J Vet Sci.
2018 Mar;19(2):207-215. 10.4142/jvs.2018.19.2.207.
In vitro treatment of lipopolysaccharide increases invasion of Pasteurella multocida serotype B:2 into bovine aortic endothelial cells
- Affiliations
-
- 1Institute of Bioscience, Universiti Putra Malaysia, 43400 Serdang, Malaysia. aro@upm.edu.my
- 2Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 Serdang, Malaysia.
- 3Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, 43400 Serdang, Malaysia.
- KMID: 2407620
- DOI: http://doi.org/10.4142/jvs.2018.19.2.207
Abstract
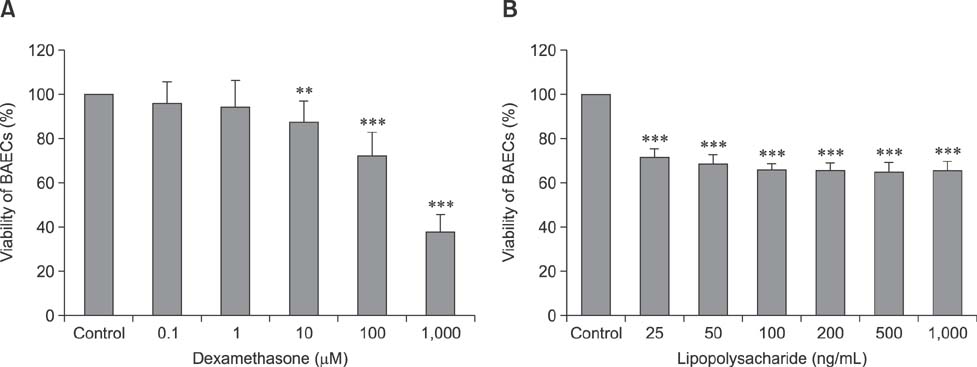
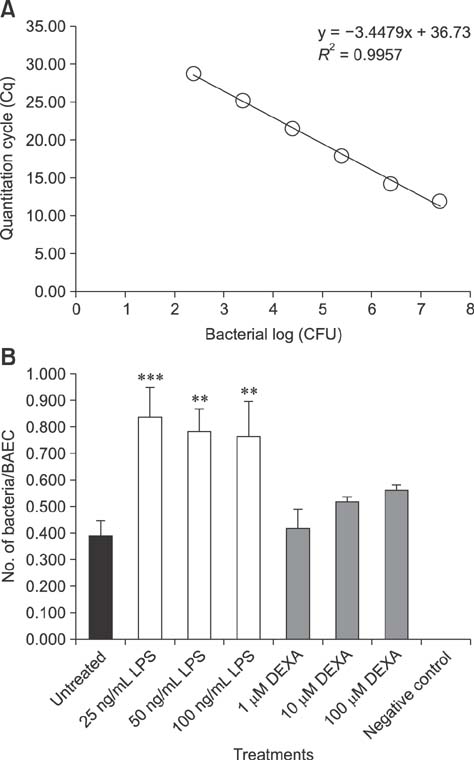
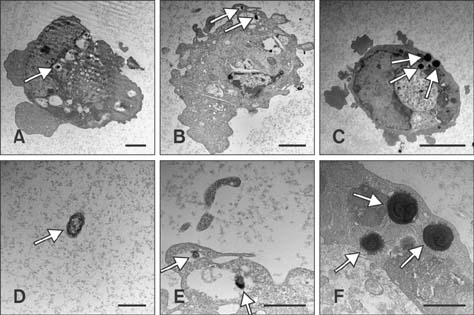
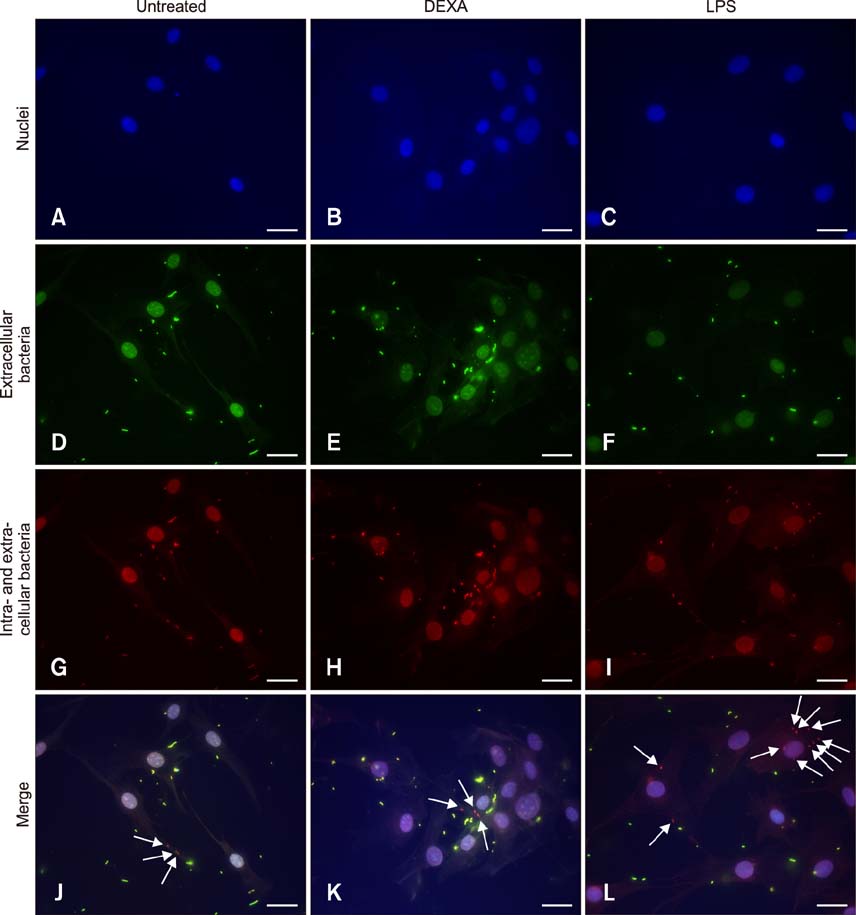
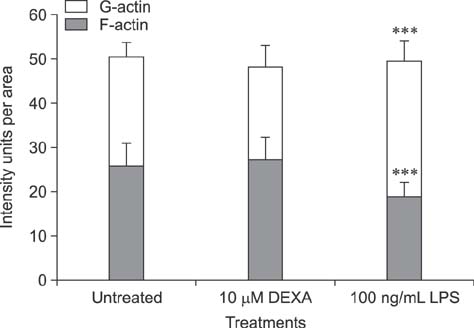
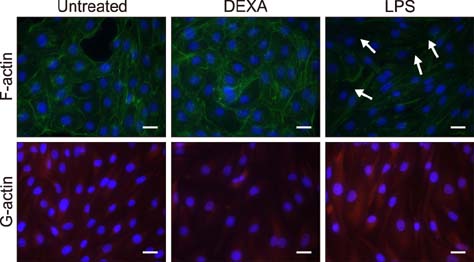
- Pasteurella multocida serotype B:2 causes hemorrhagic septicemia in cattle and buffalo. The invasion mechanism of the bacterium when invading the bloodstream is unclear. This study aimed to characterize the effects of immunomodulatory molecules, namely dexamethasone and lipopolysaccharide, on the invasion efficiency of P. multocida serotype B:2 toward bovine aortic endothelial cells (BAECs) and the involvement of actin microfilaments in the invasion mechanism. The results imply that treatment of BAECs with lipopolysaccharide at 100 ng/mL for 24 h significantly increases the intracellular bacteria number per cell (p < 0.01) compared with those in untreated and dexamethasone-treated cells. The lipopolysaccharide-treated cells showed a significant decrease in F-actin expression and an increase in G-actin expression (p < 0.001), indicating actin depolymerization of BAECs. However, no significant differences were detected in the invasion efficiency and actin filament reorganization between the dexamethasone-treated and untreated cells. Transmission electron microscopy showed that P. multocida B:2 resided in a vacuolar compartment of dexamethasone-treated and untreated cells, whereas the bacteria resided in cellular membrane of lipopolysaccharide-treated cells. The results suggest that lipopolysaccharide destabilizes the actin filaments of BAECs, which could facilitate the invasion of P. multocida B:2 into BAECs.
Keyword
MeSH Terms
Figure
Reference
-
1. Al-haj Ali H, Sawada T, Hatakeyama H, Katayama Y, Ohtsuki N, Itoh O. Invasion of chicken embryo fibroblast cells by avian Pasteurella multocida. Vet Microbiol. 2004; 104:55–62.
Article2. Anderson BH, Watson DL, Colditz IG. The effect of dexamethasone on some immunological parameters in cattle. Vet Res Commun. 1999; 23:399–413.3. Annas S, Zamri-Saad M, Jesse FF, Zunita Z. New sites of localisation of Pasteurella multocida B:2 in buffalo surviving experimental haemorrhagic septicaemia. BMC Vet Res. 2014; 10:88.4. Benkirane A, De Alwis MCL. Haemorrhagic septicaemia, its significance, prevention and control in Asia. Vet Med Czech. 2002; 47:234–240.
Article5. Boyce JD, Adler B. How does Pasteurella multocida respond to the host environment? Curr Opin Microbiol. 2006; 9:117–122.6. Chakravortty D, Koide N, Kato Y, Sugiyama T, Kawai M, Fukada M, Yoshida T, Yokochi T. Cytoskeletal alterations in lipopolysaccharide-induced bovine vascular endothelial cell injury and its prevention by sodium arsenite. Clin Diagn Lab Immunol. 2000; 7:218–225.
Article7. Chen WL, Lin CT, Yao CC, Huang YH, Chou YB, Yin HS, Hu FR. In-vitro effects of dexamethasone on cellular proliferation, apoptosis, and Na+-K+-ATPase activity of bovine corneal endothelial cells. Ocul Immunol Inflamm. 2006; 14:215–223.
Article8. Chung ELT, Abdullah FFJ, Ibrahim HH, Marza AD, Zamri-Saad M, Haron AW, Lila MAM, Norsidin MJ. Clinico-pathology, hematology and biochemistry responses in buffaloes towards Pasteurella multocida type B: 2 immunogen lypopolysaccharide via oral and intravenous routes of infection. Microb Pathog. 2016; 91:141–154.
Article9. De Alwis MCL. Haemorrhagic Septicaemia. ACIAR Monograph No. 57. Canberra: Australian Centre for International Agriculture Research (ACIAR);1999.10. de Alwis MCL, Wijewardana TG, Gomis AIU, Vipulasiri AA. Persistence of the carrier status in haemorrhagic septicaemia (Pasteurella multocida serotype 6:B infection) in buffaloes. Trop Anim Health Prod. 1990; 22:185–194.
Article11. Frey EA, Finlay BB. Lipopolysaccharide induces apoptosis in a bovine endothelial cell line via a soluble CD14 dependent pathway. Microb Pathog. 1998; 24:101–109.
Article12. Galdiero M, De Martino L, Pagnini U, Pisciotta MG, Galdiero E. Interactions between bovine endothelial cells and Pasteurella multocida: association and invasion. Res Microbiol. 2001; 152:57–65.
Article13. Grandel U, Grimminger F. Endothelial responses to bacterial toxins in sepsis. Crit Rev Immunol. 2003; 23:267–299.
Article14. Hardy MP, Gao HB, Dong Q, Ge R, Wang Q, Chai WR, Feng X, Sottas C. Stress hormone and male reproductive function. Cell Tissue Res. 2005; 322:147–153.
Article15. Harper M, Boyce JD, Cox AD, St Michael F, Wilkie IW, Blackall PJ, Adler B. Pasteurella multocida expresses two lipopolysaccharide glycoforms simultaneously, but only a single form is required for virulence: identification of two acceptor-specific heptosyl I transferases. Infect Immun. 2007; 75:3885–3893.
Article16. Horadagoda NU, Hodgson JC, Moon GM, Wijewardana TG, Eckersall PD. Development of a clinical syndrome resembling haemorrhagic septicaemia in the buffalo following intravenous inoculation of Pasteurella multocida serotype B:2 endotoxin and the role of tumour necrosis factor-alpha. Res Vet Sci. 2002; 72:194–200.
Article17. Iovane G, Pagnini P, Galdiero M, Cipollaro de l'Ero G, Vitiello M, D'Isanto M, Marcatili A. Role of Pasteurella multocida porin on cytokine expression and release by murine splenocytes. Vet Immunol Immunopathol. 1998; 66:391–404.
Article18. Jesse FFA, Osman AY, Adamu L, Yusof MSM, Omar AR, Saharee AA, Haron AW, Abdullah R, Zamri-Saad M. Polymerase chain reaction detection of Pasteurella multocida type B:2 in mice following oral inoculation. Asian J Anim Vet Adv. 2013; 8:493–501.19. Kyoui D, Takahashi H, Miya S, Kuda T, Kimura B. Comparison of the major virulence-related genes of Listeria monocytogenes in internalin A truncated strain 36-25-1 and a clinical wild-type strain. BMC Microbiol. 2014; 14:15.
Article20. Lee EJ, Pontes MH, Groisman EA. A bacterial virulence protein promotes pathogenicity by inhibiting the bacterium's own F1Fo ATP synthase. Cell. 2013; 154:146–156.
Article21. Lee MD, Wooley RE, Glisson JR. Invasion of epithelial cell monolayers by turkey strains of Pasteurella multocida. Avian Dis. 1994; 38:72–77.
Article22. Macé S, Mamlouk K, Chipchakova S, Prévost H, Joffraud JJ, Dalgaard P, Pilet MF, Dousset X. Development of a rapid real-time PCR method as a tool to quantify viable Photobacterium phosphoreum bacteria in salmon (Salmo salar) steaks. Appl Environ Microbiol. 2013; 79:2612–2619.
Article23. Mooney A, Byrne C, Clyne M, Johnson-Henry K, Sherman P, Bourke B. Invasion of human epithelial cells by Campylobacter upsaliensis. Cell Microbiol. 2003; 5:835–847.24. Nadkarni MA, Martin FE, Jacques NA, Hunter N. Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiology. 2002; 148:257–266.
Article25. Othman S, Parton R, Coote J. Interaction between mammalian cells and Pasteurella multocida B:2. Adherence, invasion and intracellular survival. Microb Pathog. 2012; 52:353–358.
Article26. Petersen A, Bisgaard M, Townsend K, Christensen H. MLST typing of Pasteurella multocida associated with haemorrhagic septicaemia and development of a real-time PCR specific for haemorrhagic septicaemia associated isolates. Vet Microbiol. 2014; 170:335–341.
Article27. Rafelski SM, Theriot JA. Bacterial shape and ActA distribution affect initiation of Listeria monocytogenes actin-based motility. Biophys J. 2005; 89:2146–2158.
Article28. Rafidah O, Zamri-Saad M. Effect of dexamethasone on protective efficacy of live gdhA derivative Pasteurella multocida B:2 vaccine. Asian J Anim Vet Adv. 2013; 8:548–554.
Article29. Ramdani , Adler B. Opsonic monoclonal antibodies against lipopolysaccharide (LPS) antigens of Pasteurella multocida and the role of LPS in immunity. Vet Microbiol. 1991; 26:335–347.30. Shivachandra SB, Viswas KN, Kumar AA. A review of hemorrhagic septicemia in cattle and buffalo. Anim Health Res Rev. 2011; 12:67–82.
Article31. West NP, Sansonetti P, Mounier J, Exley RM, Parsot C, Guadagnini S, Prévost MC, Prochnicka-Chalufour A, Delepierre M, Tanguy M, Tang CM. Optimization of virulence functions through glucosylation of Shigella LPS. Science. 2005; 307:1313–1317.
Article32. Wheeler R. Outer membrane proteomics of Pasteurella multocida isolates to identify putative host-specificity determinants. Biosci Horiz. 2009; 2:1–12.
Article33. Yap HY, Ghazali K, Wan Mohamad Nazarie WF, Mat Isa MN, Zakaria Z, Omar AR. Draft genome sequence of Pasteurella multocida subsp. multocida strain PMTB, isolated from a buffalo. Genome Announc. 2013; 1:e00872–e00813.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pasteurella multocida isolation from pigs with respiratory disease in Korea
- A Case of pasteurella multocida pleural empyema
- Hydrogen Peroxide as an Effective Disinfectant for Pasteurella multocida
- Meningoencephalitis and pneumonia caused by Pasteurella multocida in rabbits
- Immunologic reactivity of a lipopolysaccharide-protein complex of type A Pasteurella multocida in mice