Immune Netw.
2016 Dec;16(6):344-357. 10.4110/in.2016.16.6.344.
Reference Values for a Panel of Cytokinergic and Regulatory Lymphocyte Subpopulations
- Affiliations
-
- 1Maria Paola Belloni Center for Personalized Medicine, Data Medica Group, Padova 35100, Italy. alessandro.buriani@gmail.com
- 2Department of Pharmaceutical and Pharmacological Sciences, University of Padova, Padova 35100, Italy.
- 3Department of Biomedical Sciences, University of Padova, Padova 35100, Italy.
- KMID: 2362799
- DOI: http://doi.org/10.4110/in.2016.16.6.344
Abstract
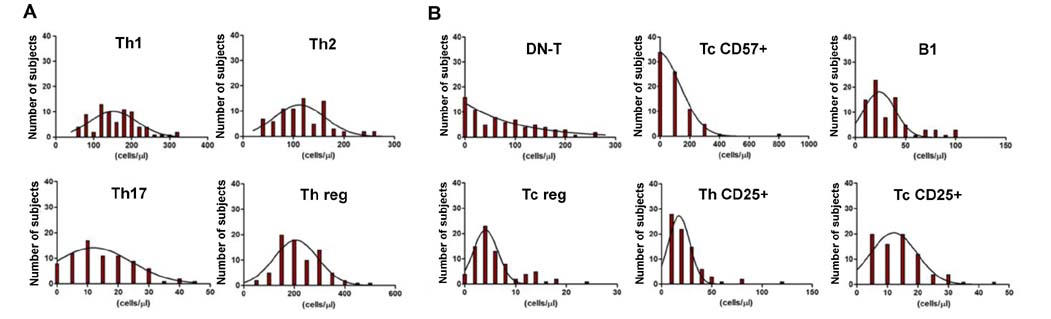
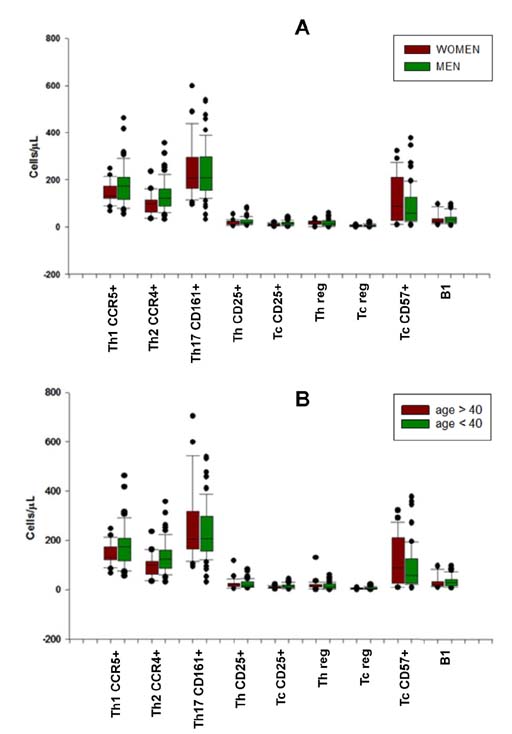
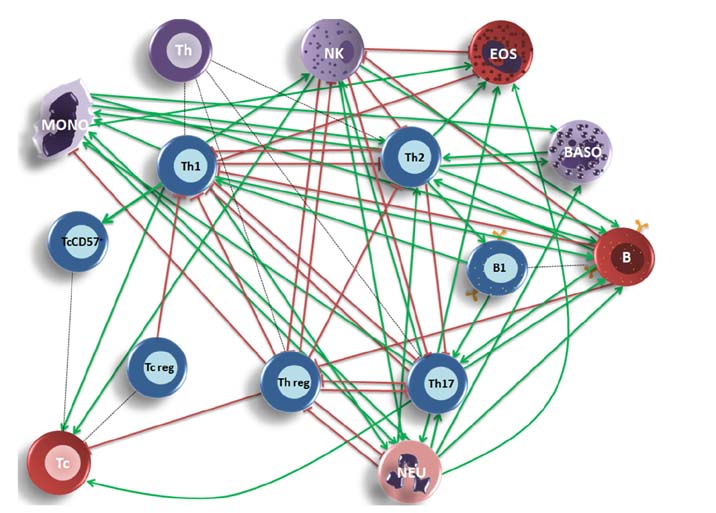
- Lymphocyte subpopulations producing cytokines and exerting regulatory functions represent key immune elements. Given their reciprocal interdependency lymphocyte subpopulations are usually assayed as diagnostic panels, rather than single biomarkers for specialist clinical use. This retrospective analysis on lymphocyte subpopulations, analyzed over the last few years in an outpatient laboratory in Northeast Italy, contributes to the establishment of reference values for several regulatory lymphocytes currently lacking such reference ranges for the general population. Mean values and ranges in a sample of Caucasian patients (mean age 42±8,5 years), were provided for Th1, Th2, Th17, Th-reg, Tc-reg, Tc-CD57⺠and B1 lymphocytes. The results are consistent with what is found in literature for the single subtypes and are: Th1 157.8±60.3/µl (7.3%±2.9); Th2 118.2±52.2/µl (5.4%±2.5); Th17 221.6±90.2/µl (10.5%±4.4); Th-reg 15.1±10.2/µl (0.7%±0.4); Tc-reg 5.8±4.7/µl (0.3%±0.2); Tc-CD57+ 103.7±114.1/µl (4.6%±4.7); B1 33.7±22.8/µl (1.5%±0.9); (Values are mean±SD). The results show that despite their variability, mean values are rather consistent in all age or sex groups and can be used as laboratory internal reference for this regulatory panel. Adding regulatory cells to lymphocyte subpopulations panels allows a more complete view of the state of the subject's immune network balance, thus improving the personalization and the "actionability" of diagnostic data in a systems medicine perspective.
Keyword
MeSH Terms
Figure
Reference
-
1. Tosato F, Bernardi D, Sanzari MC, Pantano G, Plebani M. Biological variability of lymphocyte subsets of human adults' blood. Clin Chim Acta. 2013; 424:159–163.
Article2. Choi J, Lee SJ, Lee YA, Maeng HG, Lee JK, Kang YW. Reference values for peripheral blood lymphocyte subsets in a healthy korean population. Immune Netw. 2014; 14:289–295.
Article3. McCoy JP Jr, Keren DF. Current practices in clinical flow cytometry. A practice survey by the American Society of Clinical Pathologists. Am J Clin Pathol. 1999; 111:161–168.
Article4. Yanaba K, Bouaziz JD, Matsushita T, Magro CM, Clair EW St, Tedder TF. B-lymphocyte contributions to human autoimmune disease. Immunol Rev. 2008; 223:284–299.
Article5. Doreau A, Belot A, Bastid J, Riche B, Trescol-Biemont MC, Ranchin B, Fabien N, Cochat P, Pouteil-Noble C, Trolliet P, Durieu I, Tebib J, Kassai B, Ansieau S, Puisieux A, Eliaou JF, Bonnefoy-Berard N. Interleukin 17 acts in synergy with B cell-activating factor to influence B cell biology and the pathophysiology of systemic lupus erythematosus. Nat Immunol. 2009; 10:778–785.
Article6. Victorino RM, Hodgson HJ. Alteration in T lymphocyte subpopulations in inflammatory bowel disease. Clin Exp Immunol. 1980; 41:156–165.7. Blanco P, Viallard JF, Pellegrin JL, Moreau JF. Cytotoxic T lymphocytes and autoimmunity. Curr Opin Rheumatol. 2005; 17:731–734.
Article8. Niwa F, Kuriyama N, Nakagawa M, Imanishi J. Effects of peripheral lymphocyte subpopulations and the clinical correlation with Parkinson's disease. Geriatr Gerontol Int. 2012; 12:102–107.
Article9. Kieseier BC, Hartung HP, Wiendl H. Immune circuitry in the peripheral nervous system. Curr Opin Neurol. 2006; 19:437–445.
Article10. Stinissen P, Raus J, Zhang J. Autoimmune pathogenesis of multiple sclerosis: role of autoreactive T lymphocytes and new immunotherapeutic strategies. Crit Rev Immunol. 1997; 17:33–75.
Article11. Prat A, Biernacki K, Lavoie JF, Poirier J, Duquette P, Antel JP. Migration of multiple sclerosis lymphocytes through brain endothelium. Arch Neurol. 2002; 59:391–397.
Article12. Luckheeram RV, Zhou R, Verma AD, Xia B. CD4(+)T cells: differentiation and functions. Clin Dev Immunol. 2012; 2012:925135.13. Libby P, Hansson GK. Inflammation and immunity in diseases of the arterial tree: players and layers. Circ Res. 2015; 116:307–311.
Article14. Kim JD, Lee SH, Seo EH, Woo SY, Kim SH, Chung SM, Kim HJ. Role of Th1 and Th17 cells in the development and complexity of coronary artery disease: comparison analysis by the methods of flow cytometry and SYNTAX score. Coron Artery Dis. 2015; 26:604–611.
Article15. Robertson MJ, Schacterle RS, Mackin GA, Wilson SN, Bloomingdale KL, Ritz J, Komaroff AL. Lymphocyte subset differences in patients with chronic fatigue syndrome, multiple sclerosis and major depression. Clin Exp Immunol. 2005; 141:326–332.
Article16. Fehervari Z. Lymphocytes in Alzheimer's disease. Nat Immunol. 2016; 17:355.
Article17. Asano M, Toda M, Sakaguchi N, Sakaguchi S. Autoimmune disease as a consequence of developmental abnormality of a T cell subpopulation. J Exp Med. 1996; 184:387–396.
Article18. McNally A, Hill GR, Sparwasser T, Thomas R, Steptoe RJ. CD4+CD25+ regulatory T cells control CD8+ T-cell effector differentiation by modulating IL-2 homeostasis. Proc Natl Acad Sci U S A. 2011; 108:7529–7534.
Article19. Van Parijs L, Abbas AK. Homeostasis and self-tolerance in the immune system: turning lymphocytes off. Science. 1998; 280:243–248.
Article20. Stumhofer JS, Laurence A, Wilson EH, Huang E, Tato CM, Johnson LM, Villarino AV, Huang Q, Yoshimura A, Sehy D, Saris CJ, O'Shea JJ, Hennighausen L, Ernst M, Hunter CA. Interleukin 27 negatively regulates the development of interleukin 17-producing T helper cells during chronic inflammation of the central nervous system. Nat Immunol. 2006; 7:937–945.
Article21. Hood L, Tian Q. Systems approaches to biology and disease enable translational systems medicine. Genomics Proteomics Bioinformatics. 2012; 10:181–185.
Article22. Lemberger T. Systems biology in human health and disease. Mol Syst Biol. 2007; 3:136.
Article23. Lindquist JA, Schraven B. Systems biology of T cell activation. Ernst Schering Found Symp Proc. 2007; 43–61.
Article24. Hood L, Lovejoy JC, Price ND. Integrating big data and actionable health coaching to optimize wellness. BMC Med. 2015; 13:4.
Article25. Kutukculer N, Azarsiz E, Aksu G, Karaca NE. CD4+CD25+Foxp3+ T regulatory cells, Th1 (CCR5, IL-2, IFN-gamma) and Th2 (CCR4, IL-4, Il-13) type chemokine receptors and intracellular cytokines in children with common variable immunodeficiency. Int J Immunopathol Pharmacol. 2016; 29:241–251.
Article26. Yamamoto J, Adachi Y, Onoue Y, Adachi YS, Okabe Y, Itazawa T, Toyoda M, Seki T, Morohashi M, Matsushima K, Miyawaki T. Differential expression of the chemokine receptors by the Th1- and Th2-type effector populations within circulating CD4+ T cells. J Leukoc Biol. 2000; 68:568–574.27. Maggi L, Santarlasci V, Capone M, Peired A, Frosali F, Crome SQ, Querci V, Fambrini M, Liotta F, Levings MK, Maggi E, Cosmi L, Romagnani S, Annunziato F. CD161 is a marker of all human IL-17-producing T-cell subsets and is induced by RORC. Eur J Immunol. 2010; 40:2174–2181.
Article28. Gonzalez Y, Herrera MT, Juarez E, Salazar-Lezama MA, Bobadilla K, Torres M. CD161 expression defines a Th1/Th17 polyfunctional subset of resident memory T lymphocytes in bronchoalveolar cells. PLoS One. 2015; 10:e0123591.
Article29. Hannet I, Erkeller-Yuksel F, Lydyard P, Deneys V, DeBruyere M. Developmental and maturational changes in human blood lymphocyte subpopulations. Immunol Today. 1992; 13:215. 218.
Article30. Shearer WT, Rosenblatt HM, Gelman RS, Oyomopito R, Plaeger S, Stiehm ER, Wara DW, Douglas SD, Luzuriaga K, McFarland EJ, Yogev R, Rathore MH, Levy W, Graham BL, Spector SA. Lymphocyte subsets in healthy children from birth through 18 years of age: the pediatric AIDS clinical trials group P1009 study. J Allergy Clin Immunol. 2003; 112:973–980.
Article31. Nebe T, Bentzien F, Bruegel M, Fiedler GM, Gutensohn K, Heimpel H, Krebs N, Ossendorf M, Schuff-Werner P, Stamminger G, Baum H. Multi-centric determination of reference ranges for automated blood counts. Laboratoriums Medizin. 2011; 35.32. Herklotz R, Luthi U, Ottiger C, Huber AR. Metaanalysis of reference values in hematology. Ther Umsch. 2006; 63:5–24.33. Rout N. Enhanced Th1/Th17 functions of CD161+ CD8+ T cells in mucosal tissues of rhesus macaques. PLoS One. 2016; 11:e0157407.34. Ohkura N, Hamaguchi M, Sakaguchi S. FOXP3+ regulatory T cells: control of FOXP3 expression by pharmacological agents. Trends Pharmacol Sci. 2011; 32:158–166.
Article35. Rudensky AY. Regulatory T cells and Foxp3. Immunol Rev. 2011; 241:260–268.
Article36. Beres AJ, Haribhai D, Chadwick AC, Gonyo PJ, Williams CB, Drobyski WR. CD8+ Foxp3+ regulatory T cells are induced during graft-versus-host disease and mitigate disease severity. J Immunol. 2012; 189:464–474.
Article37. Santagostino A, Garbaccio G, Pistorio A, Bolis V, Camisasca G, Pagliaro P, Girotto M. An Italian national multicenter study for the definition of reference ranges for normal values of peripheral blood lymphocyte subsets in healthy adults. Haematologica. 1999; 84:499–504.38. Maini MK, Gilson RJ, Chavda N, Gill S, Fakoya A, Ross EJ, Phillips AN, Weller IV. Reference ranges and sources of variability of CD4 counts in HIV-seronegative women and men. Genitourin Med. 1996; 72:27–31.
Article39. Buchwald J, Berggren JL, Lützen J, Renn J, Fraser C, Sauer T, Shapiro A, Fischer , Hans . A history of the central limit theorem: from classical to modern probability theory. sources and studies in the history of mathematics and physical sciences. New York: springer;2011. p. 17–30. Chapter 2.40. Kim S, Zhao Z, Shao X. Nonparametric functional central limit theorem for time series regression with application to self-normalized confidence interval. J Multivar Anal. 2015; 133:277–290.
Article41. Shevach EM. CD4+ CD25+ suppressor T cells: more questions than answers. Nat Rev Immunol. 2002; 2:389–400.
Article42. Churlaud G, Pitoiset F, Jebbawi F, Lorenzon R, Bellier B, Rosenzwajg M, Klatzmann D. Human and mouse CD8(+)CD25(+)FOXP3(+) regulatory T cells at steady state and during interleukin-2 therapy. Front Immunol. 2015; 6:171.
Article43. Kared H, Martelli S, Ng TP, Pender SL, Larbi A. CD57 in human natural killer cells and T-lymphocytes. Cancer Immunol Immunother. 2016; 65:441–452.
Article44. Focosi D, Bestagno M, Burrone O, Petrini M. CD57+ T lymphocytes and functional immune deficiency. J Leukoc Biol. 2010; 87:107–116.45. Perez-Andres M, Grosserichter-Wagener C, Teodosio C, van Dongen JJ, Orfao A, van Zelm MC. The nature of circulating CD27+CD43+ B cells. J Exp Med. 2011; 208:2565–2566.46. Griffin DO, Holodick NE, Rothstein TL. Human B1 cells are CD3-: A reply to “A human equivalent of mouse B-1 cells?” and “The nature of circulating CD27+CD43+ B cells”. J Exp Med. 2011; 208:2566–2569.
Article47. Talaat RM, Elmaghraby AM, Barakat SS, El-Shahat M. Alterations in immune cell subsets and their cytokine secretion profile in childhood idiopathic thrombocytopenic purpura (ITP). Clin Exp Immunol. 2014; 176:291–300.
Article48. Wood KL, Twigg HL III, Doseff AI. Dysregulation of CD8+ lymphocyte apoptosis, chronic disease, and immune regulation. Front Biosci (Landmark Ed.). 2009; 14:3771–3781.49. Saunders JA, Estes KA, Kosloski LM, Allen HE, Dempsey KM, Torres-Russotto DR, Meza JL, Santamaria PM, Bertoni JM, Murman DL, Ali HH, Standaert DG, Mosley RL, Gendelman HE. CD4+ regulatory and effector/memory T cell subsets profile motor dysfunction in Parkinson's disease. J Neuroimmune Pharmacol. 2012; 7:927–938.
Article50. Salonen R, Ilonen J, Jagerroos H, Syrjala H, Nurmi T, Reunanen M. Lymphocyte subsets in the cerebrospinal fluid in active multiple sclerosis. Ann Neurol. 1989; 25:500–502.
Article51. Vojdani A, Lambert J. The Role of Th17 in Neuroimmune Disorders: Target for CAM Therapy. Part I. Evid Based Complement Alternat Med. 2011; 2011:927294.
Article52. Belardelli F. Role of interferons and other cytokines in the regulation of the immune response. APMIS. 1995; 103:161–179.
Article53. Sugita S, Kawazoe Y, Imai A, Yamada Y, Horie S, Mochizuki M. Inhibition of Th17 differentiation by anti-TNF-alpha therapy in uveitis patients with Behcet's disease. Arthritis Res Ther. 2012; 14:R99.54. Fletcher JM, Lalor SJ, Sweeney CM, Tubridy N, Mills KH. T cells in multiple sclerosis and experimental autoimmune encephalomyelitis. Clin Exp Immunol. 2010; 162:1–11.
Article55. Korn T, Bettelli E, Oukka M, Kuchroo VK. IL-17 and Th17 Cells. Annu Rev Immunol. 2009; 27:485–517.
Article56. Eisenstein EM, Williams CB. The T(reg)/Th17 cell balance: a new paradigm for autoimmunity. Pediatr Res. 2009; 65:26R–31R.
Article57. Kuo ML, Huang JL, Yeh KW, Li PS, Hsieh KH. Evaluation of Th1/Th2 ratio and cytokine production profile during acute exacerbation and convalescence in asthmatic children. Ann Allergy Asthma Immunol. 2001; 86:272–276.
Article58. Nanba T, Watanabe M, Inoue N, Iwatani Y. Increases of the Th1/Th2 cell ratio in severe Hashimoto's disease and in the proportion of Th17 cells in intractable Graves' disease. Thyroid. 2009; 19:495–501.
Article59. Falivene J, Ghiglione Y, Laufer N, Socias ME, Holgado MP, Ruiz MJ, Maeto C, Figueroa MI, Giavedoni LD, Cahn P, Salomon H, Sued O, Turk G, Gherardi MM. Th17 and Th17/Treg ratio at early HIV infection associate with protective HIV-specific CD8(+) T-cell responses and disease progression. Sci Rep. 2015; 5:11511.
Article60. Yu ZX, Ji MS, Yan J, Cai Y, Liu J, Yang HF, Li Y, Jin ZC, Zheng JX. The ratio of Th17/Treg cells as a risk indicator in early acute respiratory distress syndrome. Crit Care. 2015; 19:82.
Article61. Ji L, Zhan Y, Hua F, Li F, Zou S, Wang W, Song D, Min Z, Chen H, Cheng Y. The ratio of Treg/Th17 cells correlates with the disease activity of primary immune thrombocytopenia. PLoS One. 2012; 7:e50909.
Article62. Cunnusamy K, Baughman EJ, Franco J, Ortega SB, Sinha S, Chaudhary P, Greenberg BM, Frohman EM, Karandikar NJ. Disease exacerbation of multiple sclerosis is characterized by loss of terminally differentiated autoregulatory CD8+ T cells. Clin Immunol. 2014; 152:115–126.
Article63. Baughman EJ, Mendoza JP, Ortega SB, Ayers CL, Greenberg BM, Frohman EM, Karandikar NJ. Neuroantigen-specific CD8+ regulatory T-cell function is deficient during acute exacerbation of multiple sclerosis. J Autoimmun. 2011; 36:115–124.
Article64. Baumgarth N. The double life of a B-1 cell: self-reactivity selects for protective effector functions. Nat Rev Immunol. 2011; 11:34–46.
Article65. Villoslada P, Baranzini S. Data integration and systems biology approaches for biomarker discovery: challenges and opportunities for multiple sclerosis. J Neuroimmunol. 2012; 248:58–65.
Article66. Buriani A, Fortinguerra S, Carrara M. Clinical perspectives in diagnostic-omics and personalised medicine approach to monitor effectiveness and toxicity of phytocomplexes. In : Pelkonen O, Duez P, Vuorela PM, Vuorela H, editors. Toxicology of herbal products. Springer: 2017. Chapter 16. DOI: 10.1007/978-3-319-43806-1_16.67. Buriani A, Pelkonen O, Fortinguerra S, Carrara M. Systems network pharmaco-toxicology in the study of herbal medicines. In : Pelkonen O, Duez P, Vuorela PM, Vuorela H, editors. Toxicology of herbal products. Springer;2017. Chapter 7. DOI: 10.1007/978-3-319-43806-1_7.68. Buriani A, Fortinguerra S. The systems biology oriented. Holistic vision of personalized medicine and the emerging concept of proactive herbal medicine. J J Intern Medicine. 2015; 1:007.69. Oriss TB, McCarthy SA, Morel BF, Campana MA, Morel PA. Crossregulation between T helper cell (Th)1 and Th2: inhibition of Th2 proliferation by IFN-gamma involves interference with IL-1. J Immunol. 1997; 158:3666–3672.70. Damsker JM, Hansen AM, Caspi RR. Th1 and Th17 cells: adversaries and collaborators. Ann N Y Acad Sci. 2010; 1183:211–221.71. Song K, Coleman RA, Alber C, Ballas ZK, Waldschmidt TJ, Mortari F, LaBrecque DR, Cook RT. TH1 cytokine response of CD57+ T-cell subsets in healthy controls and patients with alcoholic liver disease. Alcohol. 2001; 24:155–167.
Article72. Ekkens MJ, Shedlock DJ, Jung E, Troy A, Pearce EL, Shen H, Pearce EJ. Th1 and Th2 cells help CD8 T-cell responses. Infect Immun. 2007; 75:2291–2296.
Article73. Geering B, Stoeckle C, Conus S, Simon HU. Living and dying for inflammation: neutrophils, eosinophils, basophils. Trends Immunol. 2013; 34:398–409.
Article74. de Visser KE, Eichten A, Coussens LM. Paradoxical roles of the immune system during cancer development. Nat Rev Cancer. 2006; 6:24–37.
Article75. Brombacher F, Kastelein RA, Alber G. Novel IL-12 family members shed light on the orchestration of Th1 responses. Trends Immunol. 2003; 24:207–212.
Article76. Chong WP, van PN, Chen J, Silver PB, Jittayasothorn Y, Mattapallil MJ, Germain RN, Caspi RR. NK-DC crosstalk controls the autopathogenic Th17 response through an innate IFN-gamma-IL-27 axis. J Exp Med. 2015; 212:1739–1752.
Article77. Garlanda C, Dinarello CA, Mantovani A. The interleukin-1 family: back to the future. Immunity. 2013; 39:1003–1018.
Article78. Korn T, Oukka M, Kuchroo V, Bettelli E. Th17 cells: effector T cells with inflammatory properties. Semin Immunol. 2007; 19:362–371.
Article79. Romagnani S. The Th1/Th2 paradigm. Immunol Today. 1997; 18:263–266.
Article80. Janeway JCA, Travers P, Walport M, Shlomchik MJ. Macrophage activation by armed CD4 TH1 cells. In : Charles J, Janeway A, Travers P, Walport M, Shlomchik MJ, editors. Immunobiology, The immune system in health and disease. 5th Edition. New York: Garland science;2001. p. 333–337. Part IV, Chapter 8.81. Basu R, Hatton RD, Weaver CT. The Th17 family: flexibility follows function. Immunol Rev. 2013; 252:89–103.
Article82. de Blic J, Tillie-Leblond I, Tonnel AB, Jaubert F, Scheinmann P, Gosset P. Difficult asthma in children: an analysis of airway inflammation. J Allergy Clin Immunol. 2004; 113:94–100.83. Meyer-Hoffert U, Lezcano-Meza D, Bartels J, Montes-Vizuet AR, Schroder JM, Teran LM. Th2- and to a lesser extent Th1-type cytokines upregulate the production of both CXC (IL-8 and gro-alpha) and CC (RANTES, eotaxin, eotaxin-2, MCP-3 and MCP-4) chemokines in human airway epithelial cells. Int Arch Allergy Immunol. 2003; 131:264–271.
Article84. Min B. Basophils induce Th2 immunity: is this final answer? Virulence. 2010; 1:399–401.85. Knoops L, Louahed J, Renauld JC. IL-9-induced expansion of B-1b cells restores numbers but not function of B-1 lymphocytes in xid mice. J Immunol. 2004; 172:6101–6106.
Article86. Zhu J. T helper 2 (Th2) cell differentiation, type 2 innate lymphoid cell (ILC2) development and regulation of interleukin-4 (IL-4) and IL-13 production. Cytokine. 2015; 75:14–24.
Article87. Wang C, Liu Q, Chen F, Xu W, Zhang C, Xiao W. IL-25 promotes Th2 Immunity responses in asthmatic mice via nuocytes activation. PLoS One. 2016; 11:e0162393.
Article88. Su J, Chen T, Ji XY, Liu C, Yadav PK, Wu R, Yang P, Liu Z. IL-25 downregulates Th1/Th17 immune response in an IL-10-dependent manner in inflammatory bowel disease. Inflamm Bowel Dis. 2013; 19:720–728.
Article89. Tian Y, Zajac AJ. IL-21 and T Cell Differentiation: Consider the Context. Trends Immunol. 2016; 37:557–568.
Article90. Leonard WJ, Wan CK. IL-21 Signaling in Immunity. F1000Res. 2016; 5.
Article91. Tosolini M, Kirilovsky A, Mlecnik B, Fredriksen T, Mauger S, Bindea G, Berger A, Bruneval P, Fridman WH, Pages F, Galon J. Clinical impact of different classes of infiltrating T cytotoxic and helper cells (Th1, th2, treg, th17) in patients with colorectal cancer. Cancer Res. 2011; 71:1263–1271.
Article92. Saito S, Nakashima A, Shima T, Ito M. Th1/Th2/Th17 and regulatory T-cell paradigm in pregnancy. Am J Reprod Immunol. 2010; 63:601–610.
Article93. van den Berg WB, McInnes IB. Th17 cells and IL-17 a--focus on immunopathogenesis and immunotherapeutics. Semin Arthritis Rheum. 2013; 43:158–170.
Article94. Busman-Sahay KO, Walrath T, Huber S, O'Connor W Jr. Cytokine crowdsourcing: multicellular production of TH17-associated cytokines. J Leukoc Biol. 2015; 97:499–510.
Article95. Gharibi T, Majidi J, Kazemi T, Dehghanzadeh R, Motallebnezhad M, Babaloo Z. Biological effects of IL-21 on different immune cells and its role in autoimmune diseases. Immunobiology. 2016; 221:357–367.
Article96. Ju B, Li D, Ji X, Liu J, Peng H, Wang S, Liu Y, Hao Y, Yee C, Liang H, Shao Y. Interleukin-21 administration leads to enhanced antigen-specific T cell responses and natural killer cells in HIV-1 vaccinated mice. Cell Immunol. 2016; 303:55–65.
Article97. Cope A, Le FG, Cardone J, Kemper C. The Th1 life cycle: molecular control of IFN-gamma to IL-10 switching. Trends Immunol. 2011; 32:278–286.98. Pedroza-Pacheco I, Madrigal A, Saudemont A. Interaction between natural killer cells and regulatory T cells: perspectives for immunotherapy. Cell Mol Immunol. 2013; 10:222–229.
Article99. Taams LS, van Amelsfort JM, Tiemessen MM, Jacobs KM, de Jong EC, Akbar AN, Bijlsma JW, Lafeber FP. Modulation of monocyte/macrophage function by human CD4+CD25+ regulatory T cells. Hum Immunol. 2005; 66:222–230.
Article100. Lewkowicz N, Klink M, Mycko MP, Lewkowicz P. Neutrophil--CD4+CD25+ T regulatory cell interactions: a possible new mechanism of infectious tolerance. Immunobiology. 2013; 218:455–464.
Article101. McNally A, Hill GR, Sparwasser T, Thomas R, Steptoe RJ. CD4+CD25+ regulatory T cells control CD8+ T-cell effector differentiation by modulating IL-2 homeostasis. Proc Natl Acad Sci U S A. 2011; 108:7529–7534.
Article102. Rother N, van der Vlag J. Disturbed T cell signaling and altered Th17 and Regulatory T cell subsets in the pathogenesis of systemic lupus erythematosus. Front Immunol. 2015; 6:610.
Article103. Diehl S, Rincon M. The two faces of IL-6 on Th1/Th2 differentiation. Mol Immunol. 2002; 39:531–536.
Article104. Newcomb DC, Boswell MG, Zhou W, Huckabee MM, Goleniewska K, Sevin CM, Hershey GK, Kolls JK, Peebles RS Jr. Human TH17 cells express a functional IL-13 receptor and IL-13 attenuates IL-17A production. J Allergy Clin Immunol. 2011; 127:1006–1013.e1-4.
Article105. Kimura A, Kishimoto T. IL-6: regulator of Treg/Th17 balance. Eur J Immunol. 2010; 40:1830–1835.
Article106. Patera AC, Pesnicak L, Bertin J, Cohen JI. Interleukin 17 modulates the immune response to vaccinia virus infection. Virology. 2002; 299:56–63.
Article107. Zhong X, Gao W, Degauque N, Bai C, Lu Y, Kenny J, Oukka M, Strom TB, Rothstein TL. Reciprocal generation of Th1/Th17 and T(reg) cells by B1 and B2 B cells. Eur J Immunol. 2007; 37:2400–2404.
Article108. Wang Y, Rothstein TL. Induction of Th17 cell differentiation by B-1 cells. Front Immunol. 2012; 3:281.
Article109. Kotake S, Nanke Y, Yago T, Kawamoto M, Kobashigawa T, Yamanaka H. Elevated ratio of Th17 cell-derived Th1 cells (CD161(+)Th1 cells) to CD161(+)Th17 cells in peripheral blood of early-onset rheumatoid arthritis patients. Biomed Res Int. 2016; 2016:4186027.110. Li J, Dong X, Zhao L, Wang X, Wang Y, Yang X, Wang H, Zhao W. Natural killer cells regulate Th1/Treg and Th17/Treg balance in chlamydial lung infection. J Cell Mol Med. 2016; 20:1339–1351.
Article111. Hsing CH, Hsu CC, Chen WY, Chang LY, Hwang JC, Chang MS. Expression of IL-19 correlates with Th2 cytokines in uraemic patients. Nephrol Dial Transplant. 2007; 22:2230–2238.
Article112. Liao SC, Cheng YC, Wang YC, Wang CW, Yang SM, Yu CK, Shieh CC, Ceng KC, Lee MF, Chiang SR, Shieh JM, Chang MS. IL-19 induced Th2 cytokines and was up-regulated in asthma patients. J Immunol. 2004; 173:6712–6718.
Article113. Allen JE, Sutherland TE, Ruckerl D. IL-17 and neutrophils: unexpected players in the type 2 immune response. Curr Opin Immunol. 2015; 34:99–106.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Influence of Halothane Anesthesia on the T - lymphocyte Subpopulations
- Clinical Significance of Lymphocyte Subpopulations in Peripheral Blood from Gastric Cancer Patients
- Evaluation of In-house Lymphocyte Panel of 72 Wells for the Identification of HLA Antibody Specificity
- Flow Cytometric Analysis of Lymphocyte Subpopulations of Cattle Infected with Bovine Leukemia Virus
- Postirradiation Changes of White Blood Cellsand Lymphocyte Subpopulations in Cancer Patients