J Vet Sci.
2015 Sep;16(3):385-388. 10.4142/jvs.2015.16.3.385.
Anaplasma sp. and hemoplasma infection in leopard cats (Prionailurus bengalensis euptilurus) from Korea
- Affiliations
-
- 1Department of Veterinary Pathology, College of Veterinary Medicine, University of Georgia, Athens, GA30605, USA.
- 2College of Veterinary Medicine, Seoul National University, Seoul 151-742, Korea. jdchun@snu.ac.kr
- KMID: 2344313
- DOI: http://doi.org/10.4142/jvs.2015.16.3.385
Abstract
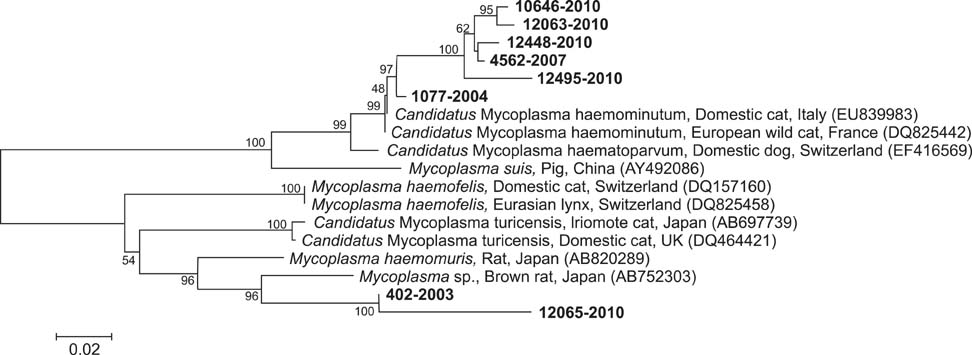
- This study examined the occurrence of Anaplasma spp. and hemoplasma infection in leopard cats, Prionailurus bengalensis euptilurus, in Korea. Twenty-nine biological samples were tested by molecular analysis. Two (6.9%) and eight (27.6%) tested specimens were positive for Anaplasma bovis and hemoplasma infection, respectively. Based on our results, Anaplasma/Ehrlichia spp. and hemoplasma are regularly infecting leopard cat populations of Korea. Considering their endangered status, regular monitoring of infection by arthropod-borne pathogens known to cause clinical symptoms in feline hosts such as Anaplasma/Ehrlichia spp. and hemoplasma would be crucial as part of ongoing conservation efforts.
MeSH Terms
-
Anaplasma/*isolation & purification
Anaplasmosis/*epidemiology/microbiology
Animals
DNA, Bacterial/genetics
*Felidae
Molecular Sequence Data
Mycoplasma/*isolation & purification
Mycoplasma Infections/epidemiology/microbiology/*veterinary
Phylogeny
Polymerase Chain Reaction/veterinary
RNA, Ribosomal, 16S/genetics
Republic of Korea/epidemiology
Sequence Analysis, DNA/veterinary
DNA, Bacterial
RNA, Ribosomal, 16S
Figure
Reference
-
1. Choi TY, Kwon HS, Woo DG, Park CH. Habitat selection and management of the leopard cat (Prionailurus bengalensis) in a rural area of Korea. Korean J Environ Ecol. 2012; 26:322–332.2. Criado-Fornelio A, Martinez-Marcos A, Buling-Saraña A, Barba-Carretero JC. Presence of Mycoplasma haemofelis, Mycoplasma haemominutum and piroplasmids in cats from southern Europe: a molecular study. Vet Microbiol. 2003; 93:307–317.
Article3. Duarte A, Marques V, Correia JHD, Neto I, Bráz BS, Rodrigues C, Martins T, Rosado R, Ferreira JP, Santos-Reis M, Tavares L. Molecular detection of haemotropic Mycoplasma species in urban and rural cats from Portugal. J Feline Med Surg. 2015; 17:516–522.
Article4. Eddlestone SM, Diniz PPVP, Neer TM, Gaunt SD, Corstvet R, Cho D, Hosgood G, Hegarty B, Breitschwerdt EB. Doxycycline clearance of experimentally induced chronic Ehrlichia canis infection in dogs. J Vet Intern Med. 2007; 21:1237–1242.
Article5. Hirata M, Tateno M, Sakuma M, Nakanishi N, Izawa M, Asari Y, Okamura M, Shimokawa Miyama T, Setoguchi A, Endo Y. An epidemiological survey of hemoplasma infection in Iriomote cats (Prionailurus bengalensis iriomotensis). J Vet Med Sci. 2012; 74:1531–1537.
Article6. Jilintai , Seino N, Hayakawa D, Suzuki M, Hata H, Kondo S, Matsumoto K, Yakoyama N, Inokuma H. Molecular survey for Anaplasma bovis and Anaplasma phagocytophilum infection in cattle in a pastureland where sika deer appear in Hokkaido, Japan. Jpn J Infect Dis. 2009; 62:73–75.7. Kim HC, Hang SH, Chong ST, Klein TA, Choi CY, Nam HY, Chae HY, Lee H, Ko S, Kang JG, Chae JS. Ticks collected from selected mammalian hosts surveyed in the Republic of Korea during 2008-2009. Korean J Parasitol. 2011; 49:331–335.
Article8. Kim CM, Yi YH, Yu DH, Lee MJ, Cho MR, Desai AR, Shringi S, Klein TA, Kim HC, Song JW, Baek LJ, Chong ST, O'Guinn ML, Lee JS, Lee IY, Park JH, Foley J, Chae JS. Tick-borne rickettsial pathogens in ticks and small mammals in Korea. Appl Environ Microbiol. 2006; 72:5766–5776.
Article9. Kang JG, Ko S, Kim YJ, Yang HJ, Lee H, Shin NS, Choi KS, Chae JS. New genetic variants of Anaplasma phagocytophilum and Anaplasma bovis from Korean water deer (Hydropotes inermis argyropus). Vector Borne Zoonotic Dis. 2011; 11:929–938.
Article10. Meli ML, Cattori V, Martínez F, López G, Vargas A, Simón MA, Zorrilla I, Muñoz A, Palomares F, López-Bao JV, Pastor J, Tandon R, Willi B, Hofmann-Lehmann R, Lutz H. Feline leukemia virus and other pathogens as important threat to the survival of the critically endangered Iberian lynx (Lynx pardinus). PLoS One. 2009; 4:e4744.11. Spada E, Proverbio D, Galluzzo P, Della Pepa A, Bagnagatti De Giorgi G, Perego R, Ferro E. Prevalence of haemoplasma infections in stray cats in northern Italy. ISRN Microbiol. 2014; 2014:298352.
Article12. Suzán G, Ceballos G. The role of feral mammals on wildlife infectious disease prevalence in two nature reserves within Mexico City limits. J Zoo Wildl Med. 2005; 36:479–484.
Article13. Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: Molecular Evolutionary Genetics Analysis using Maximum Likelihood, Evolutionary Distance, and Maximum Parsimony Methods. Mol Biol Evol. 2011; 28:2731–2739.
Article14. Tateno M, Nishio T, Sakuma M, Nakanishi N, Izawa M, Asari Y, Okamura M, Maruyama S, Miyama TS, Setoguchi A, Endo Y. Molecular epidemiologic survey of Bartonella, Ehrlichia, and Anaplasma infections in Japanese Iriomote and Tsushima leopard cats. J Wildl Dis. 2013; 49:646–652.
Article15. Vanak AT, Gompper ME. Interference competition at the landscape level: the effect of free-ranging dogs on a native mesocarnivore. J Appl Ecol. 2010; 47:1225–1232.
Article16. Yu DH, Kim HW, Desai AR, Han IA, Li YH, Lee MJ, Kim IS, Chae JS, Park J. Molecular detection of feline hemoplasmas in feral cats in Korea. J Vet Med Sci. 2007; 69:1299–1301.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Fibrinous pleuritis associated with Streptococcus canis in a leopard cat (Prionailurus bengalensis euptilurus)
- A Clonorchiasis Case of a Leopard Cat, Prionailurus bengalensis euptilurus, Diagnosed by Ultrasonography and Egg Detection in Republic of Korea
- Diagnostic laparoscopy in a leopard cat (Prionailurus bengalensis) with intercostal abdominal hernia and hepatic lipidosis
- Infections by pathogens with different transmission modes in feral cats from urban and rural areas of Korea
- Detection of Anaplasma sp. in Korean Native Goats (Capra aegagrus hircus) on Jeju Island, Korea