Korean J Ophthalmol.
2014 Dec;28(6):436-443. 10.3341/kjo.2014.28.6.436.
Prevalence and Spatial Concordance of Visual Field Deterioration in Fellow Eyes of Glaucoma Patients
- Affiliations
-
- 1Siloam Eye Hospital, Seoul, Korea. eyejune.lee@gmail.com
- 2The Jules Stein Eye Institute, David Geffen School of Medicine at UCLA, Los Angeles, CA, USA.
- KMID: 2345819
- DOI: http://doi.org/10.3341/kjo.2014.28.6.436
Abstract
- PURPOSE
To examine the prevalence of visual field deterioration in contralateral eyes of patients with worsening open-angle glaucoma and to evaluate the spatial concordance of visual field deterioration between both eyes.
METHODS
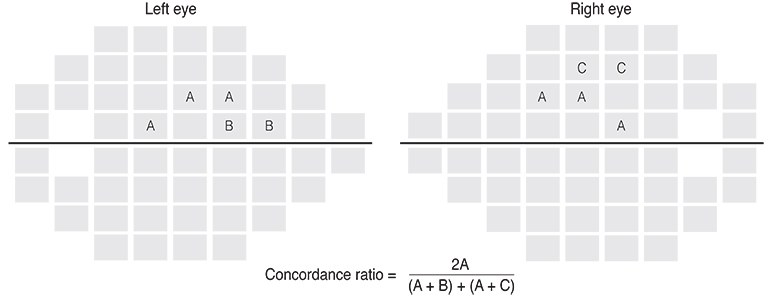
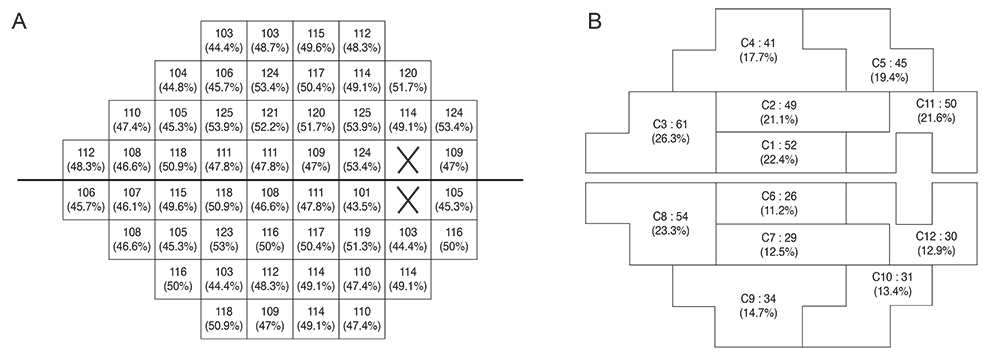
One hundred sixteen open-angle glaucoma patients who underwent 8 or more visual field examinations over > or =6 years of follow-up were included. The rates of the fast and slow components of visual field decay for each of 52 visual field test locations were calculated with point-wise exponential regression analysis. The spatial concordance of visual field deterioration in contralateral eyes was evaluated with a concordance ratio (calculated as the number of overlapping locations divided by the total number of deteriorating locations) and by comparing the rate of decay in corresponding modified glaucoma hemifield test clusters.
RESULTS
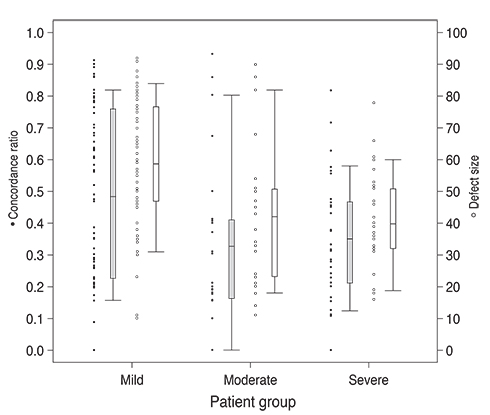
The average visual field mean deviation (+/-standard deviation [SD]) was -8.5 (+/-6.4) dB and the mean (+/-SD) follow-up time was 9.0 (+/-1.6) years. Sixty-three patients had mild damage, 23 had moderate damage, and 30 had severe damage. The mean concordance ratio (+/-SD) was 0.46 (+/-0.32) for the mild group, 0.33 (+/-0.27) for the moderate group, and 0.35 (+/-0.21) for the severe group. Thirty-one patients (27%) had deterioration in concordant locations (p < 0.05). Visual field deterioration was greater in the superior hemifield than the inferior hemifield (p < 0.05) when evaluated with both the concordance ratio and modified glaucoma hemifield test cluster analysis methods.
CONCLUSIONS
There is only fair spatial concordance with regard to visual field deterioration between the both eyes of an individual. We conclude that testing algorithms taking advantage of inter-eye spatial concordance would not be particularly advantageous in the early detection of glaucomatous deterioration.
MeSH Terms
Figure
Reference
-
1. Lee AJ, Wang JJ, Rochtchina E, et al. Patterns of glaucomatous visual field defects in an older population: the Blue Mountains Eye Study. Clin Experiment Ophthalmol. 2003; 31:331–335.2. Boden C, Hoffmann EM, Medeiros FA, et al. Intereye concordance in locations of visual field defects in primary open-angle glaucoma: diagnostic innovations in glaucoma study. Ophthalmology. 2006; 113:918–923.3. Chen PP, Bhandari A. Fellow eye prognosis in patients with severe visual field loss in 1 eye from chronic open-angle glaucoma. Arch Ophthalmol. 2000; 118:473–478.4. Chen PP. Correlation of visual field progression between eyes in patients with open-angle glaucoma. Ophthalmology. 2002; 109:2093–2099.5. Asman P, Heijl A. Glaucoma hemifield test: automated visual field evaluation. Arch Ophthalmol. 1992; 110:812–819.6. Ederer F, Gaasterland DE, Sullivan EK. AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS). 1. Study design and methods and baseline characteristics of study patients. Control Clin Trials. 1994; 15:299–325.7. AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS). 4. Comparison of treatment outcomes within race: seven-year results. Ophthalmology. 1998; 105:1146–1164.8. Bengtsson B, Heijl A. False-negative responses in glaucoma perimetry: indicators of patient performance or test reliability? Invest Ophthalmol Vis Sci. 2000; 41:2201–2204.9. Caprioli J, Mock D, Bitrian E, et al. A method to measure and predict rates of regional visual field decay in glaucoma. Invest Ophthalmol Vis Sci. 2011; 52:4765–4773.10. Thissen D, Steinberg L, Kuang D. Quick and easy implementation of the Benjamini-Hochberg procedure for controlling the false positive rate in multiple comparisons. J Educ Behav Stat. 2002; 27:77–83.11. Team RC. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing;2010.12. Heijl A, Lindgren G, Olsson J, Asman P. Visual field interpretation with empiric probability maps. Arch Ophthalmol. 1989; 107:204–208.13. Heijl A, Leske MC, Bengtsson B, et al. Measuring visual field progression in the Early Manifest Glaucoma Trial. Acta Ophthalmol Scand. 2003; 81:286–293.14. Smith SD, Katz J, Quigley HA. Analysis of progressive change in automated visual fields in glaucoma. Invest Ophthalmol Vis Sci. 1996; 37:1419–1428.15. Nouri-Mahdavi K, Brigatti L, Weitzman M, Caprioli J. Comparison of methods to detect visual field progression in glaucoma. Ophthalmology. 1997; 104:1228–1236.16. Nouri-Mahdavi K, Caprioli J, Coleman AL, et al. Pointwise linear regression for evaluation of visual field outcomes and comparison with the advanced glaucoma intervention study methods. Arch Ophthalmol. 2005; 123:193–199.17. Katz J, Gilbert D, Quigley HA, Sommer A. Estimating progression of visual field loss in glaucoma. Ophthalmology. 1997; 104:1017–1025.18. Chauhan BC, Drance SM, Douglas GR. The use of visual field indices in detecting changes in the visual field in glaucoma. Invest Ophthalmol Vis Sci. 1990; 31:512–520.19. Wild JM, Hussey MK, Flanagan JG, Trope GE. Pointwise topographical and longitudinal modeling of the visual field in glaucoma. Invest Ophthalmol Vis Sci. 1993; 34:1907–1916.20. Heijl A, Lindgren A, Lindgren G. Test-retest variability in glaucomatous visual fields. Am J Ophthalmol. 1989; 108:130–135.21. Schulzer M. Errors in the diagnosis of visual field progression in normal-tension glaucoma. Ophthalmology. 1994; 101:1589–1594.22. Kim J, Dally LG, Ederer F, et al. The Advanced Glaucoma Intervention Study (AGIS). 14. Distinguishing progression of glaucoma from visual field fluctuations. Ophthalmology. 2004; 111:2109–2116.23. Wall M, Woodward KR, Doyle CK, Artes PH. Repeatability of automated perimetry: a comparison between standard automated perimetry with stimulus size III and V, matrix, and motion perimetry. Invest Ophthalmol Vis Sci. 2009; 50:974–979.24. Azarbod P, Mock D, Bitrian E, et al. Validation of pointwise exponential regression to measure the decay rates of glaucomatous visual fields. Invest Ophthalmol Vis Sci. 2012; 53:5403–5409.25. Spry PG, Sparrow JM, Diamond JP, Harris HS. Risk factors for progressive visual field loss in primary open angle glaucoma. Eye. 2005; 19:643–651.26. Pascual JP, Schiefer U, Paetzold J, et al. Spatial characteristics of visual field progression determined by Monte Carlo simulation: diagnostic innovations in glaucoma study. Invest Ophthalmol Vis Sci. 2007; 48:1642–1650.27. Boden C, Blumenthal EZ, Pascual J, et al. Patterns of glaucomatous visual field progression identified by three progression criteria. Am J Ophthalmol. 2004; 138:1029–1036.28. Mikelberg FS, Drance SM. The mode of progression of visual field defects in glaucoma. Am J Ophthalmol. 1984; 98:443–445.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Analysis of Glaucoma Patients Undergone Filtration Surgery
- The Differences of Visual Field Defects in Three Types of Primary Glaucoma
- Visual Field Changes after Internal Limiting Membrane Peeling in Glaucoma Patients with Epiretinal Membrane
- Study of Contralateral Eye of Normal Tension Glaucoma and a Unilateral Visual Field Defect
- Visual Field Progression in Patients with Primary Open Angle Glaucoma, Normal Tension Glaucoma, and Primary Angle Closure Glaucoma