Cancer Res Treat.
2017 Oct;49(4):937-946. 10.4143/crt.2016.423.
Efficacy and Safety of First-Line Necitumumab Plus Gemcitabine and Cisplatin Versus Gemcitabine and Cisplatin in East Asian Patients with Stage IV Squamous Non-small Cell Lung Cancer: A Subgroup Analysis of the Phase 3, Open-Label, Randomized SQUIRE Study
- Affiliations
-
- 1Division of Hematology/Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. kpark@skku.edu
- 2Department of Medicine, Gachon University Gil Medical Center, Incheon, Korea.
- 3Saint Luke's Medical Center, Quezon City, Philippines.
- 4Chief Division of Medical Oncology, Department of Medicine, Faculty of Medicine, Maharaj Nakorn Chiang Mai, Chiang Mai University, Chiang Mai, Thailand.
- 5Department of Internal Medicine, Chonbuk National University Hospital, Jeonju, Korea.
- 6Lilly Deutschland GmbH, Bad Homburg, Germany.
- 7Eli Lilly and Company, Gurgaon, India.
- 8Eli Lilly Interamerica Inc., Buenos Aires, Argentina.
- KMID: 2394813
- DOI: http://doi.org/10.4143/crt.2016.423
Abstract
- PURPOSE
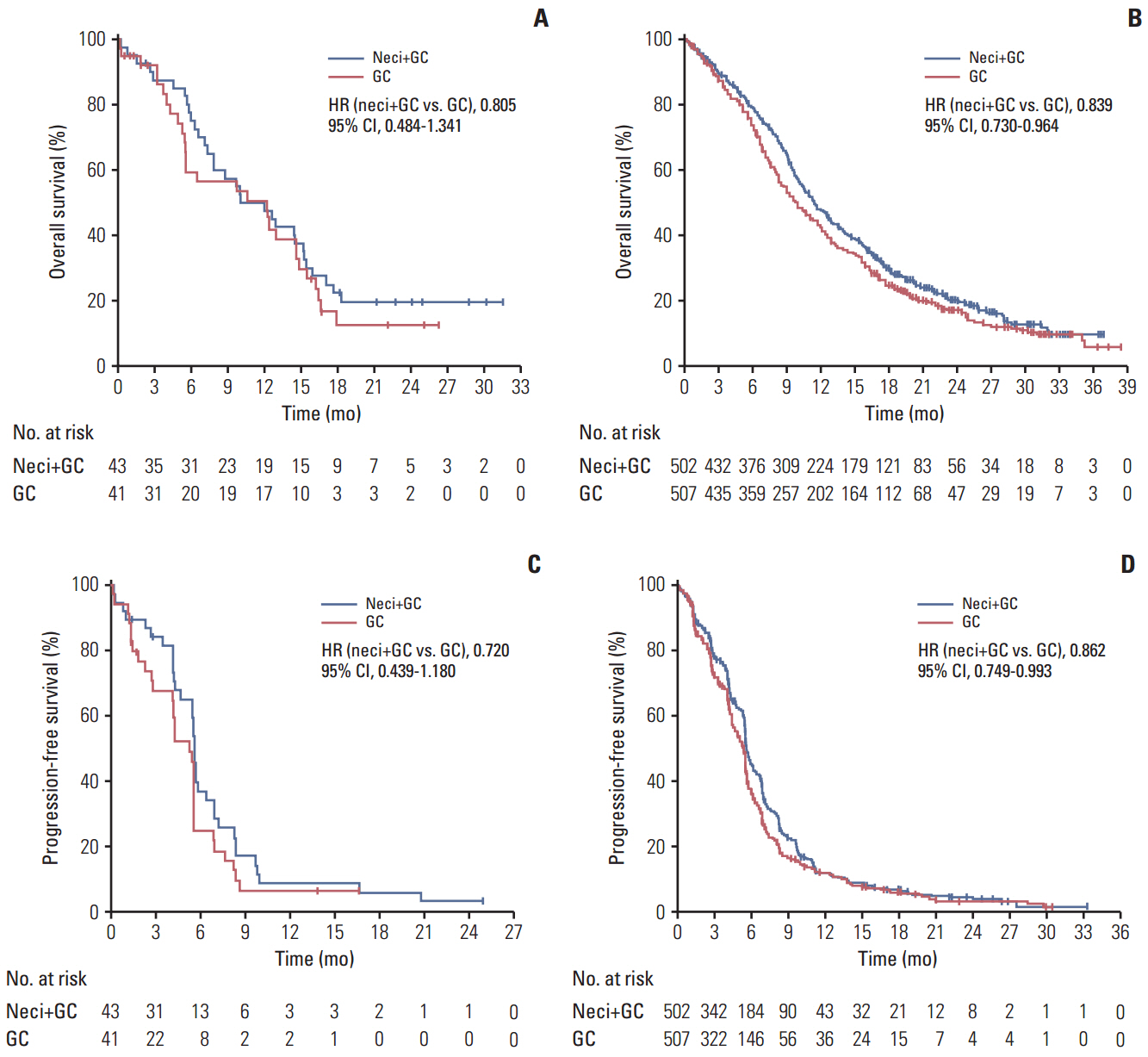
The phase 3 randomized SQUIRE study revealed significantly longer overall survival (OS) and progression-free survival (PFS) for necitumumab plus gemcitabine and cisplatin (neci+GC) than for gemcitabine and cisplatin alone (GC) in 1,093 patients with previously untreated advanced squamous non-small cell lung cancer (NSCLC). This post hoc subgroup analysis assessed the efficacy and safety of neci+GC among East Asian (EA) patients enrolled in the study.
MATERIALS AND METHODS
All patients received up to six 3-week cycles of gemcitabine (days 1 and 8, 1,250 mg/m²) and cisplatin (day 1, 75 mg/m²). Patients in the neci+GC arm also received necitumumab (days 1 and 8, 800 mg) until disease progression or unacceptable toxicity. Hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated from stratified Cox proportional hazards models.
RESULTS
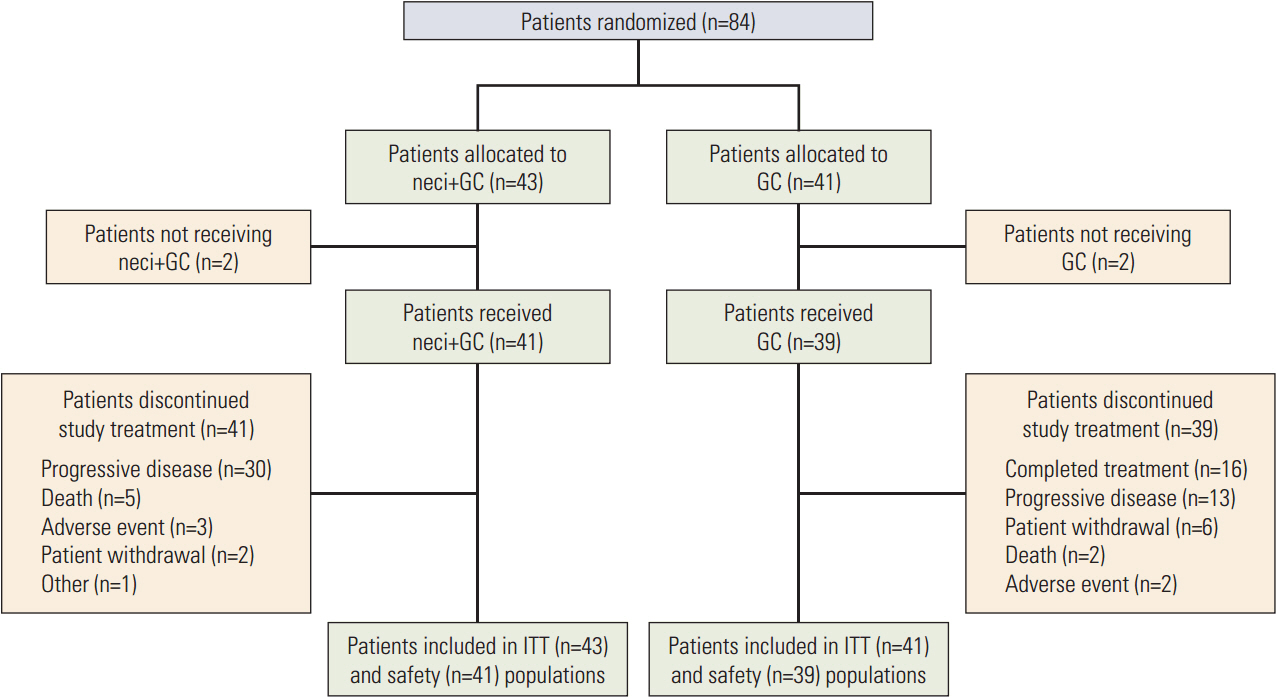
In EA patients, there were improvements for neci+GC (n=43) versus GC (n=41) in OS (HR, 0.805; 95% CI, 0.484 to 1.341) and PFS (HR, 0.720; 95% CI, 0.439 to 1.180), consistent with the results for non-EA patients observed in the present study. The overall safety data were consistent between EA and non-EA patients. A numerically higher proportion of patients experienced serious adverse events (AEs), grade ≥ 3 AEs, and AEs with an outcome of death for neci+GC versus GC in EA patients and EA patients versus non-EA patients for neci+GC.
CONCLUSION
Although limited by the small sample size and post hoc nature of the analysis, these findings are consistent with those of the overall study and suggest that neci+GC offers a survival advantage and favorable benefit/risk for EA patients with advanced squamous NSCLC.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Perez-Moreno P, Brambilla E, Thomas R, Soria JC. Squamous cell carcinoma of the lung: molecular subtypes and therapeutic opportunities. Clin Cancer Res. 2012; 18:2443–51.
Article2. Gandara DR, Hammerman PS, Sos ML, Lara PN Jr, Hirsch FR. Squamous cell lung cancer: from tumor genomics to cancer therapeutics. Clin Cancer Res. 2015; 21:2236–43.
Article3. Gentzler RD, Johnson ML. Complex decisions for first-line and maintenance treatment of advanced wild-type non-small cell lung cancer. Oncologist. 2015; 20:299–306.
Article4. Pirker R, Pereira JR, Szczesna A, von Pawel J, Krzakowski M, Ramlau R, et al. Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (FLEX): an openlabel randomised phase III trial. Lancet. 2009; 373:1525–31.
Article5. Sgambato A, Casaluce F, Maione P, Rossi A, Ciardiello F, Gridelli C. Cetuximab in advanced non-small cell lung cancer (NSCLC): the showdown? J Thorac Dis. 2014; 6:578–80.6. Hirsch FR, Varella-Garcia M, Bunn PA Jr, Di Maria MV, Veve R, Bremmes RM, et al. Epidermal growth factor receptor in non-small-cell lung carcinomas: correlation between gene copy number and protein expression and impact on prognosis. J Clin Oncol. 2003; 21:3798–807.
Article7. Dacic S, Flanagan M, Cieply K, Ramalingam S, Luketich J, Belani C, et al. Significance of EGFR protein expression and gene amplification in non-small cell lung carcinoma. Am J Clin Pathol. 2006; 125:860–5.
Article8. Greillier L, Tomasini P, Barlesi F. Necitumumab for non-small cell lung cancer. Expert Opin Biol Ther. 2015; 15:1231–9.
Article9. Thatcher N, Hirsch FR, Luft AV, Szczesna A, Ciuleanu TE, Dediu M, et al. Necitumumab plus gemcitabine and cisplatin versus gemcitabine and cisplatin alone as first-line therapy in patients with stage IV squamous non-small-cell lung cancer (SQUIRE): an open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2015; 16:763–74.
Article10. Sekine I, Yamamoto N, Nishio K, Saijo N. Emerging ethnic differences in lung cancer therapy. Br J Cancer. 2008; 99:1757–62.
Article11. Iwasaki M, Hinotsu S, Katsura J. Clinical trials and approval of anti-cancer agents. Jpn J Clin Oncol. 2010; 40 Suppl 1:i65–9.
Article12. Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004; 304:1497–500.13. Shigematsu H, Lin L, Takahashi T, Nomura M, Suzuki M, Wistuba II, et al. Clinical and biological features associated with epidermal growth factor receptor gene mutations in lung cancers. J Natl Cancer Inst. 2005; 97:339–46.
Article14. O’Donnell PH, Dolan ME. Cancer pharmacoethnicity: ethnic differences in susceptibility to the effects of chemotherapy. Clin Cancer Res. 2009; 15:4806–14.
Article15. Edge S, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC cancer staging manual. 7th ed. New York: Springer;2010.16. Pirker R, Pereira JR, von Pawel J, Krzakowski M, Ramlau R, Park K, et al. EGFR expression as a predictor of survival for first-line chemotherapy plus cetuximab in patients with advanced non-small-cell lung cancer: analysis of data from the phase 3 FLEX study. Lancet Oncol. 2012; 13:33–42.
Article17. Segaert S, Van Cutsem E. Clinical signs, pathophysiology and management of skin toxicity during therapy with epidermal growth factor receptor inhibitors. Ann Oncol. 2005; 16:1425–33.
Article18. Chen P, Wang L, Li H, Liu B, Zou Z. Incidence and risk of hypomagnesemia in advanced cancer patients treated with cetuximab: a meta-analysis. Oncol Lett. 2013; 5:1915–20.
Article19. Petrelli F, Cabiddu M, Borgonovo K, Barni S. Risk of venous and arterial thromboembolic events associated with anti-EGFR agents: a meta-analysis of randomized clinical trials. Ann Oncol. 2012; 23:1672–9.
Article20. Socinski MA, Bondarenko I, Karaseva NA, Makhson AM, Vynnychenko I, Okamoto I, et al. Weekly nab-paclitaxel in combination with carboplatin versus solvent-based paclitaxel plus carboplatin as first-line therapy in patients with advanced non-small-cell lung cancer: final results of a phase III trial. J Clin Oncol. 2012; 30:2055–62.
Article21. Brahmer J, Reckamp KL, Baas P, Crino L, Eberhardt WE, Poddubskaya E, et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. 2015; 373:123–35.
Article22. Li S, Kussie P, Ferguson KM. Structural basis for EGF receptor inhibition by the therapeutic antibody IMC-11F8. Structure. 2008; 16:216–27.
Article23. Pujol JL, Pirker R, Lynch TJ, Butts CA, Rosell R, Shepherd FA, et al. Meta-analysis of individual patient data from randomized trials of chemotherapy plus cetuximab as first-line treatment for advanced non-small cell lung cancer. Lung Cancer. 2014; 83:211–8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Gemcitabine/Cisplatin Combination Chemotherapy in Advanced non-Small Cell lung Cancer
- A Comparison of Gemcitabine in Two Doses for Stage III or IV Non-small Cell Lung Cancer : a Multi-Institutional Phase II Study
- 3-week-scheduled combination chemotherapy of gemcitabine and cisplatin in patients with advanced NSCLC
- Efficacy of Combination Chemotherapy with Paclitaxel and Cisplatin in Patients with Advanced Non-Small Cell Lung Cancer
- Three-Weekly Gemcitabine Plus Cisplatin Chemotherapy in Patients with Locally Advanced or Metastatic Non-small-cell Lung Cancer: Phase II Study of the Korean Association for the Study of Lung Cancer