Ann Surg Treat Res.
2018 Apr;94(4):219-221. 10.4174/astr.2018.94.4.219.
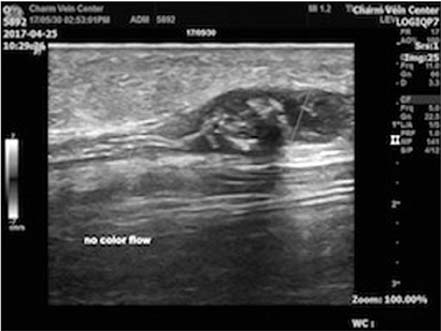
Successful use of VenaSeal system for the treatment of large great saphenous vein of 2.84-cm diameter
- Affiliations
-
- 1Department of Varicose Vein Clinic, Charm Vein Center, Seoul, Korea. charmsurgery@gmail.com
- KMID: 2407753
- DOI: http://doi.org/10.4174/astr.2018.94.4.219
Abstract
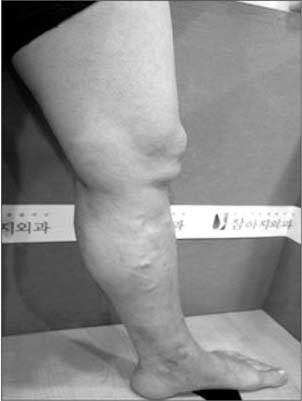
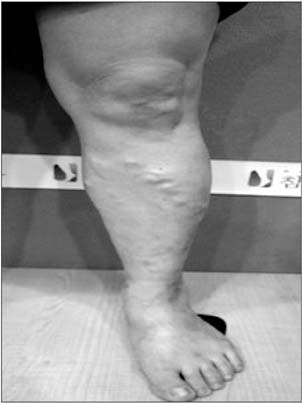
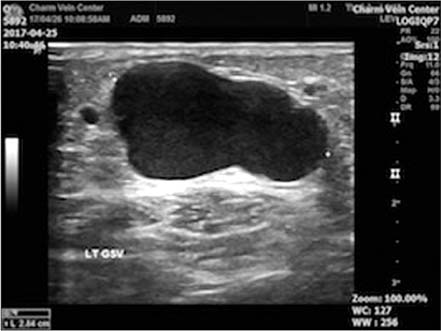
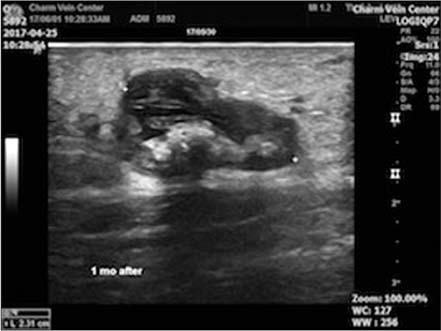
- Cyanoacrylate closure, VenaSeal system, for the treatment of incompetent saphenous vein is a new technique. We report a successful case with a large great saphenous vein of 2.84 cm in diameter.
Keyword
Figure
Cited by 1 articles
-
Hydrophilic guidewire usage under ultrasound guidance in facilitating catheter advancement during endovenous treatment of incompetent great saphenous veins
Kyosoo Hwang, Sang Woo Park, Jin Ho Hwang, Yong Wonn Kwon, Jeeyoung Min, Hyemin Jang, Il Soo Chang, Kun Woo Kim
Ann Surg Treat Res. 2022;102(2):117-124. doi: 10.4174/astr.2022.102.2.117.
Reference
-
1. US markets for varicose vein treatment devices 2013: Medtech 360. Toronto (Canada): Millennium Research Group Inc.;2013.2. U.S. Food and Drug Administration. Approval Order: Venaseal Closure System-P140018 [Internet]. Silver Spring (MD): U.S. Food and Drug Administration;2015. 02. 20. updated 2018 Feb 23. cited 2017 Mar 28. http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfTopic/pma/pma.cfm?num=p140018.3. Almeida JI, Javier JJ, Mackay EG, Bautista C, Cher DJ, Proebstle TM. Two-year follow-up of first human use of cyanoacrylate adhesive for treatment of saphenous vein incompetence. Phlebology. 2015; 30:397–404.
Article4. Proebstle TM, Alm J, Dimitri S, Rasmussen L, Whiteley M, Lawson J, et al. The European multicenter cohort study on cyanoacrylate embolization of refluxing great saphenous veins. J Vasc Surg Venous Lymphat Disord. 2015; 3:2–7.
Article5. Morrison N, Gibson K, McEnroe S, Goldman M, King T, Weiss R, et al. Randomized trial comparing cyanoacrylate embolization and radiofrequency ablation for incompetent great saphenous veins (VeClose). J Vasc Surg. 2015; 61:985–994.
Article6. Gibson K, Ferris B. Cyanoacrylate closure of incompetent great, small and accessory saphenous veins without the use of post-procedure compression: Initial outcomes of a post-market evaluation of the VenaSeal System (the WAVES Study). Vascular. 2017; 25:149–156.
Article7. Chan YC, Law Y, Cheung GC, Ting AC, Cheng SW. Cyanoacrylate glue used to treat great saphenous reflux: measures of outcome. Phlebology. 2017; 32:99–106.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The cutoff value of saphenous vein diameter to predict reflux
- Outcome of ClosureFAST radiofrequency ablation for large-diameter incompetent great saphenous vein
- Impact of the Variations in the Termination of Small Saphenous Vein on Small Saphenous Vein Incompetence
- Ethical Considerations and Adverse Events in Cyanoacrylate Embolization for Non-Saphenous Veins: A Case Report
- Anatomical observation on draining patterns of saphenous tributaries in Korean adults