J Gastric Cancer.
2012 Sep;12(3):179-186.
18F-2-Deoxy-2-Fluoro-D-Glucose Positron Emission Tomography: Computed Tomography for Preoperative Staging in Gastric Cancer Patients
- Affiliations
-
- 1Department of Surgery, Kosin University College of Medicine, Busan, Korea. yoonkiyoung@naver.com
Abstract
- PURPOSE
The use of 18F-2-deoxy-2-fluoro-D-glucose positron emission tomography-computed tomography as a routine preoperative modality is increasing for gastric cancer despite controversy with its usefulness in preoperative staging. In this study we aimed to determine the usefulness of preoperative positron emission tomography-computed tomography scans for staging of gastric cancer.
MATERIALS AND METHODS
We retrospectively analyzed 396 patients' positron emission tomography-computed tomography scans acquired for preoperative staging from January to December 2009.
RESULTS
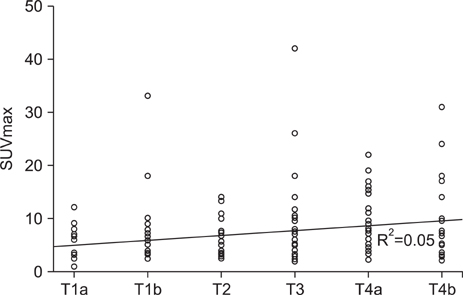
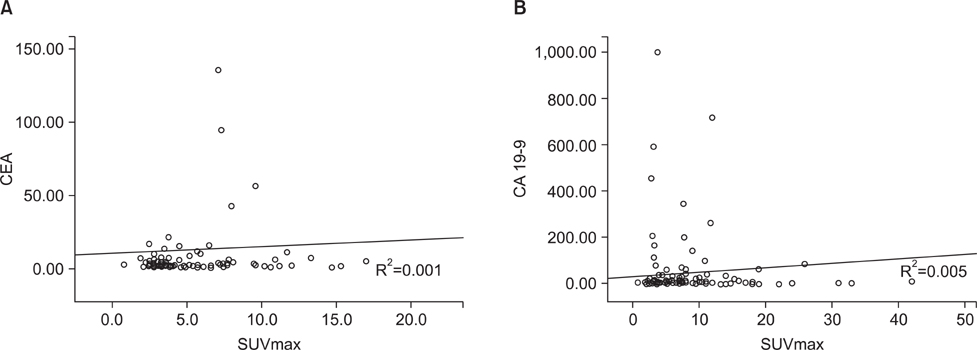
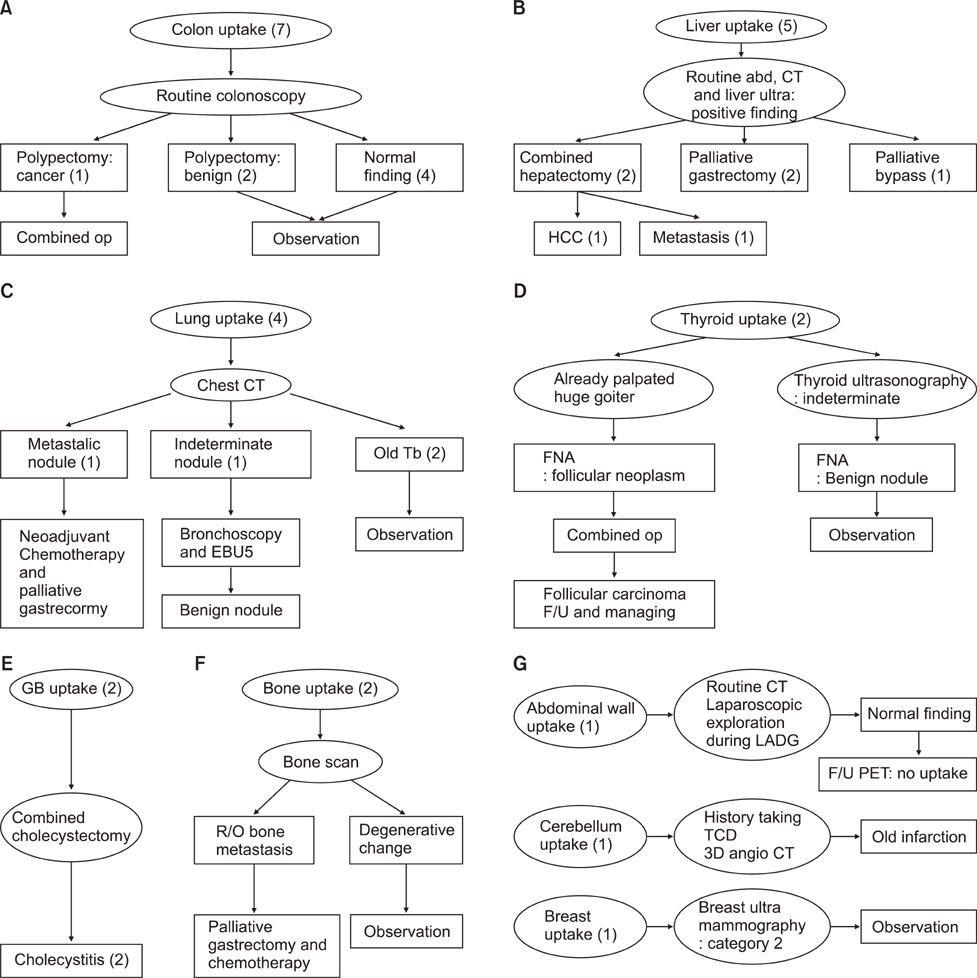
The sensitivity of positron emission tomography-computed tomography for detecting early gastric cancer was 20.7% and it was 74.2% for advanced gastric cancer. The size of the primary tumor was correlated with sensitivity, and there was a positive correlation between T stage and sensitivity. For regional lymph node metastasis, the sensitivity and specificity of the positron emission tomography-computed tomography were 30.7% and 94.7%, respectively. There was no correlation between T stage and maximum standardized uptake value or between tumor markers and maximum standardized uptake value. Fluorodeoxyglucose uptake was detected by positron emission tomography-computed tomography in 24 lesions other than the primary tumors. Among them, nine cases were found to be malignant, including double primary cancers and metastatic cancers. Only two cases were detected purely by positron emission tomography-computed tomography.
CONCLUSIONS
Positron emission tomography-computed tomography could be useful in detecting metastasis or another primary cancer for preoperative staging in gastric cancer patients, but not for T or N staging. More prospective studies are needed to determine whether positron emission tomography-computed tomography scans should be considered a routine preoperative imaging modality.
MeSH Terms
Figure
Reference
-
1. Kelley JR, Duggan JM. Gastric cancer epidemiology and risk factors. J Clin Epidemiol. 2003. 56:1–9.
Article2. Kim JP. Surgical results in gastric cancer. Semin Surg Oncol. 1999. 17:132–138.
Article3. Kim EY, Lee WJ, Choi D, Lee SJ, Choi JY, Kim BT, et al. The value of PET/CT for preoperative staging of advanced gastric cancer: comparison with contrast-enhanced CT. Eur J Radiol. 2011. 79:183–188.
Article4. Mochiki E, Kuwano H, Katoh H, Asao T, Oriuchi N, Endo K. Evaluation of 18F-2-deoxy-2-fluoro-D-glucose positron emission tomography for gastric cancer. World J Surg. 2004. 28:247–253.
Article5. Dassen AE, Lips DJ, Hoekstra CJ, Pruijt JF, Bosscha K. FDG-PET has no definite role in preoperative imaging in gastric cancer. Eur J Surg Oncol. 2009. 35:449–455.
Article6. Kim SK, Kang KW, Lee JS, Kim HK, Chang HJ, Choi JY, et al. Assessment of lymph node metastases using 18F-FDG PET in patients with advanced gastric cancer. Eur J Nucl Med Mol Imaging. 2006. 33:148–155.
Article7. Hiki Y, Shimao J, Yamao Y, Kobayashi N, Kuranami M, Kikuchi S, et al. The concepts, procedures, and problems related in endoscopic laser therapy of early gastric cancer. A retrospective study on early gastric cancer. Surg Endosc. 1989. 3:1–6.
Article8. Ohgami M, Otani Y, Kumai K, Kubota T, Kim YI, Kitajima M. Curative laparoscopic surgery for early gastric cancer: five years experience. World J Surg. 1999. 23:187–192.
Article9. Marczell AP, Rosen HR, Hentschel E. Diagnosis and tactical approach to surgery for early gastric carcinoma: a retrospective analysis of the past 16 years in an Austrian general hospital. Gastroenterol Jpn. 1989. 24:732–736.
Article10. Han EJ, Choi WH, Chung YA, Kim KJ, Maeng LS, Sohn KM, et al. Comparison between FDG uptake and clinicopathologic and immunohistochemical parameters in pre-operative PET/CT scan of primary gastric carcinoma. Nucl Med Mol Imaging. 2009. 43:26–34.11. Sarela AI, Miner TJ, Karpeh MS, Coit DG, Jaques DP, Brennan MF. Clinical outcomes with laparoscopic stage M1, unresected gastric adenocarcinoma. Ann Surg. 2006. 243:189–195.
Article12. Yeung HW, Macapinlac H, Karpeh M, Finn RD, Larson SM. Accuracy of FDG-PET in gastric cancer. Preliminary experience. Clin Positron Imaging. 1998. 1:213–221.
Article13. Stahl A, Ott K, Weber WA, Becker K, Link T, Siewert JR, et al. FDG PET imaging of locally advanced gastric carcinomas: correlation with endoscopic and histopathological findings. Eur J Nucl Med Mol Imaging. 2003. 30:288–295.
Article14. Yun M, Lim JS, Noh SH, Hyung WJ, Cheong JH, Bong JK, et al. Lymph node staging of gastric cancer using (18)F-FDG PET: a comparison study with CT. J Nucl Med. 2005. 46:1582–1588.15. Park MJ, Lee WJ, Lim HK, Park KW, Choi JY, Kim BT. Detecting recurrence of gastric cancer: the value of FDG PET/CT. Abdom Imaging. 2009. 34:441–447.
Article16. Lordick F, Ott K, Krause BJ, Weber WA, Becker K, Stein HJ, et al. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: the MUNICON phase II trial. Lancet Oncol. 2007. 8:797–805.
Article17. Pieterman RM, van Putten JW, Meuzelaar JJ, Mooyaart EL, Vaalburg W, Koëter GH, et al. Preoperative staging of non-small-cell lung cancer with positron-emission tomography. N Engl J Med. 2000. 343:254–261.
Article18. Flamen P, Lerut A, Van Cutsem E, De Wever W, Peeters M, Stroobants S, et al. Utility of positron emission tomography for the staging of patients with potentially operable esophageal carcinoma. J Clin Oncol. 2000. 18:3202–3210.
Article19. Valk PE, Pounds TR, Hopkins DM, Haseman MK, Hofer GA, Greiss HB, et al. Staging non-small cell lung cancer by whole-body positron emission tomographic imaging. Ann Thorac Surg. 1995. 60:1573–1581.
Article20. Vitola JV, Delbeke D, Sandler MP, Campbell MG, Powers TA, Wright JK, et al. Positron emission tomography to stage suspected metastatic colorectal carcinoma to the liver. Am J Surg. 1996. 171:21–26.
Article21. Kole AC, Plukker JT, Nieweg OE, Vaalburg W. Positron emission tomography for staging of oesophageal and gastroesophageal malignancy. Br J Cancer. 1998. 78:521–527.
Article22. Ott K, Fink U, Becker K, Stahl A, Dittler HJ, Busch R, et al. Prediction of response to preoperative chemotherapy in gastric carcinoma by metabolic imaging: results of a prospective trial. J Clin Oncol. 2003. 21:4604–4610.
Article23. Mukai K, Ishida Y, Okajima K, Isozaki H, Morimoto T, Nishiyama S. Usefulness of preoperative FDG-PET for detection of gastric cancer. Gastric Cancer. 2006. 9:192–196.
Article24. Kawamura T, Kusakabe T, Sugino T, Watanabe K, Fukuda T, Nashimoto A, et al. Expression of glucose transporter-1 in human gastric carcinoma: association with tumor aggressiveness, metastasis, and patient survival. Cancer. 2001. 92:634–641.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Positron Emission Tomography: Application in Pediatric Epilepsy
- 18F-fluoro-2-deoxy-D-glucose positron emission tomography findings of neurolymphomatosis
- Role of 18F 2-fluoro-2-deoxyglucose Positron Emission Tomography in Upper Gastrointestinal Malignancies
- Non-Malignant 18F-FDG Uptake in the Thorax by Positron Emission Tomography Computed Tomography Fusion Imaging
- F18-fluorodeoxyglucose-positron emission tomography and computed tomography is not accurate in preoperative staging of gastric cancer