Ann Dermatol.
2017 Jun;29(3):263-267. 10.5021/ad.2017.29.3.263.
Low-Dose Systemic Methotrexate Therapy for Recalcitrant Alopecia Areata
- Affiliations
-
- 1Department of Dermatology, Chungnam National University School of Medicine, Daejeon, Korea. resina20@cnu.ac.kr
- 2Clinical Trials Center, Chungnam National University Hospital, Daejeon, Korea.
- KMID: 2378523
- DOI: http://doi.org/10.5021/ad.2017.29.3.263
Abstract
- BACKGROUND
Alopecia areata (AA) is an autoimmune skin disease difficult to manage and treat. The pathogenesis of AA features a T-cell-associated autoimmune process, and systemic immunosuppressive therapy is prescribed widely for AA.
OBJECTIVE
To evaluate the efficacy and tolerance of systemic low-dose methotrexate (LD-MTX) therapy in treatment of recalcitrant AA multiplex.
METHODS
In a retrospective, non-controlled study, we evaluated 29 patients with recalcitrant AA treated with LD-MTX and assessed the therapeutic response according to severity of disease, disease duration, cumulative dose of MTX, and drug safety.
RESULTS
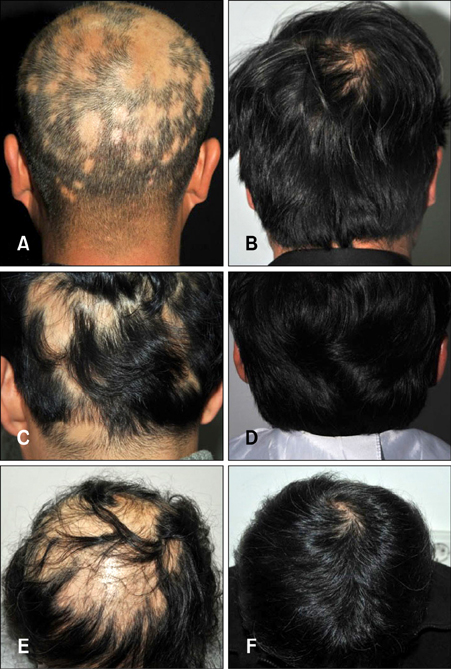
MTX was administered twice weekly, and the mean maximum weekly dose was 14.48 mg. The response was A5 (regrowth=100.0%) in 14 (48.3%) patients and A4 (regrowth of 75%~90%) in 12 (41.4%) patients. Three patients had poor response to LD-MTX treatment (A2: n=2 [6.9%], A1: n=1 [3.4%]). All three of the patients showing a poor response had disease durations exceeding 24 months. Relapse was observed in 31% of patients with more than 75% regrowth. Common side-effects were elevated liver enzyme levels and gastrointestinal discomfort.
CONCLUSION
LD-MTX appears to be an effective and well-tolerated treatment for recalcitrant AA multiplex.
Keyword
MeSH Terms
Figure
Reference
-
1. Safavi K. Prevalence of alopecia areata in the first national health and nutrition examination survey. Arch Dermatol. 1992; 128:702.
Article2. Chang KH, Rojhirunsakool S, Goldberg LJ. Treatment of severe alopecia areata with intralesional steroid injections. J Drugs Dermatol. 2009; 8:909–912.3. Kar BR, Handa S, Dogra S, Kumar B. Placebo-controlled oral pulse prednisolone therapy in alopecia areata. J Am Acad Dermatol. 2005; 52:287–290.
Article4. Luggen P, Hunziker T. High-dose intravenous corticosteroid pulse therapy in alopecia areata: own experience compared with the literature. J Dtsch Dermatol Ges. 2008; 6:375–378.
Article5. Goddard CJ, August PJ, Whorwell PJ. Alopecia totalis in a patient with Crohn's disease and its treatment with azathioprine. Postgrad Med J. 1989; 65:188–189.
Article6. Farshi S, Mansouri P, Safar F, Khiabanloo SR. Could azathioprine be considered as a therapeutic alternative in the treatment of alopecia areata? A pilot study. Int J Dermatol. 2010; 49:1188–1193.
Article7. Açıkgöz G, Calıkan E, Tunca M, Yeniay Y, Akar A. The effect of oral cyclosporine in the treatment of severe alopecia areata. Cutan Ocul Toxicol. 2014; 33:247–252.
Article8. Cipriani P, Ruscitti P, Carubbi F, Liakouli V, Giacomelli R. Methotrexate: an old new drug in autoimmune disease. Expert Rev Clin Immunol. 2014; 10:1519–1530.
Article9. Haustein UF, Rytter M. Methotrexate in psoriasis: 26 years' experience with low-dose long-term treatment. J Eur Acad Dermatol Venereol. 2000; 14:382–388.
Article10. Spring P, Gschwind I, Gilliet M. Prurigo nodularis: retrospective study of 13 cases managed with methotrexate. Clin Exp Dermatol. 2014; 39:468–473.
Article11. Olsen EA, Hordinsky MK, Price VH, Roberts JL, Shapiro J, Canfield D, et al. Alopecia areata investigational assessment guidelines--part II. national alopecia areata foundation. J Am Acad Dermatol. 2004; 51:440–447.12. Royer M, Bodemer C, Vabres P, Pajot C, Barbarot S, Paul C, et al. Efficacy and tolerability of methotrexate in severe childhood alopecia areata. Br J Dermatol. 2011; 165:407–410.
Article13. Chartaux E, Joly P. Long-term follow-up of the efficacy of methotrexate alone or in combination with low doses of oral corticosteroids in the treatment of alopecia areata totalis or universalis. Ann Dermatol Venereol. 2010; 137:507–513.14. Droitcourt C, Milpied B, Ezzedine K, Hubiche T, Belin E, Akpadjan F, et al. Interest of high-dose pulse corticosteroid therapy combined with methotrexate for severe alopecia areata: a retrospective case series. Dermatology. 2012; 224:369–373.
Article15. Kurosawa M, Nakagawa S, Mizuashi M, Sasaki Y, Kawamura M, Saito M, et al. A comparison of the efficacy, relapse rate and side effects among three modalities of systemic corticosteroid therapy for alopecia areata. Dermatology. 2006; 212:361–365.
Article16. Joly P. The use of methotrexate alone or in combination with low doses of oral corticosteroids in the treatment of alopecia totalis or universalis. J Am Acad Dermatol. 2006; 55:632–636.
Article17. Yeo IK, Ko EJ, No YA, Lim ES, Park KY, Li K, et al. Comparison of high-dose corticosteroid pulse therapy and combination therapy using oral cyclosporine with low-dose corticosteroid in severe alopecia areata. Ann Dermatol. 2015; 27:676–681.
Article18. Friedland R, Tal R, Lapidoth M, Zvulunov A, Ben Amitai D. Pulse corticosteroid therapy for alopecia areata in children: a retrospective study. Dermatology. 2013; 227:37–44.
Article19. Hammerschmidt M, Mulinari Brenner F. Efficacy and safety of methotrexate in alopecia areata. An Bras Dermatol. 2014; 89:729–734.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Congenitally Developed Alopecia Areata
- Efficacy of Oral Tofacitinib in Patients with Alopecia Areata: Tofacitinib Was Ineffective for Recalcitrant Alopecia Totalis of a Long Duration
- A Case of Alopecia Areata in a Patient taking Cyclosporine
- The Effect of Systemic Steroid Therapy on Bone Mineral Density with Alopecia Areata
- Superficial Cryotherapy of Alopecia Areata in Eyebrows