Infect Chemother.
2017 Jun;49(2):109-116. 10.3947/ic.2017.49.2.109.
Emergence of Community-Genotype Methicillin-Resistant Staphylococcus aureus in Korean Hospitals: Clinical Characteristics of Nosocomial Infections by Community-Genotype Strain
- Affiliations
-
- 1Division of Infectious Diseases, Department of Internal Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 2Division of Infectious Diseases, Department of Internal Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. iddrchung@gmail.com
- 3Asia Pacific Foundation for Infectious Diseases (APFID), Seoul, Korea.
- 4Department of Laboratory Medicine and Genetics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 2383931
- DOI: http://doi.org/10.3947/ic.2017.49.2.109
Abstract
- BACKGROUND
As community-genotype methicillin-resistant Staphylococcus aureus (MRSA) strains spread into hospitals, the genotypes of the MRSA strains causing hospital-acquired (HA) infections have become more diverse. We describe clinical characteristics of nosocomial MRSA infections by a community-genotype of sequence type (ST) 72.
MATERIALS AND METHODS
A case-control study was designed among patients with HA-MRSA infections. Forty patients with infections caused by ST72-MRSA SCCmec type IV were selected as cases. Cases were matched to the controls with 106 patients infected with ST5/ST239 MRSA, which are representative hospital genotypes in Korea.
RESULTS
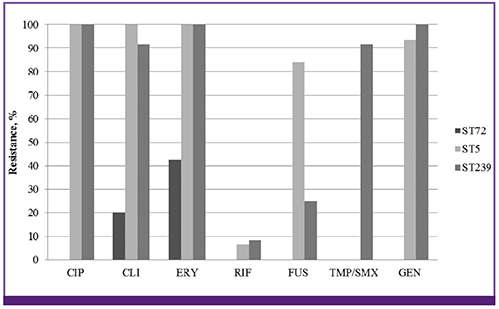
Patients infected with ST72 isolates were younger than those with ST5/ST239 isolates. Female gender predominated among ST72 MRSA group compared to ST5/ST239 MRSA group. Solid tumor was a more frequent underlying disease in MRSA infections by ST72 isolates, whereas underlying renal, lung, heart, and neurologic diseases were more frequently found in those by ST5/ST239 isolates. The most common type of infection was pneumonia in both ST72 and ST5/ST239 groups (45.0% vs. 51.9%), followed by skin and soft tissue infection (SSTI). Female gender and underlying solid tumor were identified to be independent predictors for MRSA infections by ST72 isolates. All-cause mortality rates (20.0% vs. 30.2%) were not different between the groups.
CONCLUSION
A community-genotype MRSA, ST72 isolate has emerged as a nosocomial pathogen presenting as hospital-acquired pneumonia and SSTI. Although differences in underlying disorders were found, the distribution of infection type and mortality rate did not differ between the groups.
MeSH Terms
Figure
Reference
-
1. Saiman L, O’Keefe M, Graham PL 3rd, Wu F, Saïd-Salim B, Kreiswirth B, LaSala A, Schlievert PM, Della-Latta P. Hospital transmission of community-acquired methicillin-resistant Staphylococcus aureus among postpartum women. Clin Infect Dis. 2003; 37:1313–1319.
Article2. Valsesia G, Rossi M, Bertschy S, Pfyffer GE. Emergence of SCCmec type IV and SCCmec type V methicillin-resistant Staphylococcus aureus containing the Panton-Valentine leukocidin genes in a large academic teaching hospital in central Switzerland: external invaders or persisting circulators? J Clin Microbiol. 2010; 48:720–727.
Article3. Seybold U, Kourbatova EV, Johnson JG, Halvosa SJ, Wang YF, King MD, Ray SM, Blumberg HM. Emergence of community-associated methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of health care-associated blood stream infections. Clin Infect Dis. 2006; 42:647–656.
Article4. Davis SL, Rybak MJ, Amjad M, Kaatz GW, McKinnon PS. Characteristics of patients with healthcare-associated infection due to SCCmec type IV methicillin-resistant Staphylococcus aureus . Infect Control Hosp Epidemiol. 2006; 27:1025–1031.
Article5. Strandén AM, Frei R, Adler H, Fluckiger U, Widmer AF. Emergence of SCCmec type IV as the most common type of methicillin-resistant Staphylococcus aureus in a university hospital. Infection. 2009; 37:44–48.
Article6. Vidal PM, Trindade PA, Garcia TO, Pacheco RL, Costa SF, Reinert C, Hiramatsu K, Mamizuka EM, Garcia CP, Levin AS. Differences between “classical” risk factors for infections caused by methicillin-resistant Staphylococcus aureus (MRSA) and risk factors for nosocomial bloodstream infections caused by multiple clones of the staphylococcal cassette chromosome mec type IV MRSA strain. Infect Control Hosp Epidemiol. 2009; 30:139–145.
Article7. Park SH, Park C, Yoo JH, Choi SM, Choi JH, Shin HH, Lee DG, Lee S, Kim J, Choi SE, Kwon YM, Shin WS. Emergence of community-associated methicillin-resistant Staphylococcus aureus strains as a cause of healthcare-associated bloodstream infections in Korea. Infect Control Hosp Epidemiol. 2009; 30:146–155.
Article8. Kim ES, Lee HJ, Chung GT, Lee YS, Shin DH, Jung SI, Song KH, Park WB, Kim NJ, Park KU, Kim EC, Oh MD, Kim HB. Molecular characterization of methicillin-resistant Staphylococcus aureus isolates in Korea. J Clin Microbiol. 2011; 49:1979–1982.
Article9. Park KH, Chong YP, Kim SH, Lee SO, Choi SH, Lee MS, Jeong JY, Woo JH, Kim YS. Community-associated MRSA strain ST72-SCCmecIV causing bloodstream infections: clinical outcomes and bacterial virulence factors. J Antimicrob Chemother. 2015; 70:1185–1192.
Article10. Joo EJ, Chung DR, Ha YE, Park SY, Kang SJ, Kim SH, Kang CI, Peck KR, Lee NY, Ko KS, Song JH. Community-associated Panton-Valentine leukocidin-negative meticillin-resistant Staphylococcus aureus clone (ST72-MRSA-IV) causing healthcare-associated pneumonia and surgical site infection in Korea. J Hosp Infect. 2012; 81:149–155.
Article11. Joo EJ, Chung DR, Ha YE, Park SY, Kim HA, Lim MH, Kim SH, Kang CI, Lee NY, Ko KS, Peck KR, Song JH. Clinical predictors of community-genotype ST72-methicillin-resistant Staphylococcus aureus-SCCmec type IV in patients with community-onset S. aureus infection. J Antimicrob Chemother. 2012; 67:1755–1759.
Article12. Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008; 36:309–332.
Article13. Clinical and Laboratory Standards Institutue (CLSI). Performance standards for antimicrobial susceptibility resting; 19th Informational Supplement M100-S19. Wayne, PA: CLSI;2009.14. Oliveira DC, de Lencastre H. Multiplex PCR strategy for rapid identification of structural types and variants of the mec element in methicillin-resistant Staphylococcus aureus . Antimicrob Agents Chemother. 2002; 46:2155–2161.
Article15. Milheirico C, Oliveira DC, de Lencastre H. Multiplex PCR strategy for subtyping the staphylococcal cassette chromosome mec type IV in methicillin-resistant Staphylococcus aureus: ‘SCCmec IV multiplex’. J Antimicrob Chemother. 2007; 60:42–48.16. Enright MC, Day NP, Davies CE, Peacock SJ, Spratt BG. Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J Clin Microbiol. 2000; 38:1008–1015.
Article17. Alberti KG, Zimmet P, Shaw J. Metabolic syndrome--a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet Med. 2006; 23:469–480.
Article18. Gillet Y, Issartel B, Vanhems P, Fournet JC, Lina G, Bes M, Vandenesch F, Piémont Y, Brousse N, Floret D, Etienne J. Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young immunocompetent patients. Lancet. 2002; 359:753–759.
Article19. Gillet Y, Vanhems P, Lina G, Bes M, Vandenesch F, Floret D, Etienne J. Factors predicting mortality in necrotizing community-acquired pneumonia caused by Staphylococcus aureus containing Panton-Valentine leukocidin. Clin Infect Dis. 2007; 45:315–321.
Article20. Kim SH, Park WB, Lee KD, Kang CI, Kim HB, Oh MD, Kim EC, Choe KW. Outcome of Staphylococcus aureus bacteremia in patients with eradicable foci versus noneradicable foci. Clin Infect Dis. 2003; 37:794–799.
Article21. Paul M, Kariv G, Goldberg E, Raskin M, Shaked H, Hazzan R, Samra Z, Paghis D, Bishara J, Leibovici L. Importance of appropriate empirical antibiotic therapy for methicillin-resistant Staphylococcus aureus bacteraemia. J Antimicrob Chemother. 2010; 65:2658–2665.
Article22. Patel M, Kumar RA, Stamm AM, Hoesley CJ, Moser SA, Waites KB. USA300 genotype community-associated methicillin-resistant Staphylococcus aureus as a cause of surgical site infections. J Clin Microbiol. 2007; 45:3431–3433.
Article23. Alvarez CA, Yomayusa N, Leal AL, Moreno J, Mendez-Alvarez S, Ibañez M, Vanegas N. Nosocomial infections caused by community-associated methicillin-resistant Staphylococcus aureus in Colombia. Am J Infect Control. 2010; 38:315–318.
Article24. Mangram AJ, Horan TC, Pearson ML, Silver LC, Jarvis WR. Guideline for prevention of surgical site infection, 1999. Hospital Infection Control Practices Advisory Committee. Infect Control Hosp Epidemiol. 1999; 20:250–278.25. Hermos CR, Sandora TJ, Williams LE, Mosammaparast N, McAdam AJ. Changing epidemiology of methicillin-resistant Staphylococcus aureus colonization in paediatric intensive-care units. Epidemiol Infect. 2013; 141:1983–1992.
Article26. Otto M. MRSA virulence and spread. Cell Microbiol. 2012; 14:1513–1521.
Article27. Chen Y, Chatterjee SS, Porcella SF, Yu YS, Otto M. Complete genome sequence of a Panton-Valentine leukocidin-negative community-associated methicillin-resistant Staphylococcus aureus strain of sequence type 72 from Korea. PLoS One. 2013; 8:e72803.28. Joo EJ, Choi JY, Chung DR, Song JH, Ko KS. Characteristics of the community-genotype sequence type 72 methicillin-resistant Staphylococcus aureus isolates that underlie their persistence in hospitals. J Microbiol. 2016; 54:445–450.
Article29. Cha HY, Moon DC, Choi CH, Oh JY, Jeong YS, Lee YC, Seol SY, Cho DT, Chang HH, Kim SW, Lee JC. Prevalence of the ST239 clone of methicillin-resistant Staphylococcus aureus and differences in antimicrobial susceptibilities of ST239 and ST5 clones identified in a Korean hospital. J Clin Microbiol. 2005; 43:3610–3614.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Community-Associated Methicillin-Resistant Staphylococcus aureus in Nosocomial Infections
- Clinical Observation on Staphylococcus aureus Bacteremia of Community Hospital
- Community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA)
- Nasal Carriage of Methicillin-Resistant Staphylococcus aureus among Healthcare Workers and Community Students in 1997 and 2006
- A third case of USA300 community-associated methicillin-resistant Staphylococcus aureus infection in Korea