Yonsei Med J.
2016 Mar;57(2):407-418. 10.3349/ymj.2016.57.2.407.
Is Tamsulosin 0.2 mg Effective and Safe as a First-Line Treatment Compared with Other Alpha Blockers?: A Meta-Analysis and a Moderator Focused Study
- Affiliations
-
- 1Institute for Clinical Molecular Biology Research, Soonchunhyang University Hospital, Soonchunhyang University College of Medicine, Seoul, Korea.
- 2Department of Urology, Soonchunhyang University College of Medicine, Seoul, Korea. piacekjh@hanmail.net
- 3Department of Urology, Chung-Ang University Hospital, Urological Science Institute, Chung-Ang University College of Medicine, Seoul, Korea.
- 4Department of Education, College of Education, Jeonju University, Jeonju, Korea.
- 5Department of Social Welfare, Kyungpook National University College of Social Science, Daegu, Korea.
- 6Department of Urology, Gil Hospital, Gachon University College of Medicine, Incheon, Korea.
- KMID: 2374047
- DOI: http://doi.org/10.3349/ymj.2016.57.2.407
Abstract
- PURPOSE
Tamsulosin 0.2 mg is used widely in Asian people, but the low dose has been studied less than tamsulosin 0.4 mg or other alpha blockers of standard dose. This study investigated the efficacy and safety of tamsulosin 0.2 mg by a meta-analysis and meta-regression.
MATERIALS AND METHODS
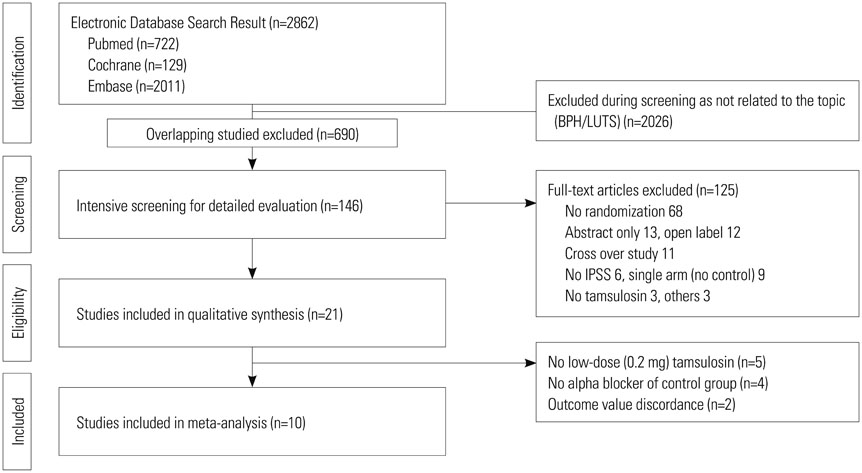
We conducted a meta-analysis of efficacy of tamsulosin 0.2 mg using International Prostate Symptom Score (IPSS), maximal urinary flow rate (Qmax), post-voided residual volume (PVR), and quality of life (QoL). Safety was analyzed using adverse events. Relevant studies were searched using MEDLINE, EMBASE, and Cochrane library from January 1980 to June 2013.
RESULTS
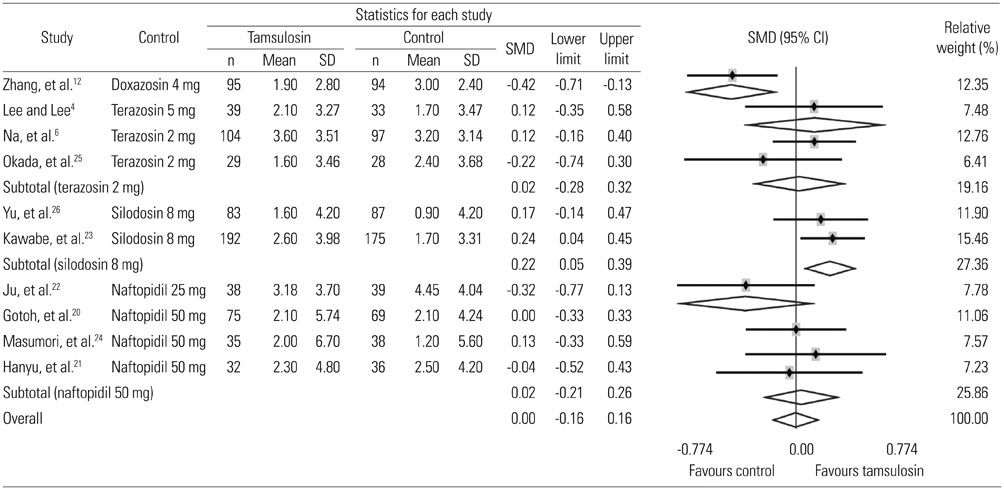
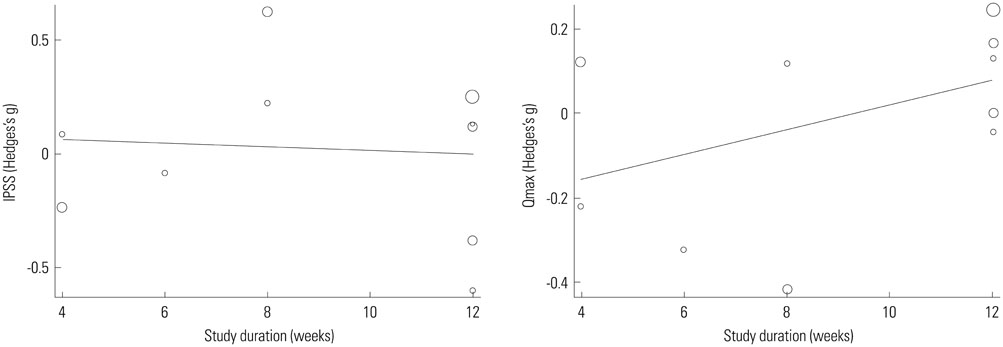
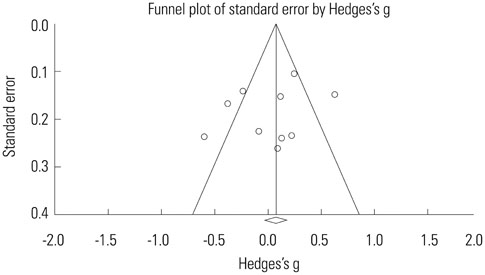
Ten studies were included with a total sample size of 1418 subjects [722 tamsulosin 0.2 mg group and 696 other alpha-blockers (terazosin, doxazosin, naftopidil, silodosin) group]. Study duration ranged from 4 to 24 weeks. The pooled overall standardized mean differences (SMD) in the mean change of IPSS from baseline for the tamsulosin group versus the control group was 0.02 [95% confidence interval (CI); -0.20, 0.25]. The pooled overall SMD in the mean change of QoL from baseline for the tamsulosin group versus the control group was 0.16 (95% CI; -0.16, 0.48). The regression analysis with the continuous variables (number of patients, study duration) revealed no significance in all outcomes as IPSS, QoL, and Qmax.
CONCLUSION
This study clarifies that tamsulosin 0.2 mg has similar efficacy and fewer adverse events compared with other alpha-blockers as an initial treatment strategy for men with lower urinary tract symptoms.
Keyword
MeSH Terms
-
Adrenergic alpha-1 Receptor Antagonists/*administration & dosage/therapeutic use
Adrenergic alpha-Antagonists
Dose-Response Relationship, Drug
Humans
Male
Middle Aged
Prostatic Hyperplasia/*complications
*Quality of Life
Sulfonamides/*administration & dosage/therapeutic use
Adrenergic alpha-1 Receptor Antagonists
Adrenergic alpha-Antagonists
Sulfonamides
Figure
Reference
-
1. Chapple CR. BHP Disease Management. Introduction and concluding remarks. Eur Urol. 1999; 36:Suppl 3. 1–6.2. Djavan B, Chapple C, Milani S, Marberger M. State of the art on the efficacy and tolerability of alpha1-adrenoceptor antagonists in patients with lower urinary tract symptoms suggestive of benign prostatic hyperplasia. Urology. 2004; 64:1081–1088.
Article3. Lee E. Comparison of tamsulosin and finasteride for lower urinary tract symptoms associated with benign prostatic hyperplasia in Korean patients. J Int Med Res. 2002; 30:584–590.
Article4. Lee E, Lee C. Clinical comparison of selective and non-selective alpha 1A-adrenoreceptor antagonists in benign prostatic hyperplasia: studies on tamsulosin in a fixed dose and terazosin in increasing doses. Br J Urol. 1997; 80:606–611.
Article5. Li NC, Chen S, Yang XH, Du LD, Wang JY, Na YQ. Beijing Tamsulosin Study Group. Efficacy of low-dose tamsulosin in chinese patients with symptomatic benign prostatic hyperplasia. Clin Drug Investig. 2003; 23:781–787.
Article6. Na YJ, Guo YL, Gu FL. The Chinese Tamsulosin Study Group. Clinical comparison of selective and non-selective alpha 1A-adrenoceptor antagonists for bladder outlet obstruction associated with benign prostatic hyperplasia: studies on tamsulosin and terazosin in Chinese patients. J Med. 1998; 29:289–304.7. Park CH, Chang HS, Oh BR, Kim HJ, Sul CK, Chung SK, et al. Efficacy of low-dose tamsulosin on lower urinary tract symptoms suggestive of benign prostatic hyperplasia: a nonblind multicentre Korean study. Clin Drug Investig. 2004; 24:41–47.
Article8. Giuliano F. Impact of medical treatments for benign prostatic hyperplasia on sexual function. BJU Int. 2006; 97:Suppl 2. 34–38.
Article9. Giuliano F. Lower urinary tract symptoms and sexual dysfunction: a common approach. BJU Int. 2008; 101:Suppl 3. 22–26.
Article10. Kim JH, Park JY, Oh MM, Lee JG, Kwon SS, Bae JH. Treatment satisfaction with low-dose tamsulosin for symptomatic benign prostatic hyperplasia: results from a multicentre cross-sectional survey. Int J Clin Pract. 2012; 66:1209–1215.
Article11. Lee HS, Kim SW, Oh SJ, Choo MS, Lee KS. Efficacy and safety of tamsulosin for treating lower urinary tract symptoms associated with benign prostatic hyperplasia: a multicenter, randomized, controlled, open-label non-inferiority study. Korean J Urol. 2012; 53:178–183.
Article12. Zhang K, Yu W, Jin J, Ye H, Wang X, Zhang N, et al. Effect of doxazosin gastrointestinal therapeutic system 4 mg vs tamsulosin 0.2 mg on nocturia in Chinese men with lower urinary tract symptoms: a prospective, multicenter, randomized, open, parallel study. Urology. 2011; 78:636–640.
Article13. Ren RM, Kou M, Lan XX. Efficacy and safety of tamsulosin for the treatment of benign prostatic hyperplasia: a meta analysis. Chin Med J (Engl). 2010; 123:234–238.14. Yuan J, Liu Y, Yang Z, Qin X, Yang K, Mao C. The efficacy and safety of alpha-1 blockers for benign prostatic hyperplasia: an overview of 15 systematic reviews. Curr Med Res Opin. 2013; 29:279–287.
Article15. Shim SR, Kim JH, Choi H, Lee WJ, Kim HJ, Bae MY, et al. General effect of low-dose tamsulosin (0.2 mg) as a first-line treatment for lower urinary tract symptoms associated with benign prostatic hyperplasia: a systematic review and meta-analysis. Curr Med Res Opin. 2015; 31:353–365.
Article16. Jonathan JD, Julian PT, Douglas GA. The Cochrane Collaboration: Cochrane Handbook for Systematic Reviews of Interventions, Version 5.0.0. Availabe at: http://www.cochrane-handbook.org.17. Moher D, Liberati A, Tetzlaff J, Altman DG. PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009; 6:e1000097.
Article18. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986; 7:177–188.
Article19. Cooper HM, Hedges LV, Valentine JC. The handbook of research synthesis and meta-analysis. 2nd ed. New York: Russell Sage Foundation;2009.20. Gotoh M, Kamihira O, Kinukawa T, Ono Y, Ohshima S, Origasa H. Tokai Urological Clinical Trial Group. Comparison of tamsulosin and naftopidil for efficacy and safety in the treatment of benign prostatic hyperplasia: a randomized controlled trial. BJU Int. 2005; 96:581–586.
Article21. Hanyu S, Hatano A, Nishiyama T, Obara K, Takahashi K. [A randomized controlled study comparing clinical effects of naftopidil and tamsulosin on benign prostatic hyperplasia]. Hinyokika Kiyo. 2010; 56:489–494.22. Ju XB, Wu HF, Su JT. [The clinical efficacy of Naftopidil tablet in the treatment of benign prostatic hyperplasia]. Zhonghua Nan Ke Xue. 2002; 8:286–288.23. Kawabe K, Yoshida M, Homma Y. Silodosin Clinical Study Group. Silodosin, a new alpha1A-adrenoceptor-selective antagonist for treating benign prostatic hyperplasia: results of a phase III randomized, placebo-controlled, double-blind study in Japanese men. BJU Int. 2006; 98:1019–1024.
Article24. Masumori N, Tsukamoto T, Iwasawa A, Furuya R, Sonoda T, Mori M. Hokkaido Urological Disorders Conference Writing Group. Ejaculatory disorders caused by alpha-1 blockers for patients with lower urinary tract symptoms suggestive of benign prostatic hyperplasia: comparison of naftopidil and tamsulosin in a randomized multicenter study. Urol Int. 2009; 83:49–54.
Article25. Okada H, Kamidono S, Yoshioka T, Okuyama A, Ozono S, Hirao Y, et al. A comparative study of terazosin and tamsulosin for symptomatic benign prostatic hyperplasia in Japanese patients. BJU Int. 2000; 85:676–681.
Article26. Yu HJ, Lin AT, Yang SS, Tsui KH, Wu HC, Cheng CL, et al. Non-inferiority of silodosin to tamsulosin in treating patients with lower urinary tract symptoms (LUTS) associated with benign prostatic hyperplasia (BPH). BJU Int. 2011; 108:1843–1848.
Article27. Kaplan SA, Goluboff ET, Olsson CA, Deverka PA, Chmiel JJ. Effect of demographic factors, urinary peak flow rates, and Boyarsky symptom scores on patient treatment choice in benign prostatic hyperplasia. Urology. 1995; 45:398–405.
Article28. Lepor H. Alpha blockade for the treatment of benign prostatic hyperplasia. Urol Clin North Am. 1995; 22:375–386.
Article29. Djavan B, Marberger M. A meta-analysis on the efficacy and tolerability of alpha1-adrenoceptor antagonists in patients with lower urinary tract symptoms suggestive of benign prostatic obstruction. Eur Urol. 1999; 36:1–13.
Article30. Lowe FC. Coadministration of tamsulosin and three antihypertensive agents in patients with benign prostatic hyperplasia: pharmacodynamic effect. Clin Ther. 1997; 19:730–742.
Article31. Schulman CC, Cortvriend J, Jonas U, Lock TM, Vaage S, Speakman MJ. European Tamsulosin Study Group. Tamsulosin, the first prostate-selective alpha 1A-adrenoceptor antagonist. Analysis of a multinational, multicentre, openlabel study assessing the long-term efficacy and safety in patients with benign prostatic obstruction (symptomatic BPH). Eur Urol. 1996; 29:145–154.32. Abrams P, Schulman CC, Vaage S. The European Tamsulosin Study Group. Tamsulosin, a selective alpha 1c-adrenoceptor antagonist: a randomized, controlled trial in patients with benign prostatic 'obstruction' (symptomatic BPH). Br J Urol. 1995; 76:325–336.
Article33. Chapple CR, Wyndaele JJ, Nordling J, Boeminghaus F, Ypma AF, Abrams P. European Tamsulosin Study Group. Tamsulosin, the first prostate-selective alpha 1A-adrenoceptorantagonist A meta-analysis of two randomized, placebo-controlled, multicentre studies in patients with benign prostatic obstruction (symptomatic BPH). Eur Urol. 1996; 29:155–167.34. Lepor H. Tamsulosin Investigator Group. Long-term evaluation of tamsulosin in benign prostatic hyperplasia: placebo-controlled, double-blind extension of phase III trial. Urology. 1998; 51:901–906.
Article35. Lepor H. Tamsulosin Investigator Group. Phase III multicenter placebo-controlled study of tamsulosin in benign prostatic hyperplasia. Urology. 1998; 51:892–900.
Article36. Narayan P, Evans CP, Moon T. Long-term safety and efficacy of tamsulosin for the treatment of lower urinary tract symptoms associated with benign prostatic hyperplasia. J Urol. 2003; 170(2 Pt 1):498–502.
Article37. Debruyne FM, Van der Poel HG. Clinical experience in Europe with uroselective alpha1-antagonists. Eur Urol. 1999; 36:Suppl 1. 54–58.38. Goktas S, Kibar Y, Kilic S, Topac H, Coban H, Seckin B. Recovery of abnormal ejaculation by intermittent tamsulosin treatment. J Urol. 2006; 175:650–652.
Article39. Naruganahalli KS. Abnormal ejaculation associated with tamsulosin in benign prostatic hyperplasia patients. GOKTAS S, KIBAR Y: Recovery of abnormal ejaculation by intermittent tamsulosin treatment. J. Urol. (2006) 175:650-652. Expert Opin Investig Drugs. 2006; 15:1635–1638.
Article40. Barqawi AB, Myers JB, O'Donnell C, Crawford ED. The effect of alpha-blocker and 5alpha-reductase inhibitor intake on sexual health in men with lower urinary tract symptoms. BJU Int. 2007; 100:853–857.
Article41. Caldwell DM, Ades AE, Higgins JP. Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ. 2005; 331:897–900.
Article42. Ioannidis JP. Integration of evidence from multiple meta-analyses: a primer on umbrella reviews, treatment networks and multiple treatments meta-analyses. CMAJ. 2009; 181:488–493.
Article43. Sutton AJ, Duval SJ, Tweedie RL, Abrams KR, Jones DR. Empirical assessment of effect of publication bias on meta-analyses. BMJ. 2000; 320:1574–1577.
Article44. Kim JH, Shim SR, Lee WJ, Kim HJ, Kwon SS, Bae JH. Sociodemographic and lifestyle factors affecting the self-perception period of lower urinary tract symptoms of international prostate symptom score items. Int J Clin Pract. 2012; 66:1216–1223.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Impact of Tamsulosin on Ureter Stone Expulsion in Korean Patients: A Meta-Analysis of Randomized Controlled Studies
- A Prospective, Multicenter, Open-Label Study of Dose Escalation Therapy in Male Patients With Nocturia Refractory to 0.2-mg Tamsulosin Monotherapy
- Evidence Is Enough?: A Systematic Review and Network Meta-Analysis of the Efficacy of Tamsulosin 0.2 mg and Tamsulosin 0.4 mg as an Initial Therapeutic Dose in Asian Benign Prostatic Hyperplasia Patients
- Evaluation of the pharmacokinetics and food effects of a novel formulation tamsulosin 0.4 mg capsule compared with a 0.2 mg capsule in healthy male volunteers
- Letter to the Editor: Commentary on “Evidence Is Enough?: A Systematic Review and Network Meta-Analysis of the Efficacy of Tamsulosin 0.2 mg and Tamsulosin 0.4 mg as an Initial Therapeutic Dose in Asian Benign Prostatic Hyperplasia Patientsâ€