Ann Lab Med.
2017 Jan;37(1):9-17. 10.3343/alm.2017.37.1.9.
Morning Spot Urine Glucose-to-Creatinine Ratios Predict Overnight Urinary Glucose Excretion in Patients With Type 2 Diabetes
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Graduate School, Yonsei University College of Medicine, Seoul, Korea. bwanlee@yuhs.ac
- 2Diabetes Center, Severance Hospital, Seoul, Korea.
- 3Department of Laboratory Medicine, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2373608
- DOI: http://doi.org/10.3343/alm.2017.37.1.9
Abstract
- BACKGROUND
With the advent of sodium glucose co-transporter 2 inhibitors to control glucose and treat diabetes, laboratory data aided by either timed or spot glucose levels in the urine could be used as an alternative marker of drug response. The aim of this study was to assess the agreement between overnight urinary glucose excretion (UGE) and morning spot urinary glucose-to-creatinine ratio (UGCR).
METHODS
In this prospective cross-sectional study, we enrolled a total of 215 participants with either normal glucose tolerance (NGT), pre-diabetes, or type 2 diabetes mellitus (T2DM). To exclude external factors such as food intake and physical activity, urine samples collected overnight at an 8-hr interval and the first-voided morning spot urine were collected and compared.
RESULTS
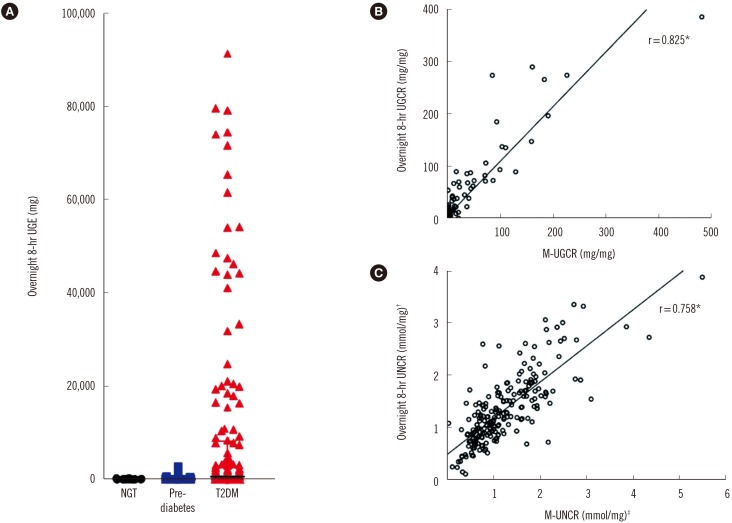
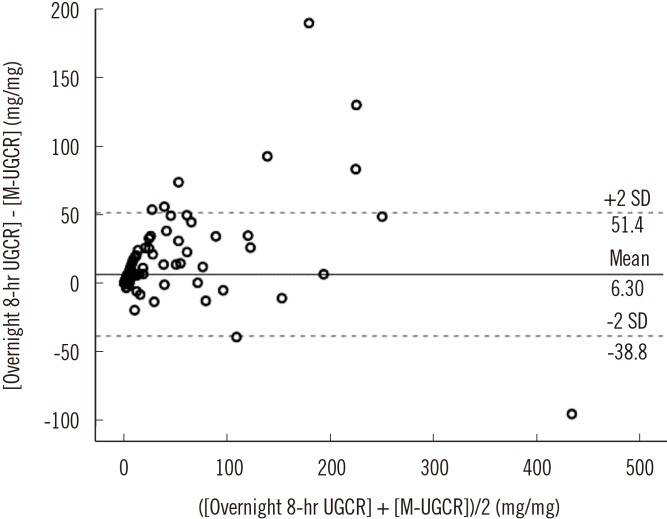
The median values of overnight 8-hr UGE in participants with NGT (N=14), pre-diabetes (N=41), and T2DM (N=160) were 35.0 mg, 35.6 mg, and 653.4 mg, respectively. In participants with T2DM, the median values of overnight 8-hr UGCR and first-voided morning spot UGCR (M-UGCR) were 1.37 mg/mg and 0.16 mg/mg, respectively. Quantitative analyses using an intraclass correlation coefficient (ICC) demonstrated a good reliability of measurement of the overnight 8-hr UGCR and M-UGCR (ICC=0.943, P<0.001). The M-UGCR was also significantly related to the overnight 8-hr UGE (r=0.828, P<0.001).
CONCLUSIONS
M-UGCR and overnight 8-hr UGCR showed good agreement, suggesting that M-UGCR be used as a simple index for estimating overnight amounts of UGE in patients with T2DM.
MeSH Terms
Figure
Reference
-
1. Aires I, Fila M, Polidori D, Santos AR, Costa AB, Calado J. Determination of the renal threshold for glucose excretion in familial renal glucosuria. Nephron. 2015; 129:300–304. PMID: 25896487.2. Tuvemo T, Ewald U, Wålinder O. Reevaluation of home urine glucose measurements in diabetic children. A computerized study of short-term control variables in the prediction of long-term diabetic control. Ups J Med Sci. 1984; 89:244–253. PMID: 6516065.3. Kverneland A, Kirkegaard BC, Wiesniewski J, Svendsen PA, Deckert T, Nerup J. [Evaluation of blood glucose determination and 24-hour urinary glucose excretion as indicators of mean blood glucose as expressed by HbA1c in patients with insulin-dependent diabetes mellitus]. Ugeskr Laeger. 1986; 148:1899–1901. PMID: 3750508.4. Kim JM, Hong JW, Won JC, Noh JH, Ko KS, Rhee BD, et al. Glycated hemoglobin value for fasting plasma glucose of 126 mg/dL in Korean: The 2011 Korea National Health and Nutrition Examination Survey. Diabetes Metab J. 2014; 38:480–483. PMID: 25541612.5. Jung CH, Jang JE, Park JY. A novel therapeutic agent for type 2 diabetes mellitus: SGLT2 inhibitor. Diabetes Metab J. 2014; 38:261–273. PMID: 25215272.6. List JF, Woo V, Morales E, Tang W, Fiedorek FT. Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes. Diabetes Care. 2009; 32:650–657. PMID: 19114612.7. Rosenstock J, Aggarwal N, Polidori D, Zhao Y, Arbit D, Usiskin K, et al. Dose-ranging effects of canagliflozin, a sodium-glucose cotransporter 2 inhibitor, as add-on to metformin in subjects with type 2 diabetes. Diabetes Care. 2012; 35:1232–1238. PMID: 22492586.8. Wilding JP. The role of the kidneys in glucose homeostasis in type 2 diabetes: clinical implications and therapeutic significance through sodium glucose co-transporter 2 inhibitors. Metabolism. 2014; 63:1228–1237. PMID: 25104103.9. Lee YH, Kown MH, Kim KJ, Lee EY, Kim D, Lee BW, et al. Inverse association between glycated albumin and insulin secretory function may explain higher levels of glycated albumin in subjects with longer duration of diabetes. PLoS One. 2014; 9:e108772. PMID: 25265016.10. Lee EY, Hwang S, Lee SH, Lee YH, Choi AR, Lee Y, et al. Postprandial C-peptide to glucose ratio as a predictor of β-cell function and its usefulness for staged management of type 2 diabetes. J Diabetes Investig. 2014; 5:517–524.11. Levey AS, Stevens LA, Schmid CH, Zhang Y, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150:604–612. PMID: 19414839.12. Brown IJ, Dyer AR, Chan Q, Cogswell ME, Ueshima H, Stamler J, et al. Estimating 24-hour urinary sodium excretion from casual urinary sodium concentrations in Western populations: the INTERSALT study. Am J Epidemiol. 2013; 177:1180–1192. PMID: 23673246.13. Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977; 33:159–174. PMID: 843571.14. Graybill FA, editor. An introduction to linear statistical models. Vol 1. [An]. New York: McGraw-Hill;1959.15. Guenther WC. Desk calculation of probabilities for the distribution of the sample correlation coefficient. Am Stat. 1977; 31:45–48.16. Zar JH. Biostatistical analysis. 2nd. Englewood Cliffs, NJ: Prentice Hall;1984.17. Hayford JT, Weydert JA, Thompson RG. Validity of urine glucose measurements for estimating plasma glucose concentration. Diabetes Care. 1983; 6:40–44.18. Wolf S, Rave K, Heinemann L, Roggen K. Renal glucose excretion and tubular reabsorption rate related to blood glucose in subjects with type 2 diabetes with a critical reappraisal of the "renal glucose threshold" model. Horm Metab Res. 2009; 41:600–604. PMID: 19418417.19. Walford S, Page MM, Allison SP. The influence of renal threshold on the interpretation of urine tests for glucose in diabetic patients. Diabetes Care. 1980; 3:672–674. PMID: 7449599.20. Kim SK, Kim HJ, Kim T, Hur KY, Kim SW, Lee MK, et al. Effectiveness of 3-day continuous glucose monitoring for improving glucose control in type 2 diabetic patients in clinical practice. Diabetes Metab J. 2014; 38:449–455. PMID: 25541608.21. Suh S, Joung JY, Jin SM, Kim MY, Bae JC, Park HD, et al. Strong correlation between glycaemic variability and total glucose exposure in type 2 diabetes is limited to subjects with satisfactory glycaemic control. Diabetes Metab. 2014; 40:272–277. PMID: 24630733.22. Baek JH, Jin SM, Kaku K, Jung JA, Kim JR, Ko JW, et al. Efficacy of mitiglinide and sitagliptin, alone or in combination, on postprandial excursion and glycemic variability assessed by continuous glucose monitoring: a post hoc analysis with single-day treatment. Expert Opin Pharmacother. 2015; 16:1127–1136. PMID: 25881690.23. Lee SW, Lee S, Kim SH, Kim TH, Kang BS, Yoo SH, et al. Parameters measuring beta-cell function are only valuable in diabetic subjects with low body mass index, high blood glucose level, or long-standing diabetes. Yonsei Med J. 2011; 52:939–947. PMID: 22028157.24. Polidori D, Sha S, Ghosh A, Plum-Mörschel L, Heise T, Rothenberg P. Validation of a novel method for determining the renal threshold for glucose excretion in untreated and canagliflozin-treated subjects with type 2 diabetes mellitus. J Clin Endocrinol Metab. 2013; 98:E867–E871. PMID: 23585665.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of Random Urine Sodium and Potassium Compensated by Creatinine as Possible Alternative Markers for 24 Hours Urinary Sodium and Potassium Excretion
- A Lower Baseline Urinary Glucose Excretion Predicts a Better Response to the Sodium Glucose Cotransporter 2 Inhibitor
- Estimation of Microalbuminuria by Urinary Albumin to Creatinine Concentration Ratio
- Spot Urine Albumin to Creatinine Ratio and Serum Cystatin C are Effective for Detection of Diabetic Nephropathy in Childhood Diabetic Patients
- The Factors Affecting Accurate Quantitaion of Proteinuria Using Spot Urine Protein/Creatinine Ratio in Children