 33:747–785.
Article10. Ji YW, Mittal SK, Hwang HS, et al. Lacrimal gland-derived IL-22 regulates IL-17-mediated ocular mucosal inflammation. Mucosal Immunol. 2017; 10:1202–1210.
Article11. O'Brien RL, Taylor MA, Hartley J, et al. Protective role of gammadelta T cells in spontaneous ocular inflammation. Invest Ophthalmol Vis Sci. 2009; 50:3266–3274.12. Misharin AV, Morales-Nebreda L, Mutlu GM, et al. Flow cytometric analysis of macrophages and dendritic cell subsets in the mouse lung. Am J Respir Cell Mol Biol. 2013; 49:503–510.
Article13. Montaldo E, Vacca P, Vitale C, et al. Human innate lymphoid cells. Immunol Lett. 2016; 179:2–8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Heterogeneity of IL-22-producing Lymphoid Tissue Inducer-like Cells in Human and Mouse
- The Role of Type 2 Innate Lymphoid Cells in Allergic Diseases
- A Case of Bilateral Conjunctival Malignant Lymphoma Misdiagnosed as Allergic Conjunctivitis
- Roles of Embryonic and Adult Lymphoid Tissue Inducer Cells in Primary and Secondary Lymphoid Tissues
- Effect of capsaicin on murine lymphocyte functions and lymphoid tissue morphology