Ann Clin Microbiol.
2016 Sep;19(3):77-81. 10.5145/ACM.2016.19.3.77.
Rapid Diagnosis of Mycobacterium abscessus Bacteremia Using Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry
- Affiliations
-
- 1Department of Laboratory Medicine, Chonnam National University Medical School, Gwangju, Korea. shinjh@chonnam.ac.kr
- KMID: 2353044
- DOI: http://doi.org/10.5145/ACM.2016.19.3.77
Abstract
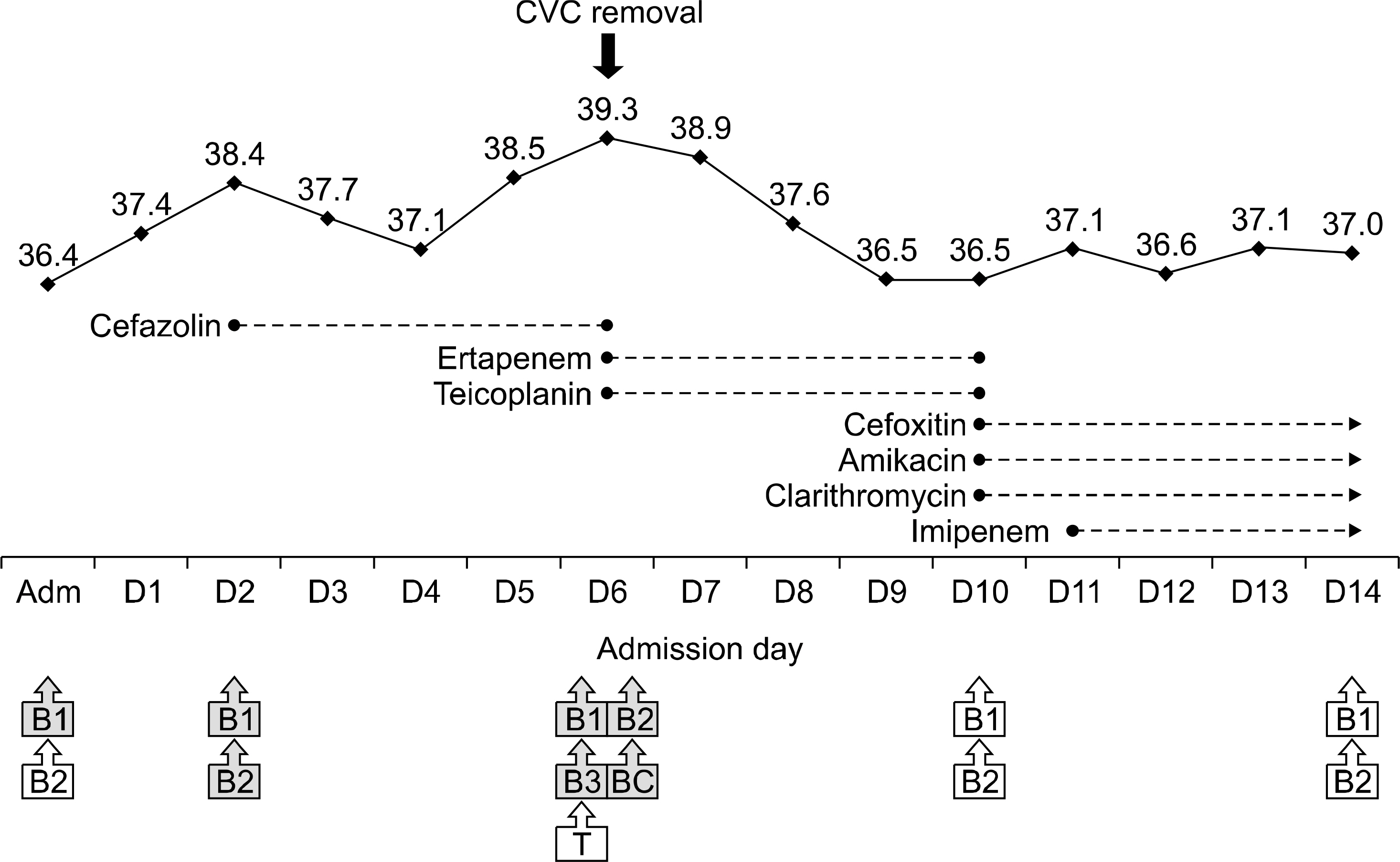
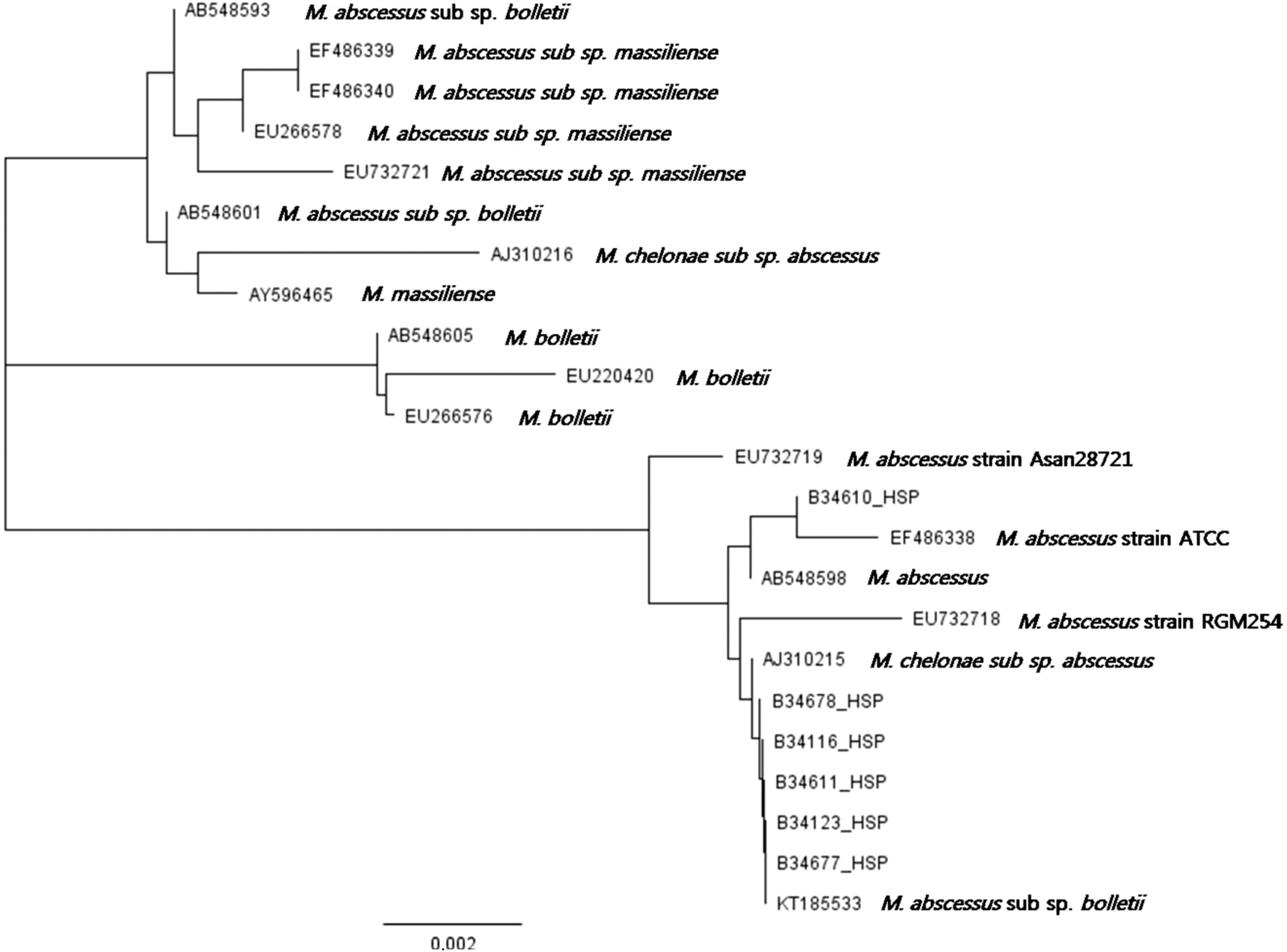
- Mycobacterium abscessus was isolated from cultures of seven blood samples from a 64-year-old diabetic female who was admitted due to steroid-unresponsive adrenal insufficiency. The isolates were difficult to identify using the conventional commercial systems, VITEK 2 (bioMérieux, France) or MicroScan (Siemens Healthcare Diagnostics, USA), but were rapidly identified as M. abscessus by a matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS)-based Bruker Biotyper system (Bruker Daltonics, USA). Identification of M. abscessus was confirmed by a reverse hybridizationbased assay (Genotype Mycobacterium CM/AS 12, Hain Lifescience) and direct sequencing of a heatshock protein gene. After removal of her central venous catheter, the patient was successfully treated with a combination therapy comprising clarithromycin, amikacin, cefoxitin, and imipenem. Our findings demonstrate that MALDI-TOF MS can facilitate rapid and accurate identification of M. abscessus from blood cultures, which enables prompt administration of appropriate therapy following catheter removal.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Wagner D and Young LS. Nontuberculous mycobacterial infections: a clinical review. Infection. 2004; 32:257–70.
Article2. Brown-Elliott BA and Wallace RJ Jr. Clinical and taxonomic status of pathogenic nonpigmented or late-pigmenting rapidly growing mycobacteria. Clin Microbiol Rev. 2002; 15:716–46.
Article3. Lee MR, Sheng WH, Hung CC, Yu CJ, Lee LN, Hsueh PR. Mycobacterium abscessus complex infections in humans. Emerg Infect Dis. 2015; 21:1638–46.4. Lee MR, Ko JC, Liang SK, Lee SW, Yen DH, Hsueh PR. Bacteraemia caused by Mycobacterium abscessus subsp. abscessus and M. abscessus subsp. bolletii: clinical features and susceptibilities of the isolates. Int J Antimicrob Agents. 2014; 43:438–41.
Article5. El Helou G, Viola GM, Hachem R, Han XY, Raad II. Rapidly growing mycobacterial bloodstream infections. Lancet Infect Dis. 2013; 13:166–74.
Article6. Kim HY, Kook Y, Yun YJ, Park CG, Lee NY, Shim TS, et al. Proportions of Mycobacterium massiliense and Mycobacterium bolletii strains among Korean Mycobacterium chelonae-Mycobac-terium abscessus group isolates. J Clin Microbiol. 2008; 46:3384–90.7. Mediavilla-Gradolph MC, De Toro-Peinado I, Bermúdez-Ruiz MP, García-Martínez Mde L, Ortega-Torres M, Montiel Quezel-Guerraz N, et al. Use of MALDI-TOF MS for identification of nontuberculous Mycobacterium species isolated from clinical specimens. Biomed Res Int. 2015; 2015:854078.8. Mareković I, Bošnjak Z, Jakopović M, Boras Z, Janković M, Popović-Grle S. Evaluation of matrix-assisted laser desorption/ionization time-of-flight mass spectrometry in identification of nontuberculous mycobacteria. Chemotherapy. 2016; 61:167–70.
Article9. Telenti A, Marchesi F, Balz M, Bally F, Böttger EC, Bodmer T. Rapid identification of mycobacteria to the species level by polymerase chain reaction and restriction enzyme analysis. J Clin Microbiol. 1993; 31:175–8.
Article10. CLSI. Susceptibility testing of mycobacteria, nocardiae and other aerobic actinomycetes: approved standard-second edition. CLSI document M24-A2. Wayne, PA: Clinical and Laboratory Standards Institute;2011.11. Teng SH, Chen CM, Lee MR, Lee TF, Chien KY, Teng LJ, et al. Matrix-assisted laser desorption ionization-time of flight mass spectrometry can accurately differentiate between Mycobacterium masilliense (M. abscessus subspecies bolletti) and M. abscessus (sensu stricto). J Clin Microbiol. 2013; 51:3113–6.12. Panagea T, Pincus DH, Grogono D, Jones M, Bryant J, Parkhill J, et al. Mycobacterium abscessus complex identification with matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol. 2015; 53:2355–8.
Article13. De Groote MA. Disease resulting from contaminated equipment and invasive procedures. Pedley S, Bartram J, Rees G, Dufour A, Cotruvo J, editors. Pathogenic Mycobacteria in Water: A Guide to Public Health Consequences, Monitoring and Management. Padstow, Cornwall: TJ International;2004. p. 131–42.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Two Cases of Campylobacter jejuni Bacteremia from Patients with Diarrhea
- Application of Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Imaging Mass Spectrometry (MALDI-TOF IMS) for Premalignant Gastrointestinal Lesions
- MALDI-TOF-MS Fingerprinting Provides Evidence of Urosepsis caused by Aerococcus urinae
- Reliability of Acinetobacter baumannii Identification with Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry
- Mass spectrometry based cellular phosphoinositides profiling and phospholipid analysis: A brief review