Infect Chemother.
2017 Jun;49(2):146-150. 10.3947/ic.2017.49.2.146.
Henoch-Schönlein Purpura with Concurrent Cytomegalovirus Duodenitis
- Affiliations
-
- 1Division of Oncology, Department of Internal Medicine, Seoul St. Mary's Hospital, The Catholic University of Korea, Seoul, Korea. jinkang@catholic.ac.kr, befamiliar@catholic.ac.kr
- 2Department of Pathology, Seoul St. Mary's Hospital, The Catholic University of Korea, Seoul, Korea.
- KMID: 2383938
- DOI: http://doi.org/10.3947/ic.2017.49.2.146
Abstract
- Henoch-Schönlein purpura (HSP) is a systemic vasculitis involving the small vessels with distinct clinical features. The etiology of HSP is diverse, and viral infection is one of the many predisposing factors. Cytomegalovirus (CMV) infection mostly affects immune-suppressed patients, but rarely patients with normal immunity can also be affected. Authors experienced a case of HSP patient, with underlying small-cell lung cancer (SCLC) with CMV duodenitis. This is a rare case of HSP diagnosed in SCLC patient with predisposing factor of CMV infection.
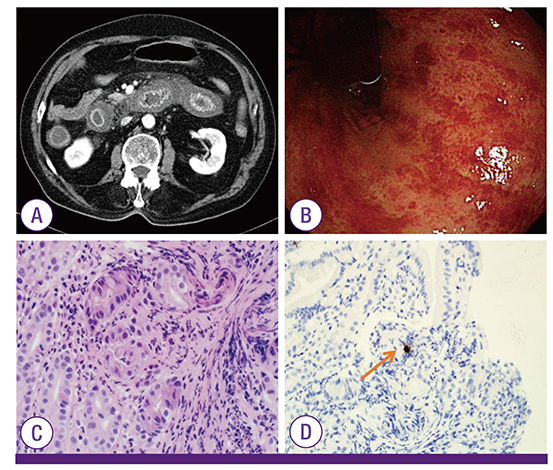
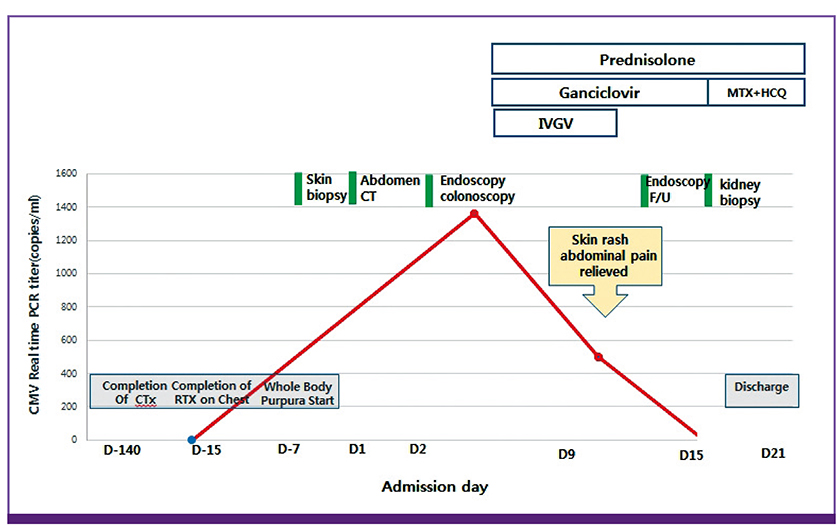
Figure
Reference
-
1. Gedalia A. Henoch-Schönlein purpura. Curr Rheumatol Rep. 2004; 6:195–202.
Article2. Zhang XD, Yang SY, Li W, Ming ZJ, Hou YL, Niu ZQ, Zhang YP. Adult Henoch-Schönlein purpura associated with small cell lung cancer: a case report and review of the literature. Oncol Lett. 2013; 5:1927–1930.
Article3. Meyer MF, Hellmich B, Kotterba S, Schatz H. Cytomegalovirus infection in systemic necrotizing vasculitis: causative agent or opportunistic infection? Rheumatol Int. 2000; 20:35–38.
Article4. Lancini D, Faddy HM, Flower R, Hogan C. Cytomegalovirus disease in immunocompetent adults. Med J Aust. 2014; 201:578–580.
Article5. Jennette JC, Falk RJ, Andrassy K, Bacon PA, Churg J, Gross WL, Hagen EC, Hoffman GS, Hunder GG, Kallenberg CG, Mccluskey RT, Alberto Sinico R, Rees AJ, Van Es LA, Waldherr R, Wiik A. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheum. 1994; 37:187–192.6. Kraft DM, McKee D, Scott C. Henoch-Schönlein purpura: a review. Am Fam Physician. 1998; 58:405–408.7. Fain O, Hamidou M, Cacoub P, Godeau B, Wechsler B, Pariès J, Stirnemann J, Morin AS, Gatfosse M, Hanslik T, Belmatoug N, Blétry O, Cevallos R, Delevaux I, Fisher E, Hayem G, Kaplan G, Le Hello C, Mouthon L, Larroche C, Lemaire V, Piette AM, Piette JC, Ponge T, Puechal X, Rossert J, Sarrot-Reynauld F, Sicard D, Ziza JM, Kahn MF, Guillevin L. Vasculitides associated with malignancies: analysis of sixty patients. Arthritis Rheum. 2007; 57:1473–1480.
Article8. Aktas B, Topcuoglu P, Kurt OK, Ensari A, Demirer T. Severe Henoch-Schönlein purpura induced by cytarabine. Ann Pharmacother. 2009; 43:792–793.
Article9. Podjasek JO, Wetter DA, Pittelkow MR, Wada DA. Henoch-Schönlein purpura associated with solid-organ malignancies: three case reports and a literature review. Acta Derm Venereol. 2012; 92:388–392.
Article10. Sánchez-Guerrero J, Gutiérrez-Ureña S, Vidaller A, Reyes E, Iglesias A, Alarcón-Segovia D. Vasculitis as a paraneoplastic syndrome. Report of 11 cases and review of the literature. J Rheumatol. 1990; 17:1458–1462.11. Fortin PR. Vasculitides associated with malignancy. Curr Opin Rheumatol. 1996; 8:30–33.
Article12. Mitsui H, Shibagaki N, Kawamura T, Matsue H, Shimada S. A clinical study of Henoch- Schönlein purpura associated with malignancy. J Eur Acad Dermatol Venereol. 2009; 23:394–401.13. Taniguchi K, Watanabe N, Sato A, Jwa SC, Suzuki T, Yamanobe Y, Sago H, Kozuka K. Changes in cytomegalovirus seroprevalence in pregnant Japanese women-a 10-year single center study. J Clin Virol. 2014; 59:192–194.
Article14. Furui Y, Satake M, Hoshi Y, Uchida S, Suzuki K, Tadokoro K. Cytomegalovirus (CMV) seroprevalence in Japanese blood donors and high detection frequency of CMV DNA in elderly donors. Transfusion. 2013; 53:2190–2197.
Article15. Mandell BF, Calabrese LH. Infections and systemic vasculitis. Curr Opin Rheumatol. 1998; 10:51–57.
Article16. de Almeida JL, Campos LM, Paim LB, Leone C, Koch VH, Silva CA. Renal involvement in Henoch-Schönlein purpura: a multivariate analysis of initial prognostic factors. J Pediatr (Rio J). 2007; 83:259–266.
Article17. Pillebout E, Thervet E, Hill G, Alberti C, Vanhille P, Nochy D. Henoch-Schönlein purpura in adults: outcome and prognostic factors. J Am Soc Nephrol. 2002; 13:1271–1278.
Article18. Hung SP, Yang YH, Lin YT, Wang LC, Lee JH, Chiang BL. Clinical manifestations and outcomes of Henoch-Schönlein purpura: comparison between adults and children. Pediatr Neonatol. 2009; 50:162–168.
Article19. Kato-Okada S, Suzuki H, Inoue T, Kikuta T, Okada H. Successful prednisolone therapy in elderly patients with severe forms of henoch-schönlein purpura nephritis. Jpn Clin Med. 2015; 6:5–7.
Article20. Satta R, Biondi G. Vasculitis and infectious diseases. G Ital Dermatol Venereol. 2015; 150:211–220.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Usefulness of Neutrophil-Lymphocyte Ratio as a Predictor of Henoch–Schönlein Nephritis
- A Case of Henoch-Schönlein Purpura Associated with Mycoplasma Pneumoniae Pneumonia
- A Case of Pulmonary Infarction Probably Related with Henoch-Schönlein Purpura
- Henoch-Schönlein Purpura Presenting as Intussusception
- Two Cases of Henoch-Sch nlein Purpura Complicated by Gastrointestinal Bleeding