J Periodontal Implant Sci.
2017 Oct;47(5):328-338. 10.5051/jpis.2017.47.5.328.
Periodontal inflamed surface area as a novel numerical variable describing periodontal conditions
- Affiliations
-
- 1Department of Periodontology, Section of Dentistry, Seoul National University Bundang Hospital, Seongnam, Korea. periolee@gmail.com
- 2Division of Statistics, Medical Research Collaborating Center, Seoul National University Bundang Hospital, Seongnam, Korea.
- 3Department of Periodontology, Dankook University Jukjeon Dental Hospital, Yongin, Korea.
- 4Department of Oral and Maxillofacial Surgery, Section of Dentistry, Seoul National University Bundang Hospital, Seongnam, Korea.
- 5Division of Rheumatology, Department of Internal Medicine, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea.
- 6Department of Molecular Medicine and Biopharmaceutical Sciences, BK21 Plus Graduate School of Convergence Science and Technology, Medical Research Institute, Seoul National University College of Medicine, Seoul, Korea.
- 7Department of Internal Medicine, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea.
- 8Division of Allergy and Clinical Immunology, Department of Internal Medicine, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea.
- KMID: 2392719
- DOI: http://doi.org/10.5051/jpis.2017.47.5.328
Abstract
- PURPOSE
A novel index, the periodontal inflamed surface area (PISA), represents the sum of the periodontal pocket depth of bleeding on probing (BOP)-positive sites. In the present study, we evaluated correlations between PISA and periodontal classifications, and examined PISA as an index integrating the discrete conventional periodontal indexes.
METHODS
This study was a cross-sectional subgroup analysis of data from a prospective cohort study investigating the association between chronic periodontitis and the clinical features of ankylosing spondylitis. Data from 84 patients without systemic diseases (the control group in the previous study) were analyzed in the present study.
RESULTS
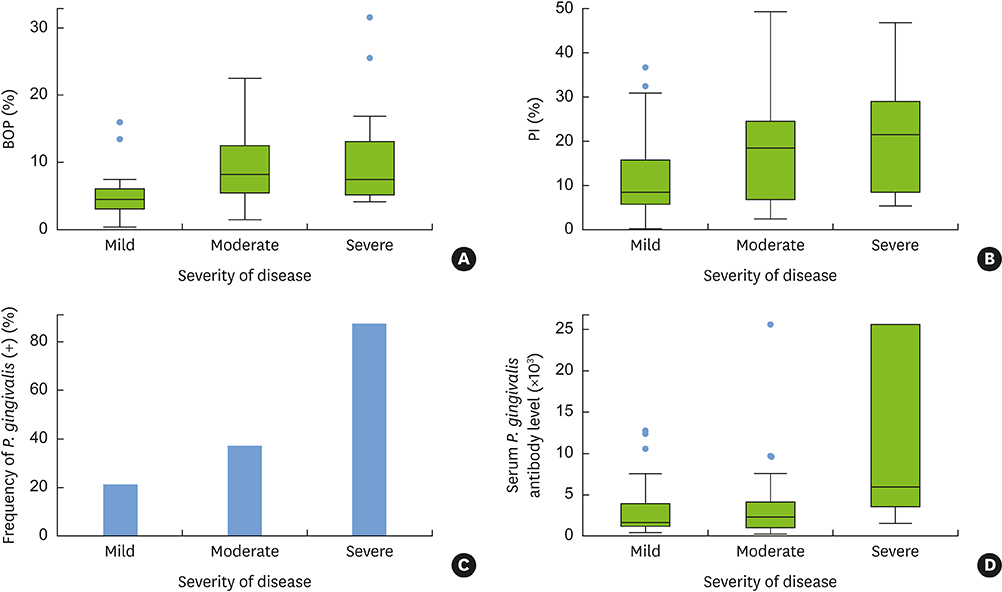
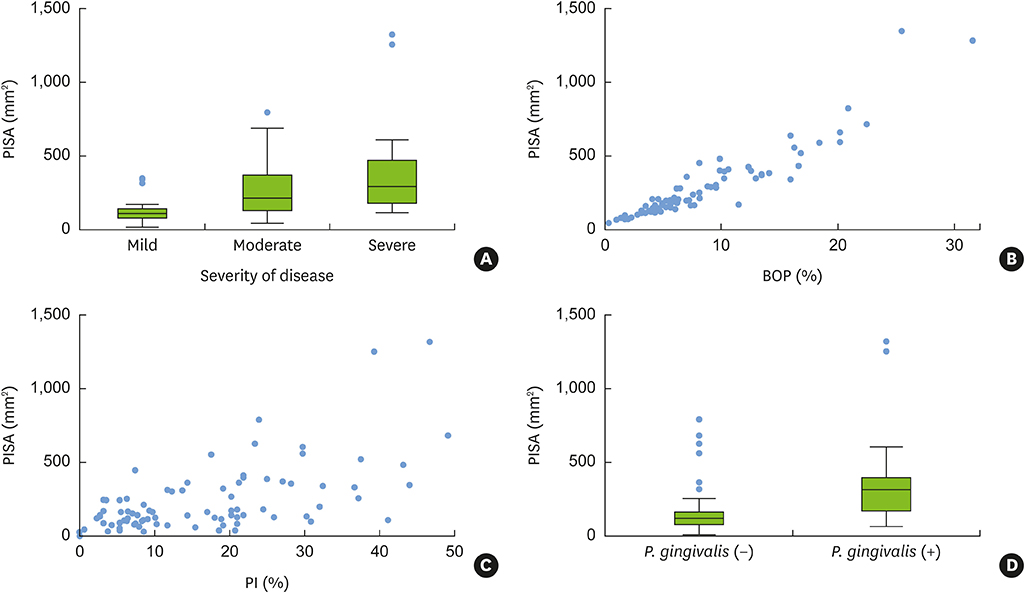
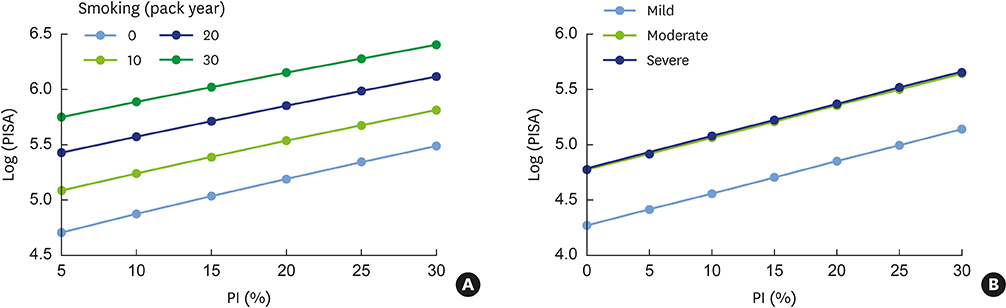
PISA values were positively correlated with conventional periodontal classifications (Spearman correlation coefficient=0.52; P<0.01) and with periodontal indexes, such as BOP and the plaque index (PI) (r=0.94; P<0.01 and r=0.60; P<0.01, respectively; Pearson correlation test). Porphyromonas gingivalis (P. gingivalis) expression and the presence of serum P. gingivalis antibodies were significant factors affecting PISA values in a simple linear regression analysis, together with periodontal classification, PI, bleeding index, and smoking, but not in the multivariate analysis. In the multivariate linear regression analysis, PISA values were positively correlated with the quantity of current smoking, PI, and severity of periodontal disease.
CONCLUSIONS
PISA integrates multiple periodontal indexes, such as probing pocket depth, BOP, and PI into a numerical variable. PISA is advantageous for quantifying periodontal inflammation and plaque accumulation.
Keyword
MeSH Terms
Figure
Reference
-
1. Consensus report. Periodontal diseases: pathogenesis and microbial factors. Ann Periodontol. 1996; 1:926–932.2. Kinane D, Bouchard P. Group E of European Workshop on Periodontology. Periodontal diseases and health: Consensus Report of the Sixth European Workshop on Periodontology. J Clin Periodontol. 2008; 35:333–337.
Article3. Kawada M, Yoshida A, Suzuki N, Nakano Y, Saito T, Oho T, et al. Prevalence of Porphyromonas gingivalis in relation to periodontal status assessed by real-time PCR. Oral Microbiol Immunol. 2004; 19:289–292.
Article4. Socransky SS, Haffajee AD. Periodontal microbial ecology. Periodontol 2000. 2005; 38:135–187.
Article5. Slots J. Casual or causal relationship between periodontal infection and non-oral disease? J Dent Res. 1998; 77:1764–1765.
Article6. Haffajee AD, Socransky SS, Lindhe J, Kent RL, Okamoto H, Yoneyama T. Clinical risk indicators for periodontal attachment loss. J Clin Periodontol. 1991; 18:117–125.
Article7. Brown LF, Beck JD, Rozier RG. Incidence of attachment loss in community-dwelling older adults. J Periodontol. 1994; 65:316–323.
Article8. Hajishengallis G, Liang S, Payne MA, Hashim A, Jotwani R, Eskan MA, et al. Low-abundance biofilm species orchestrates inflammatory periodontal disease through the commensal microbiota and complement. Cell Host Microbe. 2011; 10:497–506.
Article9. American Academy of Periodontology Task Force report on the update to the 1999 classification of periodontal diseases and conditions. J Periodontol. 2015; 86:835–838.10. Armitage GC. Development of a classification system for periodontal diseases and conditions. Ann Periodontol. 1999; 4:1–6.
Article11. Page RC, Eke PI. Case definitions for use in population-based surveillance of periodontitis. J Periodontol. 2007; 78:1387–1399.
Article12. Gilbert GH, Shelton BJ, Chavers LS, Bradford EH Jr. Predicting tooth loss during a population-based study: role of attachment level in the presence of other dental conditions. J Periodontol. 2002; 73:1427–1436.
Article13. McGuire MK, Nunn ME. Prognosis versus actual outcome. II. The effectiveness of clinical parameters in developing an accurate prognosis. J Periodontol. 1996; 67:658–665.
Article14. Nesse W, Abbas F, van der Ploeg I, Spijkervet FK, Dijkstra PU, Vissink A. Periodontal inflamed surface area: quantifying inflammatory burden. J Clin Periodontol. 2008; 35:668–673.
Article15. Greenstein G, Caton J, Polson AM. Histologic characteristics associated with bleeding after probing and visual signs of inflammation. J Periodontol. 1981; 52:420–425.
Article16. Lang NP, Joss A, Orsanic T, Gusberti FA, Siegrist BE. Bleeding on probing. A predictor for the progression of periodontal disease? J Clin Periodontol. 1986; 13:590–596.
Article17. Kang EH, Lee JT, Lee HJ, Lee JY, Chang SH, Cho HJ, et al. Chronic periodontitis is associated with spinal dysmobility in patients with ankylosing spondylitis. J Periodontol. 2015; 86:1303–1313.
Article18. O’Leary TJ, Drake RB, Naylor JE. The plaque control record. J Periodontol. 1972; 43:38.
Article19. Ainamo J, Bay I. Problems and proposals for recording gingivitis and plaque. Int Dent J. 1975; 25:229–235.20. Atieh MA. Accuracy of real-time polymerase chain reaction versus anaerobic culture in detection of Aggregatibacter actinomycetemcomitans and Porphyromonas gingivalis: a meta-analysis. J Periodontol. 2008; 79:1620–1629.
Article21. Golub LM, Kleinberg I. Gingival crevicular fluid: a new diagnostic aid in managing the periodontal patient. Oral Sci Rev. 1976; 49–61.22. Park SN, Park JY, Kook JK. Development of Porphyromonas gingivalis-specific quantitative real-time PCR primers based on the nucleotide sequence of rpoB. J Microbiol. 2011; 49:315–319.
Article23. Good PI, Hardin JW. Common errors in statistics (and how to avoid them). 3rd ed. Hoboken (NJ): Wiley;2009.24. Dietrich T, Sharma P, Walter C, Weston P, Beck J. The epidemiological evidence behind the association between periodontitis and incident atherosclerotic cardiovascular disease. J Clin Periodontol. 2013; 40:Suppl 14. S70–84.
Article25. Nesse W, Linde A, Abbas F, Spijkervet FK, Dijkstra PU, de Brabander EC, et al. Dose-response relationship between periodontal inflamed surface area and HbA1c in type 2 diabetics. J Clin Periodontol. 2009; 36:295–300.
Article26. Iwasaki M, Taylor GW, Nesse W, Vissink A, Yoshihara A, Miyazaki H. Periodontal disease and decreased kidney function in Japanese elderly. Am J Kidney Dis. 2012; 59:202–209.
Article27. Radwan-Oczko M, Jaworski A, Duś I, Plonek T, Szulc M, Kustrzycki W. Porphyromonas gingivalis in periodontal pockets and heart valves. Virulence. 2014; 5:575–580.
Article28. Belibasakis G, Thurnheer T, Bostanci N. Porphyromonas gingivalis: a heartful oral pathogen? Virulence. 2014; 5:463–464.29. Rosenstein ED, Greenwald RA, Kushner LJ, Weissmann G. Hypothesis: the humoral immune response to oral bacteria provides a stimulus for the development of rheumatoid arthritis. Inflammation. 2004; 28:311–318.
Article30. Mikuls TR, Payne JB, Yu F, Thiele GM, Reynolds RJ, Cannon GW, et al. Periodontitis and Porphyromonas gingivalis in patients with rheumatoid arthritis. Arthritis Rheumatol. 2014; 66:1090–1100.
Article31. Hitchon CA, Chandad F, Ferucci ED, Willemze A, Ioan-Facsinay A, van der Woude D, et al. Antibodies to Porphyromonas gingivalis are associated with anticitrullinated protein antibodies in patients with rheumatoid arthritis and their relatives. J Rheumatol. 2010; 37:1105–1112.
Article32. Lappin DF, Apatzidou D, Quirke AM, Oliver-Bell J, Butcher JP, Kinane DF, et al. Influence of periodontal disease, Porphyromonas gingivalis and cigarette smoking on systemic anti-citrullinated peptide antibody titres. J Clin Periodontol. 2013; 40:907–915.
Article33. Casarin RC, Del Peloso Ribeiro É, Mariano FS, Nociti FH Jr, Casati MZ, Gonçalves RB. Levels of Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, inflammatory cytokines and species-specific immunoglobulin G in generalized aggressive and chronic periodontitis. J Periodontal Res. 2010; 45:635–642.
Article34. Mooney J, Adonogianaki E, Riggio MP, Takahashi K, Haerian A, Kinane DF. Initial serum antibody titer to Porphyromonas gingivalis influences development of antibody avidity and success of therapy for chronic periodontitis. Infect Immun. 1995; 63:3411–3416.
Article35. Wilton JM, Hurst TJ, Sterne JA. Elevated opsonic activity for Porphyromonas (Bacteroides) gingivalis in serum from patients with a history of destructive periodontal disease. A case: control study. J Clin Periodontol. 1993; 20:563–569.
Article36. Fransson C, Berglundh T, Lindhe J. The effect of age on the development of gingivitis. Clinical, microbiological and histological findings. J Clin Periodontol. 1996; 23:379–385.
Article37. Kinane DF, Chestnutt IG. Smoking and periodontal disease. Crit Rev Oral Biol Med. 2000; 11:356–365.
Article38. Müller HP, Stadermann S, Heinecke A. Longitudinal association between plaque and gingival bleeding in smokers and non-smokers. J Clin Periodontol. 2002; 29:287–294.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Waiting for innovations in periodontal disease diagnosis
- Reply on "Relationship between maternal periodontal disease and Apgar score of newborns"
- An analytical on the periodontal conditions and treatment in leprous patients
- Future endeavors needed to close the socioeconomic gap in periodontal health
- ULTRASTRUCTURAL INVESTIGATIONS OF THE INTERFACE BETWEEN CULTURED PERIODONTAL LIGAMENT CELLS AND TITANIUM