J Korean Med Sci.
2017 Jun;32(6):1016-1023. 10.3346/jkms.2017.32.6.1016.
A Randomized, Placebo-Controlled, Double-Blind, Multi-Center Therapeutic Confirmatory Study to Evaluate the Safety and Efficacy of Avanafil in Korean Patients with Erectile Dysfunction
- Affiliations
-
- 1Department of Urology and Medical Research Institute of Pusan National University Hospital, Pusan National University School of Medicine, Busan, Korea. joon501@pusan.ac.kr
- 2Department of Urology, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 3Department of Urology, College of Medicine, Korea University, Seoul, Korea.
- 4Department of Urology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 5Department of Urology, Seoul National University College of Medicine, Seoul National University Hospital, Seoul, Korea.
- 6Department of Urology, Ulsan University College of Medicine, Asan Medical Center, Seoul, Korea.
- 7Department of Urology, Chonnam National University Medical School, Gwangju, Korea.
- 8Department of Urology, Chonbuk National University Medical School, Jeonju, Korea.
- KMID: 2377733
- DOI: http://doi.org/10.3346/jkms.2017.32.6.1016
Abstract
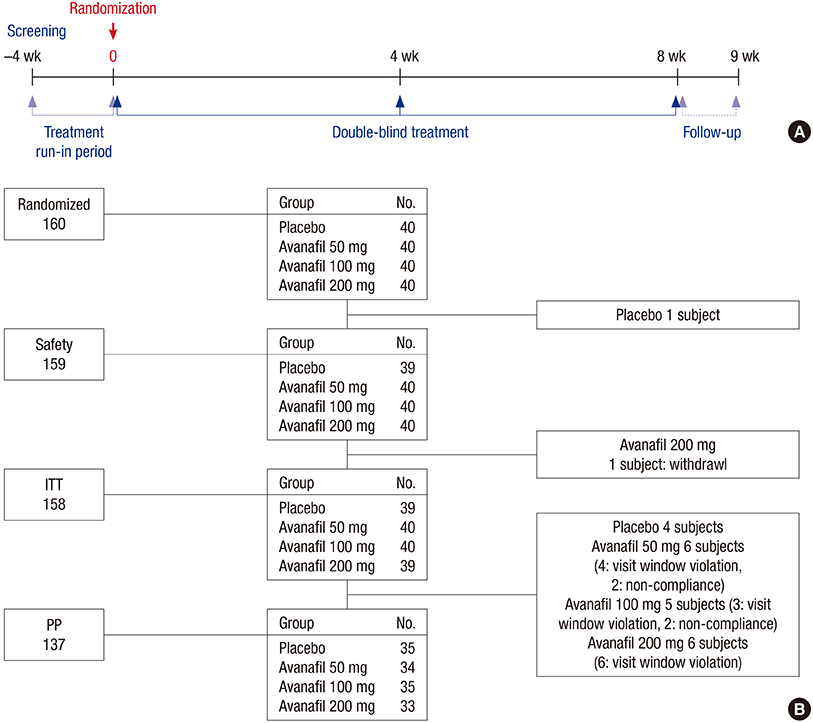
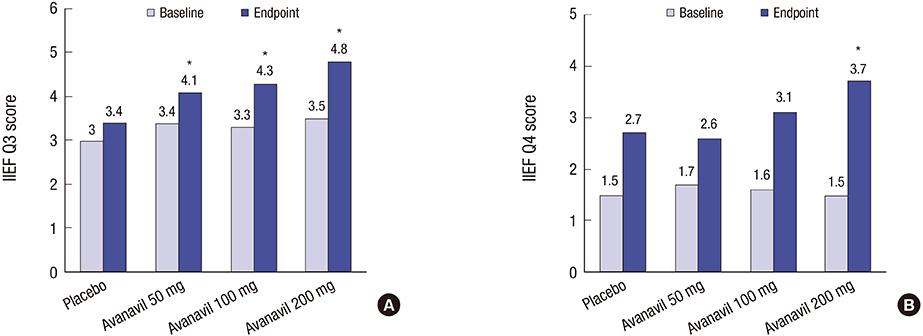
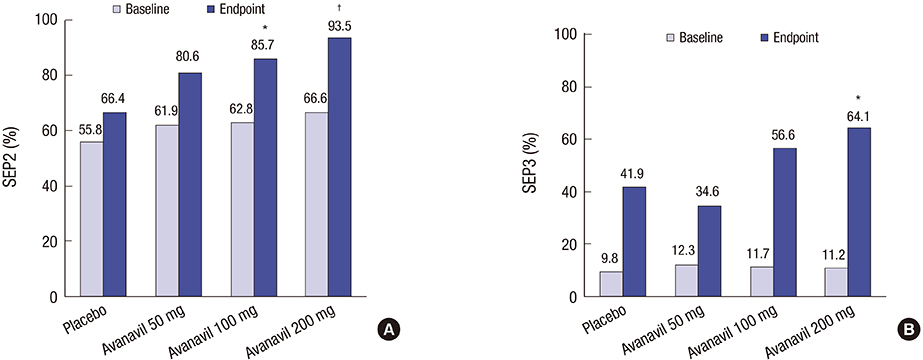
- A multi-center, randomized, double-blind, placebo-controlled study was conducted with 158 subjects who were randomized to placebo or avanafil 50, 100, and 200 mg on demand for 8 weeks to evaluate the safety, tolerability, and efficacy of avanafil in the treatment of erectile dysfunction (ED) in Korean men. The primary outcome was the erectile function (EF) domain score of the International Index of Erectile Function (IIEF) questionnaire. Secondary outcomes included changes in the scores of IIEF questions 3 and 4 (IIEF Q3, Q4) from baseline, changes in all domain scores in the IIEF from baseline, Sexual Encounter Profile questions 2-5 (SEP2-5), the Global Efficacy Assessment Question (GEAQ), and the number of subjects whose EF domain score at the 8th week visit was ≥ 26. After 8 weeks of treatment, the dose groups except avanafil 50 mg scored significantly higher on the IIEF-EF domain from baseline than the placebo group. The changes from baseline in the avanafil group in IIEF Q3 (all doses) and Q4 (200 mg alone) were higher than the placebo group. The differences between avanafil and placebo groups were significant in SEP2 (100 and 200 mg) and SEP3-5 (200 mg). The differences in the GEAQ "Yes" response were also significant in the avanafil 100 and 200 mg groups. Regarding the ratio of normal EF at the end of the study, avanafil 200 mg differed significantly from the placebo. Most treatment-associated adverse events were mild and resolved spontaneously. This is a clinical trial study and was registered at www.ClinicalTrials.gov (Identifier: NCT02477436).
MeSH Terms
Figure
Reference
-
1. Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, Wicker PA. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N Engl J Med. 1998; 338:1397–1404.2. Park JH, Cho IC, Kim YS, Kim SK, Min SK, Kye SS. Body mass index, waist-to-hip ratio, and metabolic syndrome as predictors of middle-aged men's health. Korean J Urol. 2015; 56:386–392.3. Canat L, Canat M, Guner B, Gurbuz C, Caşkurlu T. Association between renal function, erectile function and coronary artery disease: detection with coronary angiography. Korean J Urol. 2015; 56:76–81.4. Ko K, Yang DY, Lee WK, Kim SW, Moon DG, Moon KH, Park NC, Park JK, Son HC, Lee SW, et al. Effect of improvement in lower urinary tract symptoms on sexual function in men: tamsulosin monotherapy vs. combination therapy of tamsulosin and solifenacin. Korean J Urol. 2014; 55:608–614.5. DeLay KJ, Haney N, Hellstrom WJ. Modifying risk factors in the management of erectile dysfunction: a review. World J Mens Health. 2016; 34:89–100.6. Ryu JK, Cho KS, Kim SJ, Oh KJ, Kam SC, Seo KK, Shin HS, Kim SW. Korean Society for Sexual Medicine and Andrology (KSSMA) guideline on erectile dysfunction. World J Mens Health. 2013; 31:83–102.7. Hatzimouratidis K, Amar E, Eardley I, Giuliano F, Hatzichristou D, Montorsi F, Vardi Y, Wespes E; European Association of Urology. Guidelines on male sexual dysfunction: erectile dysfunction and premature ejaculation. Eur Urol. 2010; 57:804–814.8. Yuan J, Zhang R, Yang Z, Lee J, Liu Y, Tian J, Qin X, Ren Z, Ding H, Chen Q, et al. Comparative effectiveness and safety of oral phosphodiesterase type 5 inhibitors for erectile dysfunction: a systematic review and network meta-analysis. Eur Urol. 2013; 63:902–912.9. Gresser U, Gleiter CH. Erectile dysfunction: comparison of efficacy and side effects of the PDE-5 inhibitors sildenafil, vardenafil and tadalafil--review of the literature. Eur J Med Res. 2002; 7:435–446.10. Goldstein I, McCullough AR, Jones LA, Hellstrom WJ, Bowden CH, Didonato K, Trask B, Day WW. A randomized, double-blind, placebo-controlled evaluation of the safety and efficacy of avanafil in subjects with erectile dysfunction. J Sex Med. 2012; 9:1122–1133.11. Jung J, Choi S, Cho SH, Ghim JL, Hwang A, Kim U, Kim BS, Koguchi A, Miyoshi S, Okabe H, et al. Tolerability and pharmacokinetics of avanafil, a phosphodiesterase type 5 inhibitor: a single- and multiple-dose, double-blind, randomized, placebo-controlled, dose-escalation study in healthy Korean male volunteers. Clin Ther. 2010; 32:1178–1187.12. Mulhall JP, Burnett AL, Wang R, McVary KT, Moul JW, Bowden CH, DiDonato K, Shih W, Day WW. A phase 3, placebo controlled study of the safety and efficacy of avanafil for the treatment of erectile dysfunction after nerve sparing radical prostatectomy. J Urol. 2013; 189:2229–2236.13. Kedia GT, Uckert S, Assadi-Pour F, Kuczyk MA, Albrecht K. Avanafil for the treatment of erectile dysfunction: initial data and clinical key properties. Ther Adv Urol. 2013; 5:35–41.14. Rosen RC, Cappelleri JC, Gendrano N 3rd. The International Index of Erectile Function (IIEF): a state-of-the-science review. Int J Impot Res. 2002; 14:226–244.15. Hellstrom WJ, Freier MT, Serefoglu EC, Lewis RW, DiDonato K, Peterson CA. A phase II, single-blind, randomized, crossover evaluation of the safety and efficacy of avanafil using visual sexual stimulation in patients with mild to moderate erectile dysfunction. BJU Int. 2013; 111:137–147.16. Carvalheira AA, Pereira NM, Maroco J, Forjaz V. Dropout in the treatment of erectile dysfunction with PDE5: a study on predictors and a qualitative analysis of reasons for discontinuation. J Sex Med. 2012; 9:2361–2369.17. Ljunggren C, Hedelin H, Salomonsson K, Ströberg P. Giving patients with erectile dysfunction the opportunity to try all three available phosphodiesterase type 5 inhibitors contributes to better long-term treatment compliance. J Sex Med. 2008; 5:469–475.18. Leungwattanakij S, Flynn V Jr, Hellstrom WJ. Intracavernosal injection and intraurethral therapy for erectile dysfunction. Urol Clin North Am. 2001; 28:343–354.19. Vardi Y, Sprecher E, Gruenwald I. Logistic regression and survival analysis of 450 impotent patients treated with injection therapy: long-term dropout parameters. J Urol. 2000; 163:467–470.20. Sundaram CP, Thomas W, Pryor LE, Sidi AA, Billups K, Pryor JL. Long-term follow-up of patients receiving injection therapy for erectile dysfunction. Urology. 1997; 49:932–935.21. Park NC, Kim TN, Park HJ. Treatment strategy for non-responders to PDE5 inhibitors. World J Mens Health. 2013; 31:31–35.22. Wang H, Yuan J, Hu X, Tao K, Liu J, Hu D. The effectiveness and safety of avanafil for erectile dysfunction: a systematic review and meta-analysis. Curr Med Res Opin. 2014; 30:1565–1571.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Antioxidant Supplementation for Erectile Dysfunction: Systematic Review and Meta-Analysis of Double-Blind, Randomized, Placebo-Controlled Trials
- Efficacy and Safety of a Herbal Formula that Mainly Consists of Cornus Officinalis for Erectile Dysfunction: A Double-blind, Placebo-controlled Study
- Efficacy and Safety of Red Ginseng Extract Powder in Patients with Erectile Dysfunction: Multicenter, Randomized, Double-Blind, Placebo-Controlled Study
- Efficacy and Safety of Udenafil Once Daily in Patients with Erectile Dysfunction after Bilateral Nerve-Sparing Robot-Assisted Laparoscopic Radical Prostatectomy: A Randomized, Double-Blind, Placebo-Controlled Study
- Effects of cetirizine in dogs with chronic atopic dermatitis: a randomized, double blind, placebo-controlled trial