Ann Dermatol.
2016 Dec;28(6):753-756. 10.5021/ad.2016.28.6.753.
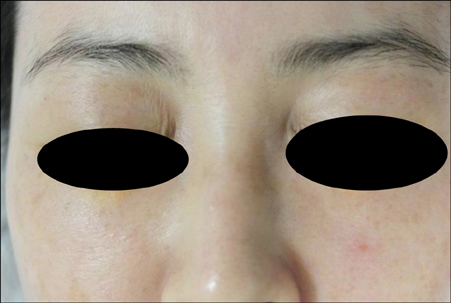
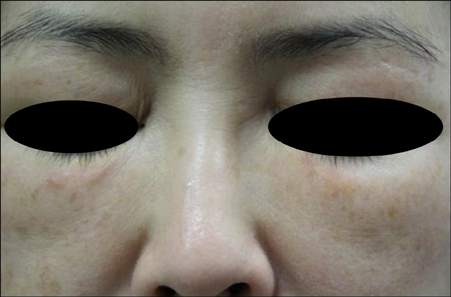
Late-Onset Complication of Fillers: Paraffinoma of the Lower Eyelids Clinically Mimicking Xanthelasma
- Affiliations
-
- 1Department of Dermatology, SMG-SNU Boramae Medical Center, Seoul, Korea. sycho@snu.ac.kr
- KMID: 2368127
- DOI: http://doi.org/10.5021/ad.2016.28.6.753
Abstract
- Injectable poly-L-lactic acid (PLLA) is world-famous filler used in lipoatrophy and facial rejuvenation because of its collagen neogenesis effect which leads to gradual volume restoration. Until recently, quite a number of unwanted adverse events of PLLA have been reported. However, to the best of our knowledge, paraffinoma as a complication of PLLA has never been reported. We herein describe the first case of paraffinoma after Sculptra® injection and propose its possible mechanism.
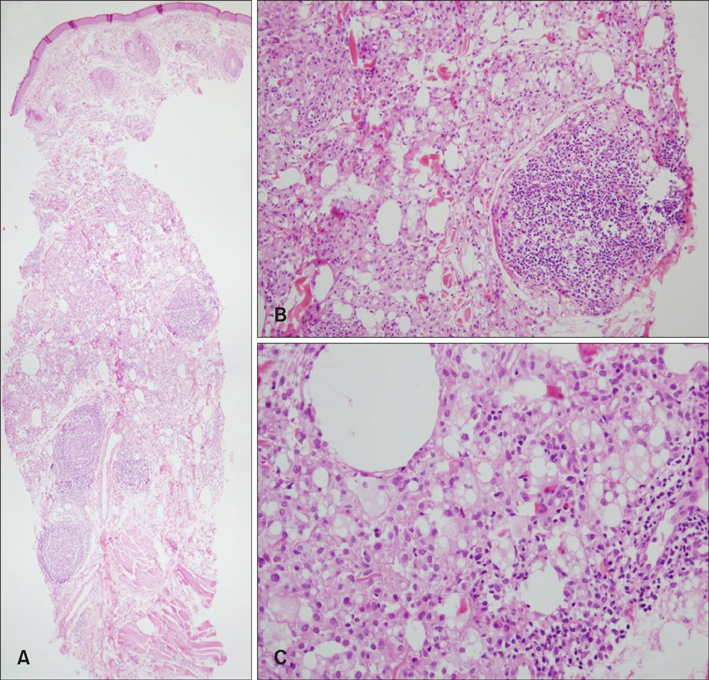
Figure
Reference
-
1. Bartus C, William Hanke C, Daro-Kaftan E. A decade of experience with injectable poly-L-lactic acid: a focus on safety. Dermatol Surg. 2013; 39:698–705.
Article2. Alijotas-Reig J, Garcia-Gimenez V, Vilardell-Tarres M. Late-onset immune-mediated adverse effects after poly-L-lactic acid injection in non-HIV patients: clinical findings and long-term follow-up. Dermatology. 2009; 219:303–308.
Article3. Lowe NJ, Maxwell CA, Lowe P, Shah A, Patnaik R. Injectable poly-l-lactic acid: 3 years of aesthetic experience. Dermatol Surg. 2009; 35:Suppl 1. 344–349.4. Duracinsky M, Leclercq P, Herrmann S, Christen MO, Dolivo M, Goujard C, et al. Safety of poly-L-lactic acid (New-Fill®) in the treatment of facial lipoatrophy: a large observational study among HIV-positive patients. BMC Infect Dis. 2014; 14:474.
Article5. Kim JH, Choi JS, Yun JH, Kang HK, Baek JO, Roh JY, et al. Foreign body reaction to injectable hyaluronic acid: late granuloma formation. Ann Dermatol. 2015; 27:224–225.
Article6. Lemperle G, Gauthier-Hazan N, Wolters M, Eisemann-Klein M, Zimmermann U, Duffy DM. Foreign body granulomas after all injectable dermal fillers: part 1. Possible causes. Plast Reconstr Surg. 2009; 123:1842–1863.
Article7. Requena L, Requena C, Christensen L, Zimmermann US, Kutzner H, Cerroni L. Adverse reactions to injectable soft tissue fillers. J Am Acad Dermatol. 2011; 64:1–34. quiz 35-36.
Article8. Park KY, Choi SY, Seo SJ, Hong CK. Posttraumatic lipogranuloma on the lower leg. J Dermatol. 2013; 40:141–142.
Article9. Shan SJ, Guo Y. Exenatide-induced eosinophilic sclerosing lipogranuloma at the injection site. Am J Dermatopathol. 2014; 36:510–512.
Article10. Abel AD, Carlson JA, Bakri S, Meyer DR. Sclerosing lipogranuloma of the orbit after periocular steroid injection. Ophthalmology. 2003; 110:1841–1845.
Article11. Dailey RA, Gray JF, Rubin MG, Hildebrand PL, Swanson NA, Wobig JL, et al. Histopathologic changes of the eyelid skin following trichloroacetic acid chemical peel. Ophthal Plast Reconstr Surg. 1998; 14:9–12.
Article12. Agarwal N, Mittal A, Kuldeep C, Gupta LK, Khare AK, Mehta S. Chemical reconstruction of skin scars therapy using 100% trichloroacetic acid in the treatment of atrophic facial post varicella scars: a pilot study. J Cutan Aesthet Surg. 2013; 6:144–147.
Article13. Haque MU, Ramesh V. Evaluation of three different strengths of trichloroacetic acid in xanthelasma palpebrarum. J Dermatolog Treat. 2006; 17:48–50.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Erratum: Late-Onset Complication of Fillers: Paraffinoma of the Lower Eyelids Clinically Mimicking Xanthelasma
- A Case of Bilateral Extensive Xanthelasma
- Atypical Facial Filler Granuloma: Comparative Histologic Analysis with Paraffinoma
- Clinical Characteristics of Xanthelasma Palpebrarum and Clinical Effects of Upper Blepharoplasty with Local Flap Technique
- The Use of Pulsed Dye Laser for the Treatment of Xanthelasma Palpebrum