Allergy Asthma Respir Dis.
2017 Jul;5(4):185-192. 10.4168/aard.2017.5.4.185.
The KAAACI Standardization Committee Report on the procedure and application of fractional exhaled nitric oxide measurement
- Affiliations
-
- 1Department of Internal Medicine, Kangwon National University School of Medicine, Chuncheon, Korea.
- 2Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea. swj0126@gmail.com
- 3Department of Internal Medicine, Ewha Womans University School of Medicine, Seoul, Korea.
- 4Department of Internal Medicine, Armed Forces Capital Hospital, Seongnam, Korea.
- 5Department of Internal Medicine, Seoul Metropolitan Government - Seoul National University Boramae Medical Center, Seoul, Korea.
- 6Department of Internal Medicine, Chung-Ang University College of Medicine, Seoul, Korea.
- 7Department of Pediatrics, Yonsei University Wonju College of Medicine, Wonju, Korea.
- 8Department of Pediatrics, Seoul National University College of Medicine, Seoul, Korea.
- 9Department of Allergy and Clinical Immunology, Ajou University School of Medicine, Suwon, Korea.
- 10Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- 11Department of Internal Medicine, Hanyang University College of Medicine, Seoul, Korea.
- 12Division of Allergy, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 2387645
- DOI: http://doi.org/10.4168/aard.2017.5.4.185
Abstract
- Fractional exhaled nitric oxide (FeNO) is a biomarker for type 2 inflammation in the airways, and its measurement has the distinct strengths of being simple, rapid, convenient, and reproducible. Since the discovery of FeNO in the 1990s, its measurement tools have been extensively developed and validated by several researchers. In particular, FeNO showed the potential to support the diagnosis of asthma and eosinophilic bronchitis. Since it has begun to be widely utilized in clinical research areas as well as daily practice, we summarized the standardized diagnostic procedures and suggested the clinical application of FeNO measurement in Korean population.
MeSH Terms
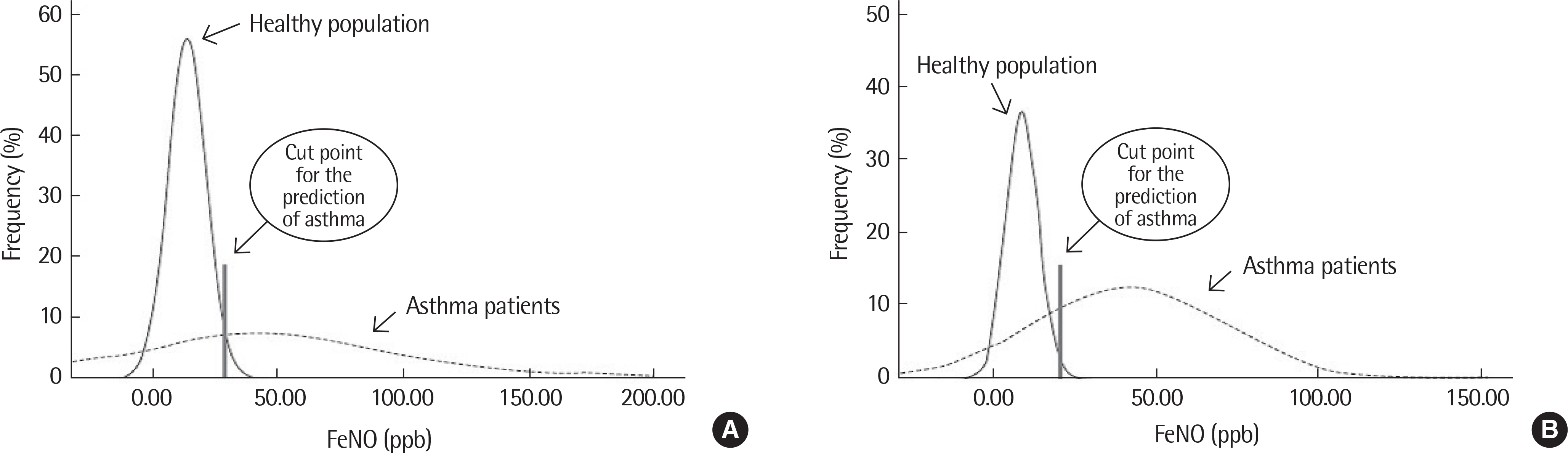
Figure
Cited by 1 articles
-
KAAACI Evidence-Based Clinical Practice Guidelines for Chronic Cough in Adults and Children in Korea
Dae Jin Song, Woo-Jung Song, Jae-Woo Kwon, Gun-Woo Kim, Mi-Ae Kim, Mi-Yeong Kim, Min-Hye Kim, Sang-Ha Kim, Sang-Heon Kim, Sang Hyuck Kim, Sun-Tae Kim, Sae-Hoon Kim, Ja Kyoung Kim, Joo-Hee Kim, Hyun Jung Kim, Hyo-Bin Kim, Kyung-Hee Park, Jae Kyun Yoon, Byung-Jae Lee, Seung-Eun Lee, Young Mok Lee, Yong Ju Lee, Kyung-Hwan Lim, You Hoon Jeon, Eun-Jung Jo, Young-Koo Jee, Hyun Jung Jin, Sun Hee Choi, Gyu Young Hur, Sang-Heon Cho, Sang-Hoon Kim, Dae Hyun Lim
Allergy Asthma Immunol Res. 2018;10(6):591-613. doi: 10.4168/aair.2018.10.6.591.
Reference
-
1. Alving K, Malinovschi A. Basic aspects of exhaled nitric oxide. Eur Respir Monogr. 2010; 49:1–31.
Article2. Persson MG, Zetterstrom O, Agrenius V, Ihre E, Gustafsson LE. Single-breath nitric oxide measurements in asthmatic patients and smokers. Lancet. 1994; 343:146–7.
Article3. Massaro AF, Gaston B, Kita D, Fanta C, Stamler JS, Drazen JM. Expired nitric oxide levels during treatment of acute asthma. Am J Respir Crit Care Med. 1995; 152:800–3.
Article4. Kharitonov SA, Yates DH, Chung KF, Barnes PJ. Changes in the dose of inhaled steroid affect exhaled nitric oxide levels in asthmatic patients. Eur Respir J. 1996; 9:196–201.
Article5. Kim MY, Jo EJ, Lee SE, Lee SY, Song WJ, Kim TW, et al. Reference ranges for induced sputum eosinophil counts in Korean adult population. Asia Pac Allergy. 2014; 4:149–55.
Article6. Ignarro LJ, Buga GM, Wood KS, Byrns RE, Chaudhuri G. Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc Natl Acad Sci U S A. 1987; 84:9265–9.
Article7. Palmer RM, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature. 1988; 333:664–6.
Article8. Nathan C, Xie QW. Nitric oxide synthases: roles, tolls, and controls. Cell. 1994; 78:915–8.
Article9. Kim SH, Yoon HJ. Use of the exhaled nitric oxide for management of asthma and respiratory diseases. Korean J Med. 2008; 74:579–86.10. Morris SM Jr, Billiar TR. New insights into the regulation of inducible nitric oxide synthesis. Am J Physiol. 1994; 266(6 Pt 1):E829–39.
Article11. Dweik RA, Comhair SA, Gaston B, Thunnissen FB, Farver C, Thomassen MJ, et al. NO chemical events in the human airway during the immediate and late antigen-induced asthmatic response. Proc Natl Acad Sci U S A. 2001; 98:2622–7.
Article12. Guo FH, Comhair SA, Zheng S, Dweik RA, Eissa NT, Thomassen MJ, et al. Molecular mechanisms of increased nitric oxide (NO) in asthma: evidence for transcriptional and post-translational regulation of NO synthesis. J Immunol. 2000; 164:5970–80.
Article13. Payne DN, Adcock IM, Wilson NM, Oates T, Scallan M, Bush A. Relationship between exhaled nitric oxide and mucosal eosinophilic inflammation in children with difficult asthma, after treatment with oral prednisolone. Am J Respir Crit Care Med. 2001; 164(8 Pt 1):1376–81.
Article14. Warke TJ, Fitch PS, Brown V, Taylor R, Lyons JD, Ennis M, et al. Exhaled nitric oxide correlates with airway eosinophils in childhood asthma. Thorax. 2002; 57:383–7.
Article15. Berry MA, Shaw DE, Green RH, Brightling CE, Wardlaw AJ, Pavord ID. The use of exhaled nitric oxide concentration to identify eosinophilic airway inflammation: an observational study in adults with asthma. Clin Exp Allergy. 2005; 35:1175–9.
Article16. Paoliello-Paschoalato AB, Oliveira SH, Cunha FQ. Interleukin 4 induces the expression of inducible nitric oxide synthase in eosinophils. Cytokine. 2005; 30:116–24.
Article17. Haldar P, Brightling CE, Hargadon B, Gupta S, Monteiro W, Sousa A, et al. Mepolizumab and exacerbations of refractory eosinophilic asthma. N Engl J Med. 2009; 360:973–84.
Article18. American Thoracic Society; European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am J Respir Crit Care Med. 2005; 171:912–30.19. Yune S, Lee JY, Choi DC, Lee BJ. Fractional exhaled nitric oxide: comparison between portable devices and correlation with sputum eosinophils. Allergy Asthma Immunol Res. 2015; 7:404–8.
Article20. Harnan SE, Tappenden P, Essat M, Gomersall T, Minton J, Wong R, et al. Measurement of exhaled nitric oxide concentration in asthma: a systematic review and economic evaluation of NIOX MINO, NIOX VERO and NObreath. Health Technol Assess. 2015; 19:1–330.
Article21. de Gouw HW, Hendriks J, Woltman AM, Twiss IM, Sterk PJ. Exhaled nitric oxide (NO) is reduced shortly after bronchoconstriction to direct and indirect stimuli in asthma. Am J Respir Crit Care Med. 1998; 158:315–9.
Article22. Silkoff PE, Wakita S, Chatkin J, Ansarin K, Gutierrez C, Caramori M, et al. Exhaled nitric oxide after beta2-agonist inhalation and spirometry in asthma. Am J Respir Crit Care Med. 1999; 159:940–4.23. de Gouw HW, Grunberg K, Schot R, Kroes AC, Dick EC, Sterk PJ. Relationship between exhaled nitric oxide and airway hyperresponsiveness following experimental rhinovirus infection in asthmatic subjects. Eur Respir J. 1998; 11:126–32.
Article24. Kharitonov SA, Yates D, Barnes PJ. Increased nitric oxide in exhaled air of normal human subjects with upper respiratory tract infections. Eur Respir J. 1995; 8:295–7.
Article25. Olin AC, Aldenbratt A, Ekman A, Ljungkvist G, Jungersten L, Alving K, et al. Increased nitric oxide in exhaled air after intake of a nitrate-rich meal. Respir Med. 2001; 95:153–8.
Article26. Bruce C, Yates DH, Thomas PS. Caffeine decreases exhaled nitric oxide. Thorax. 2002; 57:361–3.
Article27. Byrnes CA, Dinarevic S, Busst CA, Shinebourne EA, Bush A. Effect of measurement conditions on measured levels of peak exhaled nitric oxide. Thorax. 1997; 52:697–701.
Article28. Kharitonov SA, Robbins RA, Yates D, Keatings V, Barnes PJ. Acute and chronic effects of cigarette smoking on exhaled nitric oxide. Am J Respir Crit Care Med. 1995; 152:609–12.
Article29. Bergeron C, Boulet LP, Page N, Laviolette M, Zimmermann N, Rothen-berg ME, et al. Influence of cigarette smoke on the arginine pathway in asthmatic airways: increased expression of arginase I. J Allergy Clin Immunol. 2007; 119:391–7.30. Robbins RA, Millatmal T, Lassi K, Rennard S, Daughton D. Smoking cessation is associated with an increase in exhaled nitric oxide. Chest. 1997; 112:313–8.
Article31. Terada A, Fujisawa T, Togashi K, Miyazaki T, Katsumata H, Atsuta J, et al. Exhaled nitric oxide decreases during exercise-induced bronchoconstriction in children with asthma. Am J Respir Crit Care Med. 2001; 164(10 Pt 1):1879–84.
Article32. Bisgaard H, Loland L, Oj JA. NO in exhaled air of asthmatic children is reduced by the leukotriene receptor antagonist montelukast. 1999; 160:1227–31.33. Davis BE, Illamperuma C, Gauvreau GM, Watson RM, O'Byrne PM, De-schesnes F, et al. Single-dose desloratadine and montelukast and allergen-induced late airway responses. Eur Respir J. 2009; 33:1302–8.
Article34. Montuschi P, Mondino C, Koch P, Ciabattoni G, Barnes PJ, Baviera G. Effects of montelukast treatment and withdrawal on fractional exhaled nitric oxide and lung function in children with asthma. Chest. 2007; 132:1876–81.
Article35. Fardon TC, Lee DK, Hodge MR, Lipworth BJ. Addition of fexofenadine to inhaled corticosteroid therapy to reduce inflammatory biomarkers in atopic asthma. Ann Allergy Asthma Immunol. 2005; 95:259–65.
Article36. Lim S, Tomita K, Caramori G, Jatakanon A, Oliver B, Keller A, et al. Low-dose theophylline reduces eosinophilic inflammation but not exhaled nitric oxide in mild asthma. Am J Respir Crit Care Med. 2001; 164:273–6.
Article37. Song WJ, Kwon JW, Kim EJ, Lee SM, Kim SH, Lee SY, et al. Clinical application of exhaled nitric oxide measurements in a korean population. Allergy Asthma Immunol Res. 2015; 7:3–13.
Article38. Dweik RA, Boggs PB, Erzurum SC, Irvin CG, Leigh MW, Lundberg JO, et al. An official ATS clinical practice guideline: interpretation of exhaled nitric oxide levels (FENO) for clinical applications. Am J Respir Crit Care Med. 2011; 184:602–15.
Article39. Jo EJ, Song WJ, Kim TW, Park HW, Chang YS, Kim TB, et al. Reference ranges and determinant factors for exhaled nitric oxide in a healthy korean elderly population. Allergy Asthma Immunol Res. 2014; 6:504–10.
Article40. McGrath KW, Icitovic N, Boushey HA, Lazarus SC, Sutherland ER, Chinchilli VM, et al. A large subgroup of mild-to-moderate asthma is persistently noneosinophilic. Am J Respir Crit Care Med. 2012; 185:612–9.
Article41. Song WJ, Kim HJ, Shim JS, Won HK, Kang SY, Sohn KH, et al. Diagnostic accuracy of fractional exhaled nitric oxide measurement in predicting cough-variant asthma and eosinophilic bronchitis in adults with chronic cough: A systematic review and metaanalysis. J Allergy Clin Immunol 2017 Jan 11 [Epub]. pii: S0091-6749(17)30006-4.https://doi.org/10.1016/j.jaci.2016.11.037.42. Karrasch S, Linde K, Rucker G, Sommer H, Karsch-Volk M, Kleijnen J, et al. Accuracy of FENO for diagnosing asthma: a systematic review. Thorax. 2017; 72:109–16.43. Crapo RO, Casaburi R, Coates AL, Enright PL, Hankinson JL, Irvin CG, et al. Guidelines for methacholine and exercise challenge testing-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000; 161:309–29.44. Woo SI, Lee JH, Kim H, Kang JW, Sun YH, Hahn YS. Utility of fractional exhaled nitric oxide F E NO measurements in diagnosing asthma. Respir Med. 2012; 106:1103–9.45. Park SH, Kim DH, Koh YI. Usefulness of fractional exhaled nitric oxide for the diagnosis of asthma and its assessment of asthma control. Korean J Asthma Allergy Clin Immunol. 2012; 32:83–91.46. Mattes J, Storm van's Gravesande K, Reining U, Alving K, Ihorst G, Hen-schen M, et al. NO in exhaled air is correlated with markers of eosinophilic airway inflammation in corticosteroid-dependent childhood asthma. Eur Respir J. 1999; 13:1391–5.
Article47. Jatakanon A, Lim S, Kharitonov SA, Chung KF, Barnes PJ. Correlation between exhaled nitric oxide, sputum eosinophils, and methacholine responsiveness in patients with mild asthma. Thorax. 1998; 53:91–5.
Article48. Jones SL, Kittelson J, Cowan JO, Flannery EM, Hancox RJ, McLachlan CR, et al. The predictive value of exhaled nitric oxide measurements in assessing changes in asthma control. Am J Respir Crit Care Med. 2001; 164:738–43.
Article49. Korevaar DA, Westerhof GA, Wang J, Cohen JF, Spijker R, Sterk PJ, et al. Diagnostic accuracy of minimally invasive markers for detection of airway eosinophilia in asthma: a systematic review and metaanalysis. Lancet Respir Med. 2015; 3:290–300.
Article50. Kim YH, Kim KW, Baek J, Park HB, Kim H, Song KJ, et al. Usefulness of impulse oscillometry and fractional exhaled nitric oxide in children with Eosinophilic bronchitis. Pediatr Pulmonol. 2013; 48:221–8.
Article51. Chaudhuri R, McMahon AD, Thomson LJ, MacLeod KJ, McSharry CP, Livingston E, et al. Effect of inhaled corticosteroids on symptom severity and sputum mediator levels in chronic persistent cough. J Allergy Clin Immunol. 2004; 113:1063–70.
Article52. Song WJ, Won HK, Moon SD, Chung SJ, Kang SY, Sohn KH, et al. Could fractional exhaled nitric oxide test be useful in predicting inhaled corticosteroid responsiveness in chronic cough? A systematic review. J Allergy Clin Immunol Pract. 2017; 5:135–43.e1.
Article53. Oh MJ, Lee JY, Lee BJ, Choi DC. Exhaled nitric oxide measurement is useful for the exclusion of nonasthmatic eosinophilic bronchitis in patients with chronic cough. Chest. 2008; 134:990–5.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical application of fractional exhaled nitric oxide in pediatric allergic rhinitis
- Measurement and Interpretation of Fractional Exhaled Nitric Oxide
- Utility of Fractional Exhaled Nitric Oxide in the Diagnosis of Asthma and the Assessment of Asthma Control
- Measurements of fractional exhaled nitric oxide in pediatric asthma
- Use of the exhaled nitric oxide for management of asthma and respiratory diseases