Yonsei Med J.
2016 Mar;57(2):523-526. 10.3349/ymj.2016.57.2.523.
A Rare Case of Intra-Endometrial Leiomyoma of Uterus Simulating Degenerated Submucosal Leiomyoma Accompanied by a Large Sertoli-Leydig Cell Tumor
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Ewha Womans University School of Medicine, Seoul, Korea. sarahmd@ewha.ac.kr
- 2Department of Pathology, Ewha Womans University School of Medicine, Seoul, Korea.
- KMID: 2374063
- DOI: http://doi.org/10.3349/ymj.2016.57.2.523
Abstract
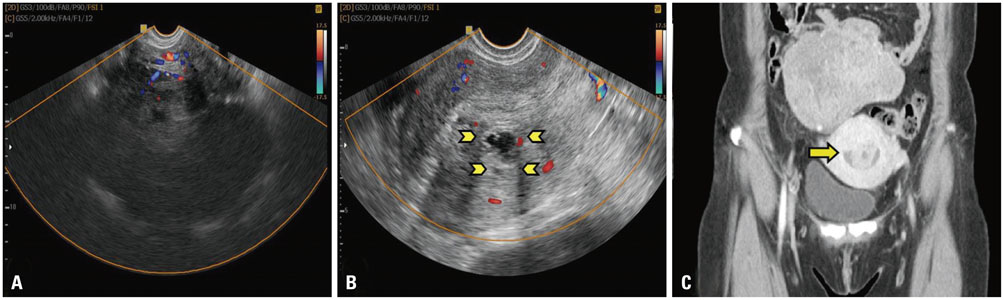
- A 50-year-old peri-menopausal woman presented with hard palpable mass on her lower abdomen and anemia from heavy menstrual bleeding. Ultrasonography showed a 13x12 cm sized hypoechoic solid mass in pelvis and a 2.5x2 cm hypoechoic cystic mass in uterine endometrium. Abdomino-pelvic computed tomography revealed a hypodense pelvic mass without enhancement, suggesting a leiomyoma of intraligamentary type or sex cord tumor of right ovary with submucosal myoma of uterus. Laparoscopy revealed a large Sertoli-Leydig cell tumor of right ovary with a very rare entity of intra-endometrial uterine leiomyoma accompanied by adenomyosis. The final diagnosis of ovarian sex-cord tumor (Sertoli-Leydig cell), stage Ia with intra-endometrial leiomyoma with adenomyosis, was made. Considering the large size of the tumor and poorly differentiated nature, 6 cycles of chemotherapy with Taxol and Carboplatin regimen were administered. There is neither evidence of major complications nor recurrence during 20 months' follow-up.
Keyword
MeSH Terms
-
Adenomyosis/*diagnosis/drug therapy
Carboplatin/therapeutic use
Female
Humans
Laparoscopy
Leiomyoma/*diagnosis/drug therapy
Male
Menorrhagia
Middle Aged
Neoplasm Recurrence, Local
Paclitaxel/therapeutic use
Sertoli-Leydig Cell Tumor/*diagnosis/drug therapy
Treatment Outcome
Uterine Neoplasms/*diagnosis/drug therapy
Carboplatin
Paclitaxel
Figure
Reference
-
1. Crum CP, Nucci MR, Lee KR. Diagnostic Gynecologic and Obstetric Pathology. 2nd ed. Philadelphia: Saunders;2011.2. Young RH, Scully RE. Ovarian Sertoli-Leydig cell tumors. A clinicopathological analysis of 207 cases. Am J Surg Pathol. 1985; 9:543–569.3. Zhang M, Cheung MK, Shin JY, Kapp DS, Husain A, Teng NN, et al. Prognostic factors responsible for survival in sex cord stromal tumors of the ovary--an analysis of 376 women. Gynecol Oncol. 2007; 104:396–400.
Article4. Bhat RA, Lim YK, Chia YN, Yam KL. Sertoli-Leydig cell tumor of the ovary: analysis of a single institution database. J Obstet Gynaecol Res. 2013; 39:305–310.
Article5. Roth LM, Anderson MC, Govan AD, Langley FA, Gowing NF, Woodcock AS. Sertoli-Leydig cell tumors: a clinicopathologic study of 34 cases. Cancer. 1981; 48:187–197.
Article6. Kurman RJ, Ellenson HL, Ronnett BM. Blaustein's Pathology of the Female Genital Tract. 6th ed. New York: Springer;2011.7. Gui T, Cao D, Shen K, Yang J, Zhang Y, Yu Q, et al. A clinicopathological analysis of 40 cases of ovarian Sertoli-Leydig cell tumors. Gynecol Oncol. 2012; 127:384–389.
Article8. Lantzsch T, Stoerer S, Lawrenz K, Buchmann J, Strauss HG, Koelbl H. Sertoli-Leydig cell tumor. Arch Gynecol Obstet. 2001; 264:206–208.
Article9. McGuire WP, Hoskins WJ, Brady MF, Kucera PR, Partridge EE, Look KY, et al. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med. 1996; 334:1–6.
Article10. Persechini ML, Motton S, Leguevaque P, Donadille F, Escourrou G, Vierasu B, et al. Virilising ovarian tumour: a case associating a Sertoli-Leydig cell tumour and a Brenner tumour. Gynecol Endocrinol. 2011; 27:345–350.
Article11. Brown J, Sood AK, Deavers MT, Milojevic L, Gershenson DM. Patterns of metastasis in sex cord-stromal tumors of the ovary: can routine staging lymphadenectomy be omitted? Gynecol Oncol. 2009; 113:86–90.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Sertoli-Leydig Cell Tumor of the Ovary
- A Case of Giant Sertoli-Leydig Cell Tumor of the Ovary with Masculinization
- A Case of Large Cystic Myxoid Leiomyoma in Uterine Round Ligament
- Ovarian serous cystadenoma associated with Sertoli-Leydig cell tumor: a case report
- Primary Squamous Cell Carcinoma of the Endometrium Covering Submucosal Leiomyoma