J Korean Med Sci.
2016 Apr;31(4):611-616. 10.3346/jkms.2016.31.4.611.
Naturalistic Pharmacotherapy Compliance among Pediatric Patients with Attention Deficit/Hyperactivity Disorder: a Study Based on Three-Year Nationwide Data
- Affiliations
-
- 1Department of Psychiatry, Myongji Hospital, Seonam University College of Medicine, Goyang, Korea.
- 2Department of Psychiatry, Sanggyepaik Hospital School of Medicine, Inje University, Seoul, Korea.
- 3Department of Psychiatry, Kangwon National University School of Medicine, Chuncheon, Korea.
- 4Department of Psychiatry, Eulji University, College of Medicine, Seoul, Korea.
- 5Department of Biostatistics, Graduate School of Public Health, Seoul National University, Seoul, Korea.
- 6Department of Preventive Medicine, Kyung Hee University, School of Medicine, Seoul, Korea.
- 7Department of Psychiatry, Soonchunhyang University, School of Medicine, Seoul, Korea.
- 8Department of Psychiatry, Kyung Hee University, School of Medicine, Seoul, Korea. mompeian@khu.ac.kr
- KMID: 2363702
- DOI: http://doi.org/10.3346/jkms.2016.31.4.611
Abstract
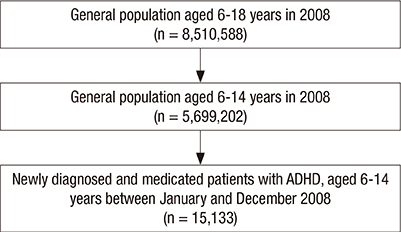
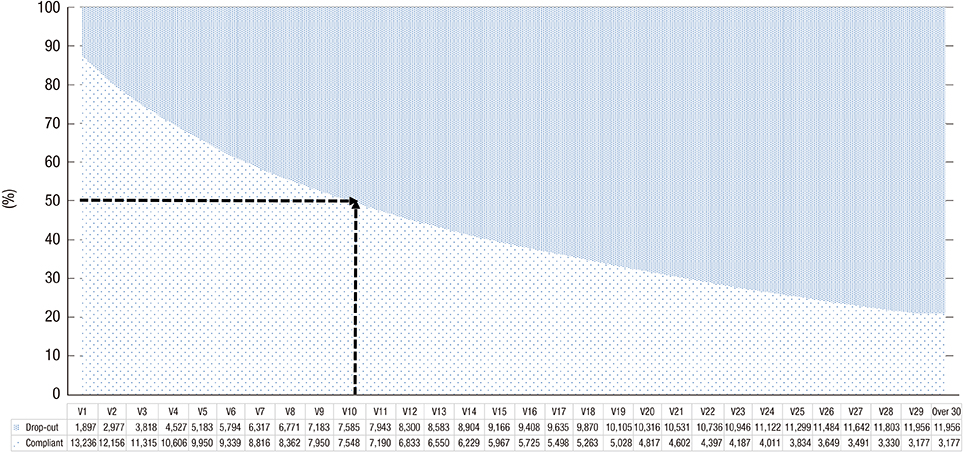
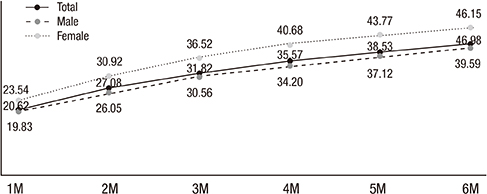
- We examined short- and long-term medication compliance among youth with attention-deficit hyperactivity disorder (ADHD), using data from the National Health Insurance database in Korea. Of the 5,699,202 6-14-year-old youth in 2008, we chose those with at least 1 medical claim containing an ICD-10 code for diagnosis of ADHD (F90.0) and no prescription for ADHD within the previous 365 days. We tracked the data every 6 months between 2008 and 2011, to determine treatment compliance among newly diagnosed, medicated patients. Further, we checked every 1 month of the 6 months after treatment commencement. Treatment continuity for each patient was calculated by sequentially counting the continuous prescriptions. For measuring compliance, we applied the medication possession ratio (MPR) as 0.6, 0.7, and 0.8, and the gap method as 15- and 30-days' intervals. There were 15,133 subjects; 11,934 (78.86%) were boys. Overall 6-month treatment compliance was 59.0%, 47.3%, 39.9%, 34.1%, 28.6%, and 23.1%. Monthly drop-out rates within the first 6 months were 20.6%, 6.5%, 4.7%, 3.7%, 3.0%, and 2.5%, respectively. When applying MPR more strictly or shorter gap days, treatment compliance lessened. This is the first nationwide report on 36-month treatment compliance of the whole population of 6-14-year-olds with ADHD. We found the beginning of the treatment, especially the first month, to be a critical period in pharmacotherapy. These results also suggest the importance of setting appropriate treatment adherence standards for patients with ADHD, considering the chronic course of ADHD.
Keyword
MeSH Terms
-
Adolescent
Attention Deficit Disorder with Hyperactivity/diagnosis/*drug therapy
Central Nervous System Stimulants/*therapeutic use
Child
Databases, Factual
Female
Humans
Male
*Medication Adherence
Methylphenidate/*therapeutic use
Republic of Korea
Retrospective Studies
Central Nervous System Stimulants
Methylphenidate
Figure
Reference
-
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association Publishing;2013.2. Faraone SV, Biederman J, Mick E. The age-dependent decline of attention deficit hyperactivity disorder: a meta-analysis of follow-up studies. Psychol Med. 2006; 36:159–165.3. Kessler RC, Adler LA, Gruber MJ, Sarawate CA, Spencer T, Van Brunt DL. Validity of the World Health Organization Adult ADHD Self-Report Scale (ASRS) screener in a representative sample of health plan members. Int J Methods Psychiatr Res. 2007; 16:52–65.4. Mannuzza S, Klein RG, Bessler A, Malloy P, LaPadula M. Adult outcome of hyperactive boys. Educational achievement, occupational rank, and psychiatric status. Arch Gen Psychiatry. 1993; 50:565–576.5. Barkley RA, Fischer M, Edelbrock CS, Smallish L. The adolescent outcome of hyperactive children diagnosed by research criteria: I. An 8-year prospective follow-up study. J Am Acad Child Adolesc Psychiatry. 1990; 29:546–557.6. Biederman J, Faraone S, Milberger S, Curtis S, Chen L, Marrs A, Ouellette C, Moore P, Spencer T. Predictors of persistence and remission of ADHD into adolescence: results from a four-year prospective follow-up study. J Am Acad Child Adolesc Psychiatry. 1996; 35:343–351.7. Claude D, Firestone P. The development of ADHD boys: a 12-year follow-up. Can J Behav Sci. 1995; 27:226–249.8. Bernfort L, Nordfeldt S, Persson J. ADHD from a socio-economic perspective. Acta Paediatr. 2008; 97:239–245.9. Mannuzza S, Klein RG. Long-term prognosis in attention-deficit/hyperactivity disorder. Child Adolesc Psychiatr Clin N Am. 2000; 9:711–726.10. Barkley RA, Fischer M, Smallish L, Fletcher K. Young adult outcome of hyperactive children: adaptive functioning in major life activities. J Am Acad Child Adolesc Psychiatry. 2006; 45:192–202.11. Gau SS, Chen SJ, Chou WJ, Cheng H, Tang CS, Chang HL, Tzang RF, Wu YY, Huang YF, Chou MC, et al. National survey of adherence, efficacy, and side effects of methylphenidate in children with attention-deficit/hyperactivity disorder in Taiwan. J Clin Psychiatry. 2008; 69:131–140.12. Atzori P, Usala T, Carucci S, Danjou F, Zuddas A. Predictive factors for persistent use and compliance of immediate-release methylphenidate: a 36-month naturalistic study. J Child Adolesc Psychopharmacol. 2009; 19:673–681.13. Rothenberger A, Becker A, Breuer D, Döpfner M. An observational study of once-daily modified-release methylphenidate in ADHD: quality of life, satisfaction with treatment and adherence. Eur Child Adolesc Psychiatry. 2011; 20:Suppl 2. S257–65.14. Yang J, Yoon BM, Lee MS, Joe SH, Jung IK, Kim SH. Adherence with electronic monitoring and symptoms in children with attention deficit hyperactivity disorder. Psychiatry Investig. 2012; 9:263–268.15. Winterstein AG, Gerhard T, Shuster J, Zito J, Johnson M, Liu H, Saidi A. Utilization of pharmacologic treatment in youths with attention deficit/hyperactivity disorder in Medicaid database. Ann Pharmacother. 2008; 42:24–31.16. Wong IC, Asherson P, Bilbow A, Clifford S, Coghill D, DeSoysa R, Hollis C, McCarthy S, Murray M, Planner C, et al. Cessation of attention deficit hyperactivity disorder drugs in the young (CADDY)--a pharmacoepidemiological and qualitative study. Health Technol Assess. 2009; 13:iii–iiv. ix–xi. 1–120.17. Garbe E, Mikolajczyk RT, Banaschewski T, Petermann U, Petermann F, Kraut AA, Langner I. Drug treatment patterns of attention-deficit/hyperactivity disorder in children and adolescents in Germany: results from a large population-based cohort study. J Child Adolesc Psychopharmacol. 2012; 22:452–458.18. Hong M, Lee WH, Moon DS, Lee SM, Chung US, Bahn GH. 36 month naturalistic retrospective study of clinic-treated youth with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2014; 24:341–346.19. Lawson KA, Johnsrud M, Hodgkins P, Sasané R, Crismon ML. Utilization patterns of stimulants in ADHD in the Medicaid population: a retrospective analysis of data from the Texas Medicaid program. Clin Ther. 2012; 34:944–956.e4.20. Hong M, Kwack YS, Joung YS, Lee SI, Kim B, Sohn SH, Chung US, Yang J, Bhang SY, Hwang JW, et al. Nationwide rate of attention-deficit hyperactivity disorder diagnosis and pharmacotherapy in Korea in 2008-2011. Asia-Pac Psychiatry. 2014; 6:379–385.21. Wolraich ML, Hannah JN, Baumgaertel A, Feurer ID. Examination of DSM-IV criteria for attention deficit/hyperactivity disorder in a county-wide sample. J Dev Behav Pediatr. 1998; 19:162–168.22. Angold A, Erkanli A, Egger HL, Costello EJ. Stimulant treatment for children: a community perspective. J Am Acad Child Adolesc Psychiatry. 2000; 39:975–984.23. Yoshimasu K, Barbaresi WJ, Colligan RC, Voigt RG, Killian JM, Weaver AL, Katusic SK. Childhood ADHD is strongly associated with a broad range of psychiatric disorders during adolescence: a population-based birth cohort study. J Child Psychol Psychiatry. 2012; 53:1036–1043.24. Steiner JF, Prochazka AV. The assessment of refill compliance using pharmacy records: methods, validity, and applications. J Clin Epidemiol. 1997; 50:105–116.25. Peterson AM, Nau DP, Cramer JA, Benner J, Gwadry-Sridhar F, Nichol M. A checklist for medication compliance and persistence studies using retrospective databases. Value Health. 2007; 10:3–12.26. Barner JC, Khoza S, Oladapo A. ADHD medication use, adherence, persistence and cost among Texas Medicaid children. Curr Med Res Opin. 2011; 27:Suppl 2. 13–22.27. Cramer JA, Roy A, Burrell A, Fairchild CJ, Fuldeore MJ, Ollendorf DA, Wong PK. Medication compliance and persistence: terminology and definitions. Value Health. 2008; 11:44–47.28. Hodgkins P, Sasané R, Meijer WM. Pharmacologic treatment of attention-deficit/hyperactivity disorder in children: incidence, prevalence, and treatment patterns in the Netherlands. Clin Ther. 2011; 33:188–203.29. Kim YJ, Oh SY, Lee J, Moon SJ, Lee WH, Bahn GH. Factors affecting adherence to pharmacotherapy in children with attention-deficit hyperactivity disorder: a retrospective study. J Korean Acad Child Adolesc Psychiatry. 2010; 21:174–181.30. Smith E, Koerting J, Latter S, Knowles MM, McCann DC, Thompson M, Sonuga-Barke EJ. Overcoming barriers to effective early parenting interventions for attention-deficit hyperactivity disorder (ADHD): parent and practitioner views. Child Care Health Dev. 2015; 41:93–102.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Non-Stimulant Medications in the Treatment of Attention-Deficit Hyperactivity Disorder
- Pharmacotherapy for attention-deficit/hyperactivity disorder
- Diagnosis and Management of Attention Deficit Hyperactivity Disorder
- Effectiveness and Tolerability of Combination Pharmacotherapy With Stimulant and Non-Stimulant in Children With Attention Deficit Hyperactivity Disorder
- OROS Methylphenidate Treatment of Secondary Adult ADHD after Traumatic Brain Injury