Clin Exp Otorhinolaryngol.
2017 Mar;10(1):50-55. 10.21053/ceo.2016.00430.
A Novel Frameshift Mutation of SLC26A4 in a Korean Family With Nonsyndromic Hearing Loss and Enlarged Vestibular Aqueduct
- Affiliations
-
- 1Department of Biology, College of Natural Sciences, Kyungpook National University, Daegu, Korea. kimuk@knu.ac.kr
- 2Department of Aroma Applied Industry, College of Herbal Bio-Industry, Daegu Haany University, Gyeongsan, Korea.
- 3Department of Otorhinolaryngology-Head and Neck Surgery, Kyungpook National University School of Medicine, Daegu, Korea. kylee@knu.ac.kr
- 4School of Life Sciences, BK21 Plus KNU Creative BioResearch Group, Kyungpook National University, Daegu, Korea.
- KMID: 2376408
- DOI: http://doi.org/10.21053/ceo.2016.00430
Abstract
OBJECTIVES
We aimed to identify the causative mutation for siblings in a Korean family with nonsyndromic hearing loss (HL) and enlarged vestibular aqueduct (EVA). The siblings were a 19-year-old female with bilateral profound HL and an 11-year-old male with bilateral moderately severe HL.
METHODS
We extracted genomic DNA from blood samples of the siblings with HL, their parents, and 100 controls. We performed mutation analysis for SLC26A4 using direct sequencing.
RESULTS
The two siblings were compound heterozygotes with the novel mutation p.I713LfsX8 and the previously described mutation p.H723R. Their parents had heterozygous mono-allelic mutations. Father had p.I713LfsX8 mutation as heterozygous, and mother had p.H723R mutation as heterozygous. However, novel mutation p.I713LfsX8 was not detected in 100 unrelated controls.
CONCLUSION
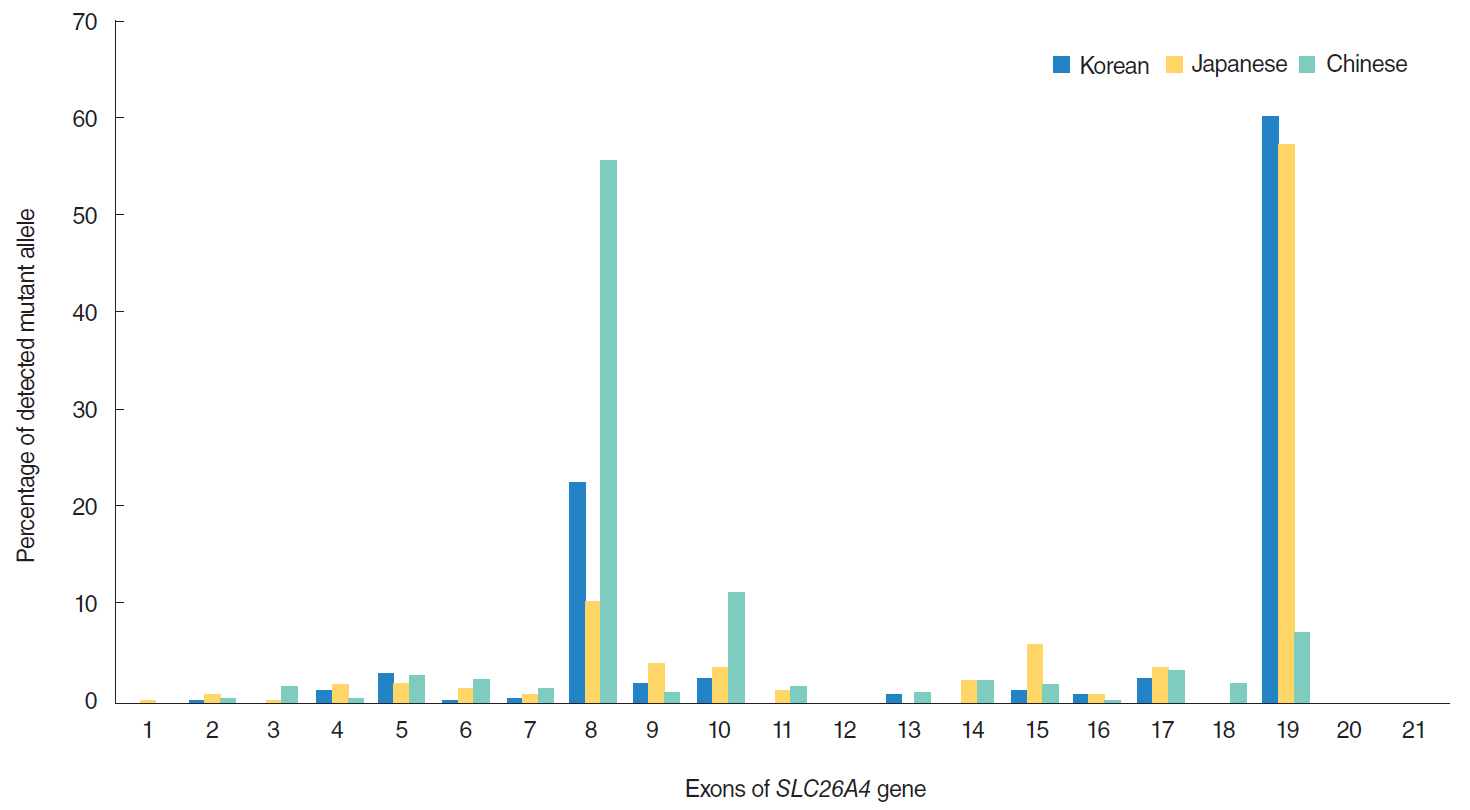
Both mutations identified in this study were located in the sulfate transporter and anti-sigma factor antagonist domain, the core region for membrane targeting of SulP/SLC26 anion transporters, which strongly suggests that failure in membrane trafficking by SLC26A4 is a direct cause of HL in this family. Our study could therefore provide a foundation for further investigations elucidating the SLC26A4-related mechanisms of HL.
MeSH Terms
Figure
Reference
-
1. Usami S, Abe S, Weston MD, Shinkawa H, Van Camp G, Kimberling WJ. Non-syndromic hearing loss associated with enlarged vestibular aqueduct is caused by PDS mutations. Hum Genet. 1999; Feb. 104(2):188–92.
Article2. Pique LM, Brennan ML, Davidson CJ, Schaefer F, Greinwald J Jr, Schrijver I. Mutation analysis of the SLC26A4, FOXI1 and KCNJ10 genes in individuals with congenital hearing loss. PeerJ. 2014; May. 2:e384.3. Madden C, Halsted M, Benton C, Greinwald J, Choo D. Enlarged vestibular aqueduct syndrome in the pediatric population. Otol Neurotol. 2003; Jul. 24(4):625–32.
Article4. Okamoto Y, Mutai H, Nakano A, Arimoto Y, Sugiuchi T, Masuda S, et al. Subgroups of enlarged vestibular aqueduct in relation to SLC26A4 mutations and hearing loss. Laryngoscope. 2014; Apr. 124(4):E134–40.5. Hilgert N, Smith RJ, Van Camp G. Forty-six genes causing nonsyndromic hearing impairment: which ones should be analyzed in DNA diagnostics? Mutat Res. 2009; Mar-Jun. 681(2-3):189–96.
Article6. Everett LA, Glaser B, Beck JC, Idol JR, Buchs A, Heyman M, et al. Pendred syndrome is caused by mutations in a putative sulphate transporter gene (PDS). Nat Genet. 1997; Dec. 17(4):411–22.
Article7. Kopp P, Pesce L, Solis-S JC. Pendred syndrome and iodide transport in the thyroid. Trends Endocrinol Metab. 2008; Sep. 19(7):260–8.8. Bizhanova A, Kopp P. Genetics and phenomics of Pendred syndrome. Mol Cell Endocrinol. 2010; Jun. 322(1-2):83–90.
Article9. Lee KY, Choi SY, Bae JW, Kim S, Chung KW, Drayna D, et al. Molecular analysis of the GJB2, GJB6 and SLC26A4 genes in Korean deafness patients. Int J Pediatr Otorhinolaryngol. 2008; Sep. 72(9):1301–9.10. Sagong B, Seo YJ, Lee HJ, Kim MJ, Kim UK, Moon IS. A mutation of the succinate dehydrogenase B gene in a Korean family with paraganglioma. Fam Cancer. 2016; Feb. 1. [Epub]. http://dx.doi.org/10.1007/s10689-016-9874-8.
Article11. Cremers CW, Admiraal RJ, Huygen PL, Bolder C, Everett LA, Joosten FB, et al. Progressive hearing loss, hypoplasia of the cochlea and widened vestibular aqueducts are very common features in Pendred’s syndrome. Int J Pediatr Otorhinolaryngol. 1998; Oct. 45(2):113–23.
Article12. Tsukamoto K, Suzuki H, Harada D, Namba A, Abe S, Usami S. Distribution and frequencies of PDS (SLC26A4) mutations in Pendred syndrome and nonsyndromic hearing loss associated with enlarged vestibular aqueduct: a unique spectrum of mutations in Japanese. Eur J Hum Genet. 2003; Dec. 11(12):916–22.13. Shin JW, Lee SC, Lee HK, Park HJ. Genetic Screening of GJB2 and SLC26A4 in Korean Cochlear Implantees: experience of Soree Ear Clinic. Clin Exp Otorhinolaryngol. 2012; Apr. 5 Suppl 1:S10–3.14. Lee HJ, Jung J, Shin JW, Song MH, Kim SH, Lee JH, et al. Correlation between genotype and phenotype in patients with bi-allelic SLC26A4 mutations. Clin Genet. 2014; Sep. 86(3):270–5.15. Song MH, Shin JW, Park HJ, Lee KA, Kim Y, Kim UK, et al. Intrafamilial phenotypic variability in families with biallelic SLC26A4 mutations. Laryngoscope. 2014; May. 124(5):E194–202.16. Park HJ, Hahn SH, Chun YM, Park K, Kim HN. Connexin26 mutations associated with nonsyndromic hearing loss. Laryngoscope. 2000; Sep. 110(9):1535–8.
Article17. Suzuki H, Oshima A, Tsukamoto K, Abe S, Kumakawa K, Nagai K, et al. Clinical characteristics and genotype-phenotype correlation of hearing loss patients with SLC26A4 mutations. Acta Otolaryngol. 2007; Dec. 127(12):1292–7.18. Miyagawa M, Nishio SY, Usami S; Deafness Gene Study Consortium. Mutation spectrum and genotype-phenotype correlation of hearing loss patients caused by SLC26A4 mutations in the Japanese: a large cohort study. J Hum Genet. 2014; May. 59(5):262–8.19. Chai Y, Huang Z, Tao Z, Li X, Li L, Li Y, et al. Molecular etiology of hearing impairment associated with nonsyndromic enlarged vestibular aqueduct in East China. Am J Med Genet A. 2013; Sep. 161A(9):2226–33.
Article20. Chen K, Zong L, Liu M, Wang X, Zhou W, Zhan Y, et al. Developing regional genetic counseling for southern Chinese with nonsyndromic hearing impairment: a unique mutational spectrum. J Transl Med. 2014; Mar. 12:64.
Article21. Hu H, Wu L, Feng Y, Pan Q, Long Z, Li J, et al. Molecular analysis of hearing loss associated with enlarged vestibular aqueduct in the mainland Chinese: a unique SLC26A4 mutation spectrum. J Hum Genet. 2007; 52(6):492–7.22. Wang QJ, Zhao YL, Rao SQ, Guo YF, Yuan H, Zong L, et al. A distinct spectrum of SLC26A4 mutations in patients with enlarged vestibular aqueduct in China. Clin Genet. 2007; Sep. 72(3):245–54.23. Yoon JS, Park HJ, Yoo SY, Namkung W, Jo MJ, Koo SK, et al. Heterogeneity in the processing defect of SLC26A4 mutants. J Med Genet. 2008; Jul. 45(7):411–9.24. Bizhanova A, Chew TL, Khuon S, Kopp P. Analysis of cellular localization and function of carboxy-terminal mutants of pendrin. Cell Physiol Biochem. 2011; 28(3):423–34.
Article25. Sharma AK, Rigby AC, Alper SL. STAS domain structure and function. Cell Physiol Biochem. 2011; 28(3):407–22.
Article26. Frischmeyer PA, Dietz HC. Nonsense-mediated mRNA decay in health and disease. Hum Mol Genet. 1999; 8(10):1893–900.27. King KA, Choi BY, Zalewski C, Madeo AC, Manichaikul A, Pryor SP, et al. SLC26A4 genotype, but not cochlear radiologic structure, is correlated with hearing loss in ears with an enlarged vestibular aqueduct. Laryngoscope. 2010; Feb. 120(2):384–9.28. Ito T, Choi BY, King KA, Zalewski CK, Muskett J, Chattaraj P, et al. SLC26A4 genotypes and phenotypes associated with enlargement of the vestibular aqueduct. Cell Physiol Biochem. 2011; 28(3):545–52.29. Pera A, Dossena S, Rodighiero S, Gandia M, Botta G, Meyer G, et al. Functional assessment of allelic variants in the SLC26A4 gene involved in Pendred syndrome and nonsyndromic EVA. Proc Natl Acad Sci U S A. 2008; Nov. 105(47):18608–13.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Family of H723R Mutation for SLC26A4 Associated with Enlarged Vestibular Aqueduct Syndrome
- Two Compound Heterozygous Were Identified in SLC26A4 Gene in Two Chinese Families With Enlarged Vestibular Aqueduct
- Carrier frequency of SLC26A4 mutations causing inherited deafness in the Korean population
- Familial Enlarged Vestibular Aqueduct Syndrome (FEVAS)
- Auditory Outcome of Cochlear Implantation in Adolescent and Adult Patients with Enlarged Vestibular Aqueduct and Biallelic SLC26A4 Mutations