Korean J Physiol Pharmacol.
2017 Nov;21(6):643-650. 10.4196/kjpp.2017.21.6.643.
Chronic cerebral hypoperfusion and plasticity of the posterior cerebral artery following permanent bilateral common carotid artery occlusion
- Affiliations
-
- 1Department of Pharmacology, Catholic Neuroscience Institute, College of Medicine, The Catholic University of Korea, Seoul 06591, Korea. syk@catholic.ac.kr
- KMID: 2395259
- DOI: http://doi.org/10.4196/kjpp.2017.21.6.643
Abstract
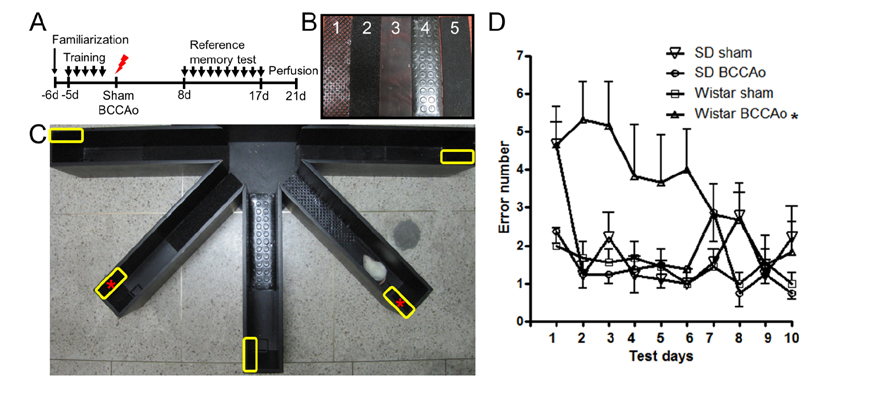
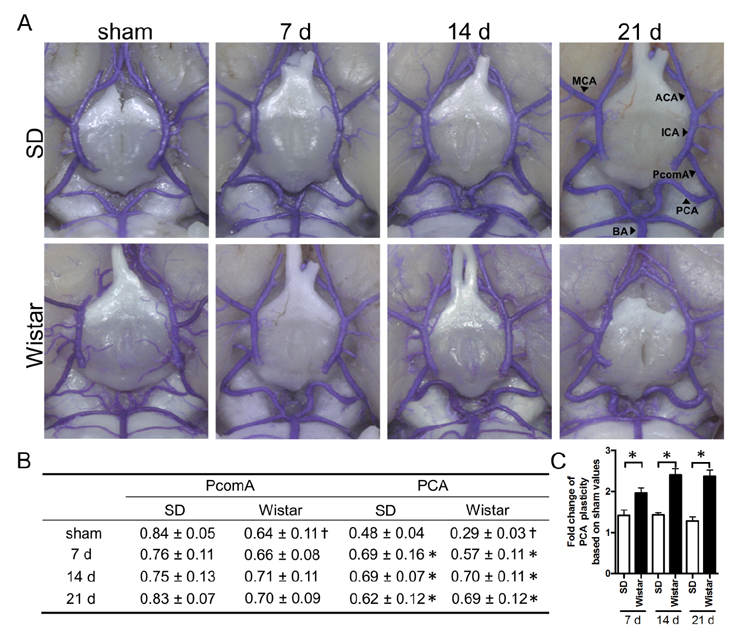
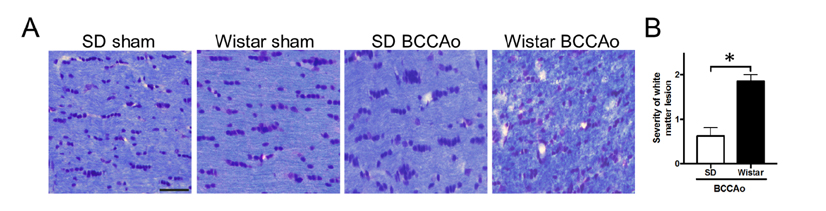
- Vascular dementia (VaD) is a group of heterogeneous diseases with the common feature of cerebral hypoperfusion. To identify key factors contributing to VaD pathophysiology, we performed a detailed comparison of Wistar and Sprague-Dawley (SD) rats subjected to permanent bilateral common carotid artery occlusion (BCCAo). Eight-week old male Wistar and SD rats underwent BCCAo, followed by a reference memory test using a five-radial arm maze with tactile cues. Continuous monitoring of cerebral blood flow (CBF) was performed with a laser Doppler perfusion imaging (LDPI) system. A separate cohort of animals was sacrificed for evaluation of the brain vasculature and white matter damage after BCCAo. We found reference memory impairment in Wistar rats, but not in SD rats. Moreover, our LDPI system revealed that Wistar rats had significant hypoperfusion in the brain region supplied by the posterior cerebral artery (PCA). Furthermore, Wistar rats showed more profound CBF reduction in the forebrain region than did SD rats. Post-mortem analysis of brain vasculature demonstrated greater PCA plasticity at all time points after BCCAo in Wistar rats. Finally, we confirmed white matter rarefaction that was only observed in Wistar rats. Our studies show a comprehensive and dynamic CBF status after BCCAo in Wistar rats in addition to severe PCA dolichoectasia, which correlated well with white matter lesion and memory decline.
Keyword
MeSH Terms
Figure
Reference
-
1. Plassman BL, Langa KM, Fisher GG, Heeringa SG, Weir DR, Ofstedal MB, Burke JR, Hurd MD, Potter GG, Rodgers WL, Steffens DC, Willis RJ, Wallace RB. Prevalence of dementia in the United States: the aging, demographics, and memory study. Neuroepidemiology. 2007; 29:125–132.2. Sopala M, Danysz W. Chronic cerebral hypoperfusion in the rat enhances age-related deficits in spatial memory. J Neural Transm (Vienna). 2001; 108:1445–1456.3. Venkat P, Chopp M, Chen J. Models and mechanisms of vascular dementia. Exp Neurol. 2015; 272:97–108.4. Román GC, Sachdev P, Royall DR, Bullock RA, Orgogozo JM, López-Pousa S, Arizaga R, Wallin A. Vascular cognitive disorder: a new diagnostic category updating vascular cognitive impairment and vascular dementia. J Neurol Sci. 2004; 226:81–87.5. Cho KO, La HO, Cho YJ, Sung KW, Kim SY. Minocycline attenuates white matter damage in a rat model of chronic cerebral hypoperfusion. J Neurosci Res. 2006; 83:285–291.6. Soria G, Tudela R, Márquez-Martín A, Camón L, Batalle D, Muñoz-Moreno E, Eixarch E, Puig J, Pedraza S, Vila E, Prats-Galino A, Planas AM. The ins and outs of the BCCAo model for chronic hypoperfusion: a multimodal and longitudinal MRI approach. PLoS One. 2013; 8:e74631.7. Wakita H, Tomimoto H, Akiguchi I, Kimura J. Glial activation and white matter changes in the rat brain induced by chronic cerebral hypoperfusion: an immunohistochemical study. Acta Neuropathol. 1994; 87:484–492.8. Jiwa NS, Garrard P, Hainsworth AH. Experimental models of vascular dementia and vascular cognitive impairment: a systematic review. J Neurochem. 2010; 115:814–828.9. Farkas E, Luiten PG, Bari F. Permanent, bilateral common carotid artery occlusion in the rat: a model for chronic cerebral hypoperfusion-related neurodegenerative diseases. Brain Res Rev. 2007; 54:162–180.10. Kim SK, Cho KO, Kim SY. The plasticity of posterior communicating artery influences on the outcome of white matter injury induced by chronic cerebral hypoperfusion in rats. Neurol Res. 2009; 31:245–250.11. Kim SK, Cho KO, Kim SY. White matter damage and hippocampal neurodegeneration induced by permanent bilateral occlusion of common carotid artery in the rat: comparison between wistar and sprague-dawley strain. Korean J Physiol Pharmacol. 2008; 12:89–94.12. Tanaka K, Wada N, Hori K, Asanuma M, Nomura M, Ogawa N. Chronic cerebral hypoperfusion disrupts discriminative behavior in acquired-learning rats. J Neurosci Methods. 1998; 84:63–68.13. Storozheva ZI, Proshin AT, Sherstnev VV, Storozhevykh TP, Senilova YE, Persiyantseva NA, Pinelis VG, Semenova NA, Zakharova EI, Pomytkin IA. Dicholine salt of succinic acid, a neuronal insulin sensitizer, ameliorates cognitive deficits in rodent models of normal aging, chronic cerebral hypoperfusion, and beta-amyloid peptide-(25-35)-induced amnesia. BMC Pharmacol. 2008; 8:1.14. Ohta H, Nishikawa H, Kimura H, Anayama H, Miyamoto M. Chronic cerebral hypoperfusion by permanent internal carotid ligation produces learning impairment without brain damage in rats. Neuroscience. 1997; 79:1039–1050.15. Zhang ZH, Shi GX, Li QQ, Wang YJ, Li P, Zhao JX, Yang JW, Liu CZ. Comparison of cognitive performance between two rat models of vascular dementia. Int J Neurosci. 2014; 124:818–823.16. Liu HX, Zhang JJ, Zheng P, Zhang Y. Altered expression of MAP-2, GAP-43, and synaptophysin in the hippocampus of rats with chronic cerebral hypoperfusion correlates with cognitive impairment. Brain Res Mol Brain Res. 2005; 139:169–177.17. Farkas E, Institóris A, Domoki F, Mihály A, Luiten PG, Bari F. Diazoxide and dimethyl sulphoxide prevent cerebral hypoperfusionrelated learning dysfunction and brain damage after carotid artery occlusion. Brain Res. 2004; 1008:252–260.18. Ma X, Sun Z, Liu Y, Jia Y, Zhang B, Zhang J. Resveratrol improves cognition and reduces oxidative stress in rats with vascular dementia. Neural Regen Res. 2013; 8:2050–2059.19. Ni J, Ohta H, Matsumoto K, Watanabe H. Progressive cognitive impairment following chronic cerebral hypoperfusion induced by permanent occlusion of bilateral carotid arteries in rats. Brain Res. 1994; 653:231–236.20. Pappas BA, de la Torre JC, Davidson CM, Keyes MT, Fortin T. Chronic reduction of cerebral blood flow in the adult rat: late-emerging CA1 cell loss and memory dysfunction. Brain Res. 1996; 708:50–58.21. Li WZ, Wu WY, Huang H, Wu YY, Yin YY. Protective effect of bilobalide on learning and memory impairment in rats with vascular dementia. Mol Med Rep. 2013; 8:935–941.22. Choy M, Ganesan V, Thomas DL, Thornton JS, Proctor E, King MD, van der Weerd L, Gadian DG, Lythgoe MF. The chronic vascular and haemodynamic response after permanent bilateral common carotid occlusion in newborn and adult rats. J Cereb Blood Flow Metab. 2006; 26:1066–1075.23. Tsuchiya M, Sako K, Yura S, Yonemasu Y. Cerebral blood flow and histopathological changes following permanent bilateral carotid artery ligation in Wistar rats. Exp Brain Res. 1992; 89:87–92.24. Otori T, Katsumata T, Muramatsu H, Kashiwagi F, Katayama Y, Terashi A. Long-term measurement of cerebral blood flow and metabolism in a rat chronic hypoperfusion model. Clin Exp Pharmacol Physiol. 2003; 30:266–272.25. Ulrich PT, Kroppenstedt S, Heimann A, Kempski O. Laser-Doppler scanning of local cerebral blood flow and reserve capacity and testing of motor and memory functions in a chronic 2-vessel occlusion model in rats. Stroke. 1998; 29:2412–2420.26. Smoker WR, Corbett JJ, Gentry LR, Keyes WD, Price MJ, McKusker S. High-resolution computed tomography of the basilar artery: 2. Vertebrobasilar dolichoectasia: clinical-pathologic correlation and review. AJNR Am J Neuroradiol. 1986; 7:61–72.27. Lin YW, Chen CH, Lai ML. The dilemma of treating vertebrobasilar dolichoectasia. Clin Pract. 2012; 2:e84.28. Pico F, Labreuche J, Touboul PJ, Leys D, Amarenco P. Intracranial arterial dolichoectasia and small-vessel disease in stroke patients. Ann Neurol. 2005; 57:472–479.29. Ince B, Petty GW, Brown RD Jr, Chu CP, Sicks JD, Whisnant JP. Dolichoectasia of the intracranial arteries in patients with first ischemic stroke: a population-based study. Neurology. 1998; 50:1694–1698.30. Pico F, Labreuche J, Seilhean D, Duyckaerts C, Hauw JJ, Amarenco P. Association of small-vessel disease with dilatative arteriopathy of the brain: neuropathologic evidence. Stroke. 2007; 38:1197–1202.31. Kalaria RN, Erkinjuntti T. Small vessel disease and subcortical vascular dementia. J Clin Neurol. 2006; 2:1–11.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Corrigendum to: Chronic cerebral hypoperfusion and plasticity of the posterior cerebral artery following permanent bilateral common carotid artery occlusion
- A Case bilateral Persistent Primitive Trigeminal Artery Combined with Cerebral Rete Mirabile
- Anesthetic management of a patient with bilateral common carotid and subclavian arteries occlusion using cerebral oximetry monitoring: A case report
- Aspiration Clot Removal by Mannual Compression of Common Carotid Artery for Acute Internal Carotid Occlusion: a Case Report
- Intracranial Aneurysm Associated with Aplasia of the Internal Cartoid Artery