J Clin Neurol.
2018 Jan;14(1):81-89. 10.3988/jcn.2018.14.1.81.
A Prospective Study of Chronic Oxaliplatin-Induced Neuropathy in Patients with Colon Cancer: Long-Term Outcomes and Predictors of Severe Oxaliplatin-Induced Neuropathy
- Affiliations
-
- 1Department of Neurology, Research Institute and Hospital, National Cancer Center, Goyang, Korea. hojinkim@ncc.re.kr
- 2Center for Colorectal Cancer, Research Institute and Hospital, National Cancer Center, Goyang, Korea.
- 3Rehabilitation Clinic, Research Institute and Hospital, National Cancer Center, Goyang, Korea. shchung@ncc.re.kr
- KMID: 2399603
- DOI: http://doi.org/10.3988/jcn.2018.14.1.81
Abstract
- BACKGROUND AND PURPOSE
The objective of this study was to determine the incidence and long-term outcomes of oxaliplatin-induced peripheral neuropathy (OIPN), as well as predictors of its severe form.
METHODS
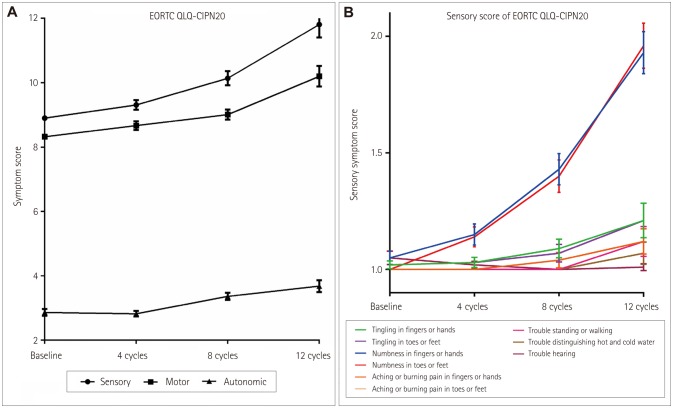
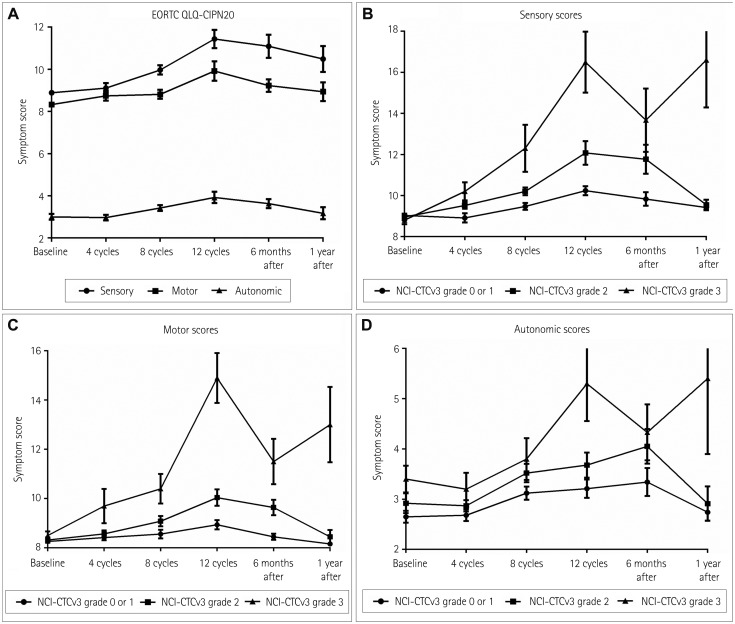
Sixty-nine patients who were taking oxaliplatin for colon cancer were prospectively followed prior to starting chemotherapy and after 4, 8, and 12 cycles of chemotherapy. Thirty-six patients completed the follow-up at 1 year after the end of chemotherapy. The patients were assessed using clinical assessment scales and nerve conduction studies (NCS) at each follow-up visit.
RESULTS
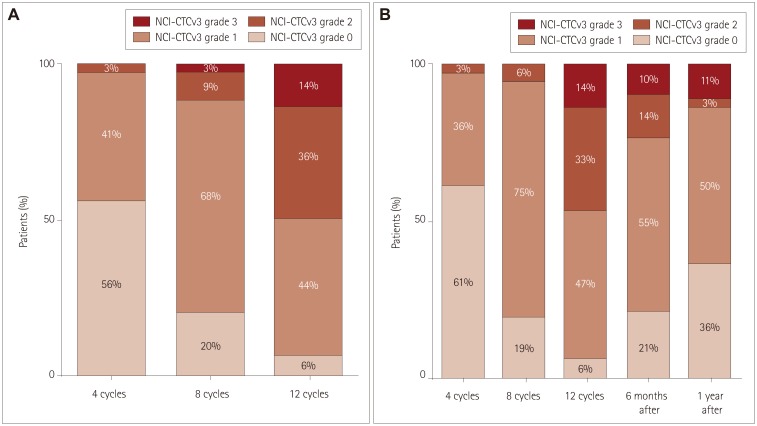
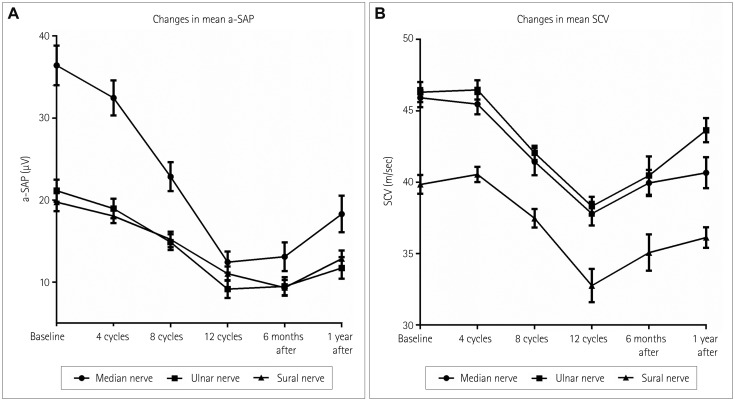
By applying the National Cancer Institute Common Toxicity criteria, OIPN was classified as grade 1 in 30 (44%) patients, grade 2 in 25 (36%), and grade 3 in 10 (14%) at the completion of therapy. At 1 year after the treatment, OIPN of grades 1, 2, and 3 was found in 50, 3, and 11% of the patients, respectively. Multivariate analysis showed that reductions of the amplitude of the sensory action potential of >11.5% in the median nerve between baseline and four cycles of chemotherapy (odds ratio=5.603, p=0.031) and of >22.5% in the sural nerve between four and eight cycles of chemotherapy (odds ratio=5.603, p=0.031) were independently associated with the risk of developing grade-3 OIPN.
CONCLUSIONS
While the severity of OIPN can improve after oxaliplatin discontinuation, more than half of the patients in this study still had OIPN at 1 year after discontinuation. Early changes in the NCS results for sensory nerves can predict the development of severe OIPN during treatment.
MeSH Terms
Figure
Cited by 1 articles
-
Oxaliplatin-induced Peripheral Neuropathy, Symptoms, Distress and Quality of Life among Korean Patients with Gastrointestinal Cancer
Hye Jeong Jung, Soo jung Ahn, Yoo Ri Yang, Kyoung A Kim, Sang Joon Shin, Min Kyu Jung, Sang Hui Chu
Asian Oncol Nurs. 2019;19(4):204-213. doi: 10.5388/aon.2019.19.4.204.
Reference
-
1. André T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004; 350:2343–2351. PMID: 15175436.
Article2. Chau I, Cunningham D. Adjuvant therapy in colon cancer--what, when and how? Ann Oncol. 2006; 17:1347–1359. PMID: 16524974.
Article3. Grothey A. Oxaliplatin-safety profile: neurotoxicity. Semin Oncol. 2003; 30(4 Suppl 15):5–13.
Article4. Krishnan AV, Goldstein D, Friedlander M, Kiernan MC. Oxaliplatin-induced neurotoxicity and the development of neuropathy. Muscle Nerve. 2005; 32:51–60. PMID: 15880395.
Article5. Grothey A, Goldberg RM. A review of oxaliplatin and its clinical use in colorectal cancer. Expert Opin Pharmacother. 2004; 5:2159–2170. PMID: 15461551.
Article6. Sisignano M, Baron R, Scholich K, Geisslinger G. Mechanism-based treatment for chemotherapy-induced peripheral neuropathic pain. Nat Rev Neurol. 2014; 10:694–707. PMID: 25366108.
Article7. Mols F, Beijers T, Lemmens V, van den Hurk CJ, Vreugdenhil G, van de Poll-Franse LV. Chemotherapy-induced neuropathy and its association with quality of life among 2- to 11-year colorectal cancer survivors: results from the population-based PROFILES registry. J Clin Oncol. 2013; 31:2699–2707. PMID: 23775951.
Article8. Yun YH, Park YS, Lee ES, Bang SM, Heo DS, Park SY, et al. Validation of the Korean version of the EORTC QLQ-C30. Qual Life Res. 2004; 13:863–868. PMID: 15129896.
Article9. Argyriou AA, Velasco R, Briani C, Cavaletti G, Bruna J, Alberti P, et al. Peripheral neurotoxicity of oxaliplatin in combination with 5-fluorouracil (FOLFOX) or capecitabine (XELOX): a prospective evaluation of 150 colorectal cancer patients. Ann Oncol. 2012; 23:3116–3122. PMID: 22865779.
Article10. Trotti A, Colevas AD, Setser A, Rusch V, Jaques D, Budach V, et al. CTCAE v3.0: development of a comprehensive grading system for the adverse effects of cancer treatment. Semin Radiat Oncol. 2003; 13:176–181. PMID: 12903007.
Article11. Kim HY, Kang JH, Youn HJ, So HS, Song CE, Chae SY, et al. Reliability and validity of the Korean version of the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire to assess Chemotherapy-induced peripheral neuropathy. J Korean Acad Nurs. 2014; 44:735–742. PMID: 25608551.
Article12. Postma TJ, Aaronson NK, Heimans JJ, Muller MJ, Hildebrand JG, Delattre JY, et al. The development of an EORTC quality of life questionnaire to assess chemotherapy-induced peripheral neuropathy: the QLQ-CIPN20. Eur J Cancer. 2005; 41:1135–1139. PMID: 15911236.
Article13. Briani C, Argyriou AA, Izquierdo C, Velasco R, Campagnolo M, Alberti P, et al. Long-term course of oxaliplatin-induced polyneuropathy: a prospective 2-year follow-up study. J Peripher Nerv Syst. 2014; 19:299–306. PMID: 25582667.
Article14. Kuebler JP, Wieand HS, O'Connell MJ, Smith RE, Colangelo LH, Yothers G, et al. Oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: results from NSABP C-07. J Clin Oncol. 2007; 25:2198–2204. PMID: 17470851.
Article15. André T, Boni C, Navarro M, Tabernero J, Hickish T, Topham C, et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol. 2009; 27:3109–3116. PMID: 19451431.
Article16. Beijers AJ, Mols F, Vreugdenhil G. A systematic review on chronic oxaliplatin-induced peripheral neuropathy and the relation with oxaliplatin administration. Support Care Cancer. 2014; 22:1999–2007. PMID: 24728618.
Article17. Padman S, Lee J, Kumar R, Slee M, Hakendorf P, Richards A, et al. Late effects of oxaliplatin-induced peripheral neuropathy (LEON)--cross-sectional cohort study of patients with colorectal cancer surviving at least 2 years. Support Care Cancer. 2015; 23:861–869. PMID: 25223350.18. Vatandoust S, Joshi R, Pittman KB, Esterman A, Broadbridge V, Adams J, et al. A descriptive study of persistent oxaliplatin-induced peripheral neuropathy in patients with colorectal cancer. Support Care Cancer. 2014; 22:513–518. PMID: 24122404.
Article19. Kokotis P, Schmelz M, Kostouros E, Karandreas N, Dimopoulos MA. Oxaliplatin-induced neuropathy: a long-term clinical and neurophysiologic follow-up study. Clin Colorectal Cancer. 2016; 15:e133–e140. PMID: 27038553.
Article20. Park SB, Lin CS, Krishnan AV, Goldstein D, Friedlander ML, Kiernan MC. Long-term neuropathy after oxaliplatin treatment: challenging the dictum of reversibility. Oncologist. 2011; 16:708–716. PMID: 21478275.
Article21. Pietrangeli A, Leandri M, Terzoli E, Jandolo B, Garufi C. Persistence of high-dose oxaliplatin-induced neuropathy at long-term follow-up. Eur Neurol. 2006; 56:13–16. PMID: 16825773.
Article22. LoMonaco M, Milone M, Batocchi AP, Padua L, Restuccia D, Tonali P. Cisplatin neuropathy: clinical course and neurophysiological findings. J Neurol. 1992; 239:199–204. PMID: 1317914.
Article23. Podratz JL, Knight AM, Ta LE, Staff NP, Gass JM, Genelin K, et al. Cisplatin induced mitochondrial DNA damage in dorsal root ganglion neurons. Neurobiol Dis. 2011; 41:661–668. PMID: 21145397.
Article24. Velasco R, Bruna J, Briani C, Argyriou AA, Cavaletti G, Alberti P, et al. Early predictors of oxaliplatin-induced cumulative neuropathy in colorectal cancer patients. J Neurol Neurosurg Psychiatry. 2014; 85:392–398. PMID: 23813745.
Article25. Lonardi S, Sobrero A, Rosati G, Di Bartolomeo M, Ronzoni M, Aprile G, et al. Phase III trial comparing 3–6 months of adjuvant FOLFOX4/XELOX in stage II–III colon cancer: safety and compliance in the TOSCA trial. Ann Oncol. 2016; 27:2074–2081. PMID: 27573560.26. Tsai YJ, Lin JK, Chen WS, Jiang JK, Teng HW, Yen CC, et al. Adjuvant FOLFOX treatment for stage III colon cancer: how many cycles are enough? Springerplus. 2016; 5:1318. PMID: 27563513.
Article27. Custodio A, Moreno-Rubio J, Aparicio J, Gallego-Plazas J, Yaya R, Maurel J, et al. Pharmacogenetic predictors of severe peripheral neuropathy in colon cancer patients treated with oxaliplatin-based adjuvant chemotherapy: a GEMCAD group study. Ann Oncol. 2014; 25:398–403. PMID: 24351404.
Article28. Sereno M, Gutiérrez-Gutiérrez G, Rubio JM, Apellániz-Ruiz M, Sánchez-Barroso L, Casado E, et al. Genetic polymorphisms of SCN9A are associated with oxaliplatin-induced neuropathy. BMC Cancer. 2017; 17:63. PMID: 28103821.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Oxaliplatin-Induced Immune-Mediated Thrombocytopenia: A Case Report
- Oxaliplatin-induced Sudden Hearing Loss in a Patient with Pancreatic Cancer
- A Case of Autoimmune Hemolytic Anemia Caused by Repeated Administration of Oxaliplatin
- Potential of histone deacetylase 6 inhibitors in alleviating chemotherapy-induced peripheral neuropathy
- A case of oxaliplatin-induced immune-mediated thrombocytopenia