J Korean Med Sci.
2017 Jul;32(7):1111-1117. 10.3346/jkms.2017.32.7.1111.
Characterization of Oropharyngeal Carriage Isolates of Neisseria meningitidis in Healthy Korean Adolescents in 2015
- Affiliations
-
- 1Center for Vaccine Evaluation and Study, Medical Research Institute, Ewha Womans University School of Medicine, Seoul, Korea. kaykim@ewha.ac.kr
- 2Department of Microbiology, Catholic Kwandong University College of Medicine, Gangneung, Korea.
- 3Department of Pediatrics, Ewha Womans University School of Medicine, Seoul, Korea.
- KMID: 2379605
- DOI: http://doi.org/10.3346/jkms.2017.32.7.1111
Abstract
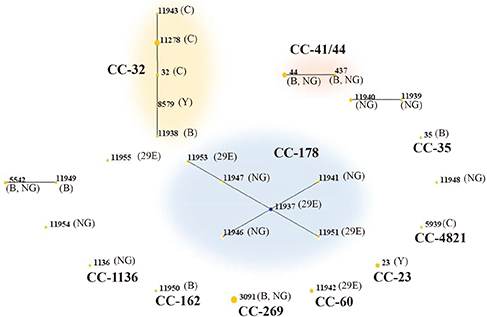
- The meningococcus carriage rate is age-dependent, with a high prevalence in adolescents and young adults. This cross-sectional study aimed to estimate the oropharyngeal carriage rate of meningococcus among healthy Korean adolescents and its relationship with several population characteristics. The survey was conducted from April to May 2015 among 1,460 first-year high-school students in 9 high schools located in Gyeonggi province, Korea. Each student answered a short questionnaire assessing risk factors for carriage, and posterior pharyngeal wall swab samples were obtained. These samples were cultured on meningococcus-selective media, with colonies resembling meningococci identified using the Vitek® MS system (bioMérieux, Marcy l'Etoile, France). All isolates were characterized by molecular serogrouping and multilocus sequence typing (MLST). Meningococci were identified from 3.4% (49/1,460) swabs. Current smokers had significantly higher carriage rates than non-smokers (8.2% vs. 2.9%, P = 0.002), and boys had significantly higher carriage rates than girls (4.4% vs. 1.6%, P = 0.004). Serogroup B was the most common serogroup, followed by serogroup C, then 29E and Y. Twenty-seven different sequence types (STs) were identified; the most common were ST-3091, ST-11278, and ST-44. These belonged to clonal complexes (CCs) 269, 32, and 41/44, respectively, known as the hypervirulent clones. Evaluating meningococcal carriage is important to understand the epidemiology of meningococcal disease; however, little data exist in Korea. Similar to western countries, meningococcal serogroup B has emerged in Korea, and hypervirulent clones were identified. It is necessary to monitor the genetic and serologic characteristics of circulating meningococci and to assess the potential strain coverage of meningococcal vaccines.
MeSH Terms
Figure
Reference
-
1. Howitz M, Lambertsen L, Simonsen JB, Christensen JJ, Mølbak K. Morbidity, mortality and spatial distribution of meningococcal disease, 1974–2007. Epidemiol Infect. 2009; 137:1631–1640.2. Gasparini R, Amicizia D, Lai PL, Panatto D. Neisseria meningitidis, pathogenetic mechanisms to overcome the human immune defences. J Prev Med Hyg. 2012; 53:50–55.3. Caugant DA. Genetics and evolution of Neisseria meningitidis: importance for the epidemiology of meningococcal disease. Infect Genet Evol. 2008; 8:558–565.4. Caugant DA, Maiden MC. Meningococcal carriage and disease--population biology and evolution. Vaccine. 2009; 27:Suppl 2. B64–B70.5. MacLennan J, Kafatos G, Neal K, Andrews N, Cameron JC, Roberts R, Evans MR, Cann K, Baxter DN, Maiden MC, et al. Social behavior and meningococcal carriage in British teenagers. Emerg Infect Dis. 2006; 12:950–957.6. Christensen H, May M, Bowen L, Hickman M, Trotter CL. Meningococcal carriage by age: a systematic review and meta-analysis. Lancet Infect Dis. 2010; 10:853–861.7. Neal KR, Nguyen-Van-Tam JS, Jeffrey N, Slack RC, Madeley RJ, Ait-Tahar K, Job K, Wale MC, Ala’Aldeen DA. Changing carriage rate of Neisseria meningitidis among university students during the first week of term: cross sectional study. BMJ. 2000; 320:846–849.8. Riordan T, Cartwright K, Andrews N, Stuart J, Burris A, Fox A, Borrow R, Douglas-Riley T, Gabb J, Miller A. Acquisition and carriage of meningococci in marine commando recruits. Epidemiol Infect. 1998; 121:495–505.9. Baker M, McNicholas A, Garrett N, Jones N, Stewart J, Koberstein V, Lennon D. Household crowding a major risk factor for epidemic meningococcal disease in Auckland children. Pediatr Infect Dis J. 2000; 19:983–990.10. Stanwell-Smith RE, Stuart JM, Hughes AO, Robinson P, Griffin MB, Cartwright K. Smoking, the environment and meningococcal disease: a case control study. Epidemiol Infect. 1994; 112:315–328.11. Lee SO, Ryu SH, Park SJ, Ryu J, Woo JH, Kim YS. Meningococcal disease in the republic of Korea army: incidence and serogroups determined by PCR. J Korean Med Sci. 2003; 18:163–166.12. Kim JH, Jun YH, Jeong YS. Study on pharyngeal Neisseria meningitidis carrier rate of Korean army recruits. J Korean Mil Med Assoc. 1990; 21:35–47.13. Hwang IU, Lee HK, Seo MY, Kim JP, Seo YB, Bang YJ. The changes of meningococcal carriage rate and the serogroup in Korean army recruits. J Korean Mil Med Assoc. 2010; 41:188–199.14. Park HD, Lee DH, Seo PW, Kim SH, Choi TY, Kim JH. Carrier rate, serogroup and vaccination effect of Neisseria meningitidis on army trainees in Korea. Chungbuk Med J. 1995; 5:45–57.15. Park HS, Chun YI. Vaccination effect on pharyngeal carrier rate of Neisseria meningitidis and its serogroups in Korean army recruits. J Korean Mil Med Assoc. 1992; 23:105–115.16. Heo JY, Bae SM, Cheong HJ, Kim WJ, Kim MY, Na W, Choe KW, Chun BC. Imapact of quadrivalent meningococcal conjugate vaccine on carried meningococci in Korean military trainee. J Korean Mil Med Assoc. 2014; 45:33–42.17. Choi S, Kim ES, Moon JS, Lee JS, Chung MH, Kim SM, Seo YH, Cho YK, Lee SO. Clinical and epidemiologic features of meningococcal infections in Incheon, Korea. Infect Chemother. 2005; 37:119–126.18. Durey A, Bae SM, Lee HJ, Nah SY, Kim M, Baek JH, Kang YH, Chung MH, Lee JS. Carriage rates and serogroups of Neisseria meningitidis among freshmen in a university dormitory in Korea. Yonsei Med J. 2012; 53:742–747.19. Kim NH, Lee J, Lee JW, Lee SY, Choi EH, Kim KH, Kang JH, Kim EC, Lee HJ. Carriage rates and serogroups of Neisseria meningitides in children attending day care centers. Korean J Pediatr Infect Dis. 2009; 16:31–39.20. Taha MK. Simultaneous approach for nonculture PCR-based identification and serogroup prediction of Neisseria meningitidis. J Clin Microbiol. 2000; 38:855–857.21. Bennett DE, Mulhall RM, Cafferkey MT. PCR-based assay for detection of Neisseria meningitidis capsular serogroups 29E, X, and Z. J Clin Microbiol. 2004; 42:1764–1765.22. Birtles A, Hardy K, Gray SJ, Handford S, Kaczmarski EB, Edwards-Jones V, Fox AJ. Multilocus sequence typing of Neisseria meningitidis directly from clinical samples and application of the method to the investigation of meningococcal disease case clusters. J Clin Microbiol. 2005; 43:6007–6014.23. Claus H, Maiden MC, Wilson DJ, McCarthy ND, Jolley KA, Urwin R, Hessler F, Frosch M, Vogel U. Genetic analysis of meningococci carried by children and young adults. J Infect Dis. 2005; 191:1263–1271.24. Gasparini R, Comanducci M, Amicizia D, Ansaldi F, Canepa P, Orsi A, Icardi G, Rizzitelli E, De Angelis G, Bambini S, et al. Molecular and serological diversity of Neisseria meningitidis carrier strains isolated from Italian students aged 14 to 22 years. J Clin Microbiol. 2014; 52:1901–1910.25. Yazdankhah SP, Kriz P, Tzanakaki G, Kremastinou J, Kalmusova J, Musilek M, Alvestad T, Jolley KA, Wilson DJ, McCarthy ND, et al. Distribution of serogroups and genotypes among disease-associated and carried isolates of Neisseria meningitidis from the Czech Republic, Greece, and Norway. J Clin Microbiol. 2004; 42:5146–5153.26. Lee SO. Commencement of the meningococcal vaccination for the Republic of Korea army. Infect Chemother. 2013; 45:113–115.27. Cunningham R, Matthews R, Lewendon G, Harrison S, Stuart JM. Improved rate of isolation of Neisseria meningitidis by direct plating of pharyngeal swabs. J Clin Microbiol. 2001; 39:4575–4576.28. Caugant DA, Tzanakaki G, Kriz P. Lessons from meningococcal carriage studies. FEMS Microbiol Rev. 2007; 31:52–63.29. Cartwright K. Epidemiology, surveillance, and population biology: carriage studies. Methods Mol Med. 2001; 67:293–311.30. Caugant DA, Høiby EA, Rosenqvist E, Frøholm LO, Selander RK. Transmission of Neisseria meningitidis among asymptomatic military recruits and antibody analysis. Epidemiol Infect. 1992; 109:241–253.31. Granoff D, Pelton S, Harrison L. Meningococcal vaccines. In : Plotkin S, Orenstein W, Offit PA, editors. Vaccines. 6th ed. Philadelphia, PA: Elsevier;2013. p. 388–418.32. Caugant DA. Population genetics and molecular epidemiology of Neisseria meningitidis. APMIS. 1998; 106:505–525.33. Kristiansen PA, Diomandé F, Ba AK, Sanou I, Ouédraogo AS, Ouédraogo R, Sangaré L, Kandolo D, Aké F, Saga IM, et al. Impact of the serogroup A meningococcal conjugate vaccine, MenAfriVac, on carriage and herd immunity. Clin Infect Dis. 2013; 56:354–363.34. Ala’Aldeen DA, Neal KR, Ait-Tahar K, Nguyen-Van-Tam JS, English A, Falla TJ, Hawkey PM, Slack RC. Dynamics of meningococcal long-term carriage among university students and their implications for mass vaccination. J Clin Microbiol. 2000; 38:2311–2316.35. Harrison LH, Shutt KA, Arnold KE, Stern EJ, Pondo T, Kiehlbauch JA, Myers RA, Hollick RA, Schmink S, Vello M, et al. Meningococcal carriage among Georgia and Maryland high school students. J Infect Dis. 2015; 211:1761–1768.36. Brehony C, Hill DM, Lucidarme J, Borrow R, Maiden MC. Meningococcal vaccine antigen diversity in global databases. Euro Surveill. 2015; 20:30084.37. Maiden MC, Bygraves JA, Feil E, Morelli G, Russell JE, Urwin R, Zhang Q, Zhou J, Zurth K, Caugant DA, et al. Multilocus sequence typing: a portable approach to the identification of clones within populations of pathogenic microorganisms. Proc Natl Acad Sci USA. 1998; 95:3140–3145.38. Bertrand S, Van Meervenne E, De Baere T, Vanhoof R, Collard JM, Ruckly C, Taha M, Carion F. Detection of a geographical and endemic cluster of hyper-invasive meningococcal strains. Microbes Infect. 2011; 13:684–690.39. Law DK, Lorange M, Ringuette L, Dion R, Giguère M, Henderson AM, Stoltz J, Zollinger WD, De Wals P, Tsang RS. Invasive meningococcal disease in Quebec, Canada, due to an emerging clone of ST-269 serogroup B meningococci with serotype antigen 17 and serosubtype antigen P1.19 (B:17:P1.19). J Clin Microbiol. 2006; 44:2743–2749.40. Sauer S, Kliem M. Mass spectrometry tools for the classification and identification of bacteria. Nat Rev Microbiol. 2010; 8:74–82.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neisseria meningitidis Infections
- Carriage Rates and Serogroups of Neisseria meningitidis among Freshmen in a University Dormitory in Korea
- Neonatal Sepsis and Meningitis Caused by Neisseria meningitidis Serogroup B: a Case Report
- Detection of Neisseria meningitidis, Haemophilus influenzae, Streptococcus pneumoniae and Streptococcus agalactiae using 16S rRNA gene
- Meningococcal Disease in the Republic of Korea Army: Incidence and Serogroups Determined by PCR