Pediatr Infect Vaccine.
2017 Dec;24(3):152-159. 10.14776/piv.2017.24.3.152.
Clinical Manifestations and Treatment in Korean Patients with X-Linked Agammaglobulinemia
- Affiliations
-
- 1Department of Pediatrics, Seoul National University Children's Hospital, Seoul, the Republic of Korea. gkim@snu.ac.kr
- 2Department of Pediatrics, Seoul National University College of Medicine, Seoul, the Republic of Korea.
- KMID: 2401390
- DOI: http://doi.org/10.14776/piv.2017.24.3.152
Abstract
- PURPOSE
X-linked agammaglobulinemia (XLA) is a primary immunodeficiency caused by mutations in the Bruton's tyrosine kinase (Btk) gene. The aim of this study was to investigate the clinical manifestations, molecular features, and treatment status of XLA in Korean patients at Seoul National University Children's Hospital.
METHODS
Fourteen Korean boys with XLA showing serum agammaglobulinemia, non-detectable to less than 2% of peripheral B-cells, and mutation of the Btk gene were enrolled. We observed the clinical features, laboratory findings, status of treatment, and complications in these XLA patients.
RESULTS
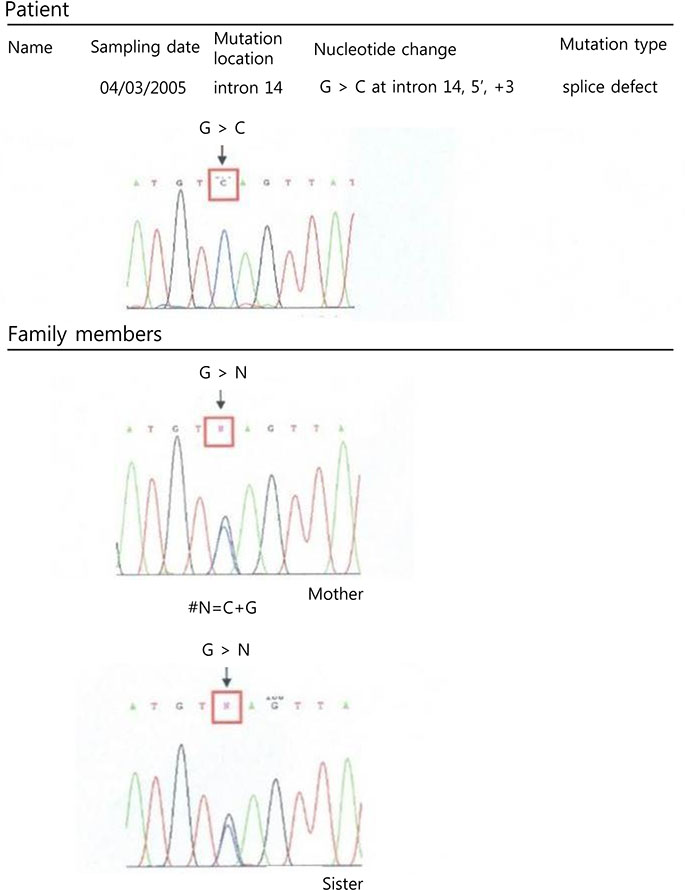
All XLA patients had a history of recurrent bacterial infections before diagnosis, and 20% of them had a neutropenia. Of the XLA patients 35.7% had a family history of XLA and 75% of their mothers were carriers. Btk gene analysis showed variable gene mutations in Xq22 including 9 amino acid substitutions, 3 frameshifts, 1 premature stop codon, and 1 splice defect. After intravenous immunoglobulin replacement therapy, infection episodes decreased, but complications such as bronchiectasis and chronic sinusitis remained.
CONCLUSIONS
In patients less than 4 years of age with recurrent infection, analysis of serum gamma globulin levels and the Btk gene are recommended for the early diagnosis of XLA and for the appropriate prevention of recurrent infection.
MeSH Terms
-
Agammaglobulinemia*
Amino Acid Substitution
B-Lymphocytes
Bacterial Infections
Bronchiectasis
Codon, Nonsense
Diagnosis
Early Diagnosis
gamma-Globulins
Humans
Immunoglobulins
Immunoglobulins, Intravenous
Mothers
Neutropenia
Protein-Tyrosine Kinases
Seoul
Sinusitis
Codon, Nonsense
Immunoglobulins
Immunoglobulins, Intravenous
Protein-Tyrosine Kinases
gamma-Globulins
Figure
Reference
-
1. Bruton OC. Agammaglobulinemia. Pediatrics. 1952; 9:722–728.
Article2. Ochs HD, Smith CI. X-linked agammaglobulinemia. A clinical and molecular analysis. Medicine (Baltimore). 1996; 75:287–299.
Article3. Khan WN, Sideras P, Rosen FS, Alt FW. The role of Bruton's tyrosine kinase in B-cell development and function in mice and man. Ann N Y Acad Sci. 1995; 764:27–38.
Article4. Vetrie D, Vorechovsky I, Sideras P, Holland J, Davies A, Flinter F, et al. The gene involved in X-linked agammaglobulinaemia is a member of the src family of protein-tyrosine kinases. Nature. 1993; 361:226–233.
Article5. Conley ME, Howard V. Clinical findings leading to the diagnosis of X-linked agammaglobulinemia. J Pediatr. 2002; 141:566–571.
Article6. Lederman HM, Winkelstein JA. X-linked agammaglobulinemia: an analysis of 96 patients. Medicine (Baltimore). 1985; 64:145–156.7. Farrar JE, Rohrer J, Conley ME. Neutropenia in X-linked agammaglobulinemia. Clin Immunol Immunopathol. 1996; 81:271–276.
Article8. European Society for Immunodeficiencies. X-linked agammaglobulinemia [Internet]. Geneva: ESID;c2017. cited 2017 Nov 16. Available from: https://esid.org/Working-Parties/Clinical/Resources/Diagnostic-criteriafor-PID2#Q15.9. Nakagawa N, Imai K, Kanegane H, Sato H, Yamada M, Kondoh K, et al. Quantification of kappa-deleting recombination excision circles in Guthrie cards for the identification of early B-cell maturation defects. J Allergy Clin Immunol. 2011; 128:223–225.10. Winkelstein JA, Marino MC, Lederman HM, Jones SM, Sullivan K, Burks AW, et al. X-linked agammaglobulinemia: report on a United States registry of 201 patients. Medicine (Baltimore). 2006; 85:193–202.11. Rohrer J, Parolini O, Belmont JW, Conley ME, Parolini O. The genomic structure of human Btk, the defective gene in X-linked agammaglobulinemia. Immunogenetics. 1994; 40:319–324.
Article12. Ohta Y, Haire RN, Litman RT, Fu SM, Nelson RP, Kratz J, et al. Genomic organization and structure of Bruton agammaglobulinemia tyrosine kinase: localization of mutations associated with varied clinical presentations and course in X chromosome-linked agammaglobulinemia. Proc Natl Acad Sci U S A. 1994; 91:9062–9066.13. Vihinen M, Brandau O, Branden LJ, Kwan SP, Lappalainen I, Lester T, et al. Btkbase, mutation database for X-linked agammaglobulinemia (XLA). Nucleic Acids Res. 1998; 26:242–247.
Article14. Sweinberg SK, Wodell RA, Grodofsky MP, Greene JM, Conley ME. Retrospective analysis of the incidence of pulmonary disease in hypogammaglobulinemia. J Allergy Clin Immunol. 1991; 88:96–104.
Article15. Quartier P, Debre M, De Blic J, de Sauverzac R, Sayegh N, Jabado N, et al. Early and prolonged intravenous immunoglobulin replacement therapy in childhood agammaglobulinemia: a retrospective survey of 31 patients. J Pediatr. 1999; 134:589–596.
Article16. van der Meer JW, Weening RS, Schellekens PT, van Munster IP, Nagengast FM. Colorectal cancer in patients with Xlinked agammaglobulinaemia. Lancet. 1993; 341:1439–1440.
Article17. Bonagura VR, Marchlewski R, Cox A, Rosenthal DW. Biologic IgG level in primary immunodeficiency disease: the IgG level that protects against recurrent infection. J Allergy Clin Immunol. 2008; 122:210–212.
Article18. Roifman CM, Berger M, Notarangelo LD. Management of primary antibody deficiency with replacement therapy: summary of guidelines. Immunol Allergy Clin North Am. 2008; 28:875–876.
Article19. Roifman CM, Schroeder H, Berger M, Sorensen R, Ballow M, Buckley RH, et al. Comparison of the efficacy of IGIVC, 10% (caprylate/chromatography) and IGIV-SD, 10% as replacement therapy in primary immune deficiency. A randomized double-blind trial. Int Immunopharmacol. 2003; 3:1325–1333.
Article20. Favre O, Leimgruber A, Nicole A, Spertini F. Intravenous immunoglobulin replacement prevents severe and lower respiratory tract infections, but not upper respiratory tract and non-respiratory infections in common variable immune deficiency. Allergy. 2005; 60:385–390.
Article21. Bayrakci B, Ersoy F, Sanal O, Kilic S, Metin A, Tezcan I. The efficacy of immunoglobulin replacement therapy in the long-term follow-up of the B-cell deficiencies (XLA, HIM, CVID). Turk J Pediatr. 2005; 47:239–246.22. Eijkhout HW, van Der, Kallenberg CG, Weening RS, van Dissel JT, Sanders LA, et al. The effect of two different dosages of intravenous immunoglobulin on the incidence of recurrent infections in patients with primary hypogammaglobulinemia. A randomized, double-blind, multicenter crossover trial. Ann Intern Med. 2001; 135:165–174.
Article23. Roifman CM, Levison H, Gelfand EW. High-dose versus low-dose intravenous immunoglobulin in hypogammaglobulinaemia and chronic lung disease. Lancet. 1987; 1:1075–1077.
Article24. Pruzanski W, Sussman G, Dorian W, Van T, Ibanez D, Redelmeier D. Relationship of the dose of intravenous gammaglobulin to the prevention of infections in adults with common variable immunodeficiency. Inflammation. 1996; 20:353–359.
Article25. Sorensen RU, Polmar SH. Efficacy and safety of high-dose intravenous immune globulin therapy for antibody deficiency syndromes. Am J Med. 1984; 76(3A):83–90.
Article26. Ochs HD, Fischer SH, Wedgwood RJ, Wara DW, Cowan MJ, Ammann AJ, et al. Comparison of high-dose and lowdose intravenous immunoglobulin therapy in patients with primary immunodeficiency diseases. Am J Med. 1984; 76(3A):78–82.
Article27. Lucas M, Lee M, Lortan J, Lopez-Granados E, Misbah S, Chapel H. Infection outcomes in patients with common variable immunodeficiency disorders: relationship to immunoglobulin therapy over 22 years. J Allergy Clin Immunol. 2010; 125:1354–1360.
Article28. Orange JS, Grossman WJ, Navickis RJ, Wilkes MM. Impact of trough IgG on pneumonia incidence in primary immunodeficiency: a meta-analysis of clinical studies. Clin Immunol. 2010; 137:21–30.
Article29. Rohrer J, Conley ME. Correction of X-linked immunodeficient mice by competitive reconstitution with limiting numbers of normal bone marrow cells. Blood. 1999; 94:3358–3365.
Article30. Howard V, Myers LA, Williams DA, Wheeler G, Turner EV, Cunningham JM, et al. Stem cell transplants for patients with X-linked agammaglobulinemia. Clin Immunol. 2003; 107:98–102.
Article