Ann Surg Treat Res.
2016 Jul;91(1):37-44. 10.4174/astr.2016.91.1.37.
Ongoing higher infection rate in ABO-incompatible kidney transplant recipient: is it a serious problem? A single-center experience
- Affiliations
-
- 1Division of Hepato-Biliary-Pancreatic Surgery and Transplantation, Department of Surgery, Pusan National University Yangsan Hospital, Pusan National University School of Medicine, Yangsan, Korea.
- 2Research Institute for Convergence of Biomedical Science and Technology, Pusan National University Yangsan Hospital, Yangsan, Korea.
- 3Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. djhan@amc.seoul.kr
- KMID: 2327415
- DOI: http://doi.org/10.4174/astr.2016.91.1.37
Abstract
- PURPOSE
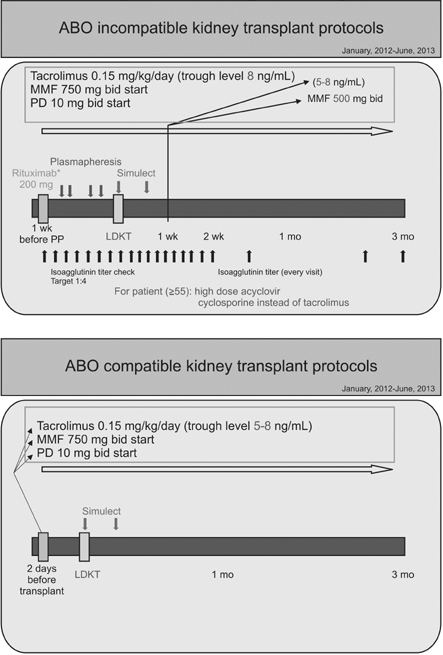
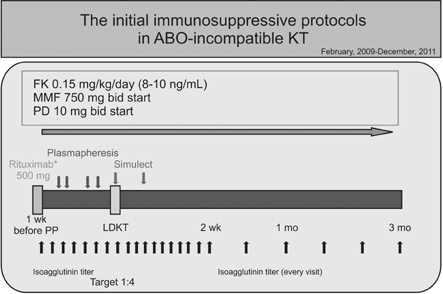
Additional clinical experience and knowledge regarding the barrier to transplantation of ABO blood type incompatibility could reduce the higher rate of infectious complications in ABO-incompatible kidney transplantation.
METHODS
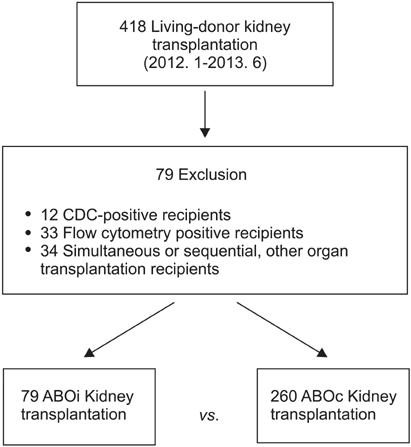
A total of 79 ABO-incompatible kidney transplantation (ABOiKT) patients were compared with 260 ABO-compatible kidney transplantation (ABOcKT) patients for basic clinical characteristics, infectious complications, rejection episodes, and graft survival.
RESULTS
There were no significant differences in baseline characteristics, rejection rates, or graft survival between the ABOiKT and ABOcKT patients. No significant difference in the infection rate was shown for cytomegalovirus (26.6% vs. 30.0%; P = 0.672), BK virus (19.0% vs. 21.5%; P = 0.752), herpes disease (10.1% vs. 5.0%; P = 0.082), pneumonia (5.3% vs. 3.8%; P = 0.746), or urinary tract infection (8.9% vs. 10.0%; P > 0.999). Female sex (hazard ratio [HR], 2.20; P = 0.003), advanced age (≥60 years) (HR, 2.5; P = 0.019), history of rejection episodes (HR, 2.28; P = 0.016), and history of surgical complications (HR, 4.64; P = 0.018) were significant risk factors for infection. ABO incompatibility demonstrated a tendency toward higher infection risk without statistical significance (HR, 1.74; P = 0.056).
CONCLUSION
In spite of immunosuppressant protocol modification, the rate of infectious complications following ABOiKT is still higher than with ABOcKT when a modified desensitization protocol is used. However, this was not sufficient to avoid ABOiKT.
MeSH Terms
Figure
Reference
-
1. Choi BH, Cho HK, Jung JH, Choi JY, Shin S, Kim YH, et al. How to reduce lethal infectious complications in ABO-incompatible kidney transplantation. Transplant Proc. 2015; 47:653–659.2. Habicht A, Broker V, Blume C, Lorenzen J, Schiffer M, Richter N, et al. Increase of infectious complications in ABO-incompatible kidney transplant recipients: a single centre experience. Nephrol Dial Transplant. 2011; 26:4124–4131.3. Lentine KL, Axelrod D, Klein C, Simpkins C, Xiao H, Schnitzler MA, et al. Early clinical complications after ABO-incompatible live-donor kidney transplantation: a national study of Medicare-insured recipients. Transplantation. 2014; 98:54–65.4. Hwang JK, Kim YK, Kim JM, Chung BH, Choi BS, Yang CW, et al. Comparative analysis of ABO-incompatible living donor kidney transplantation with ABO-compatible grafts: a single-center experience in Korea. Transplant Proc. 2013; 45:2931–2936.5. Shirakawa H, Ishida H, Shimizu T, Omoto K, Iida S, Toki D, et al. The low dose of rituximab in ABO-incompatible kidney transplantation without a splenectomy: a single-center experience. Clin Transplant. 2011; 25:878–884.6. Hatakeyama S, Fujita T, Murakami R, Suzuki Y, Sugiyama N, Yamamoto H, et al. Outcome comparison of ABO-incompatible kidney transplantation with low-dose rituximab and ABO-compatible kidney transplantation: a single-center experience. Transplant Proc. 2014; 46:445–448.7. Asberg A, Humar A, Rollag H, Jardine AG, Mouas H, Pescovitz MD, et al. Oral valganciclovir is noninferior to intravenous ganciclovir for the treatment of cytomegalovirus disease in solid organ transplant recipients. Am J Transplant. 2007; 7:2106–2113.8. Bohl DL, Brennan DC. BK virus nephropathy and kidney transplantation. Clin J Am Soc Nephrol. 2007; 2:Suppl 1. S36–S46.9. Singh R, Geerlings SE, Bemelman FJ. Asymptomatic bacteriuria and urinary tract infections among renal allograft recipients. Curr Opin Infect Dis. 2015; 28:112–116.10. Fiorante S, Lopez-Medrano F, Lizasoain M, Lalueza A, Juan RS, Andres A, et al. Systematic screening and treatment of asymptomatic bacteriuria in renal transplant recipients. Kidney Int. 2010; 78:774–781.11. Lee YM, Kim YH, Han DJ, Park SK, Park JS, Sung H, et al. Cytomegalovirus infection after acute rejection therapy in seropositive kidney transplant recipients. Transpl Infect Dis. 2014; 16:397–402.12. Haas M, Sis B, Racusen LC, Solez K, Glotz D, Colvin RB, et al. Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant. 2014; 14:272–283.13. Fuchinoue S, Ishii Y, Sawada T, Murakami T, Iwadoh K, Sannomiya A, et al. The 5-year outcome of ABO-incompatible kidney transplantation with rituximab induction. Transplantation. 2011; 91:853–857.14. Muramatsu M, Gonzalez HD, Cacciola R, Aikawa A, Yaqoob MM, Puliatti C. ABO incompatible renal transplants: Good or bad? World J Transplant. 2014; 4:18–29.15. Opelz G, Morath C, Susal C, Tran TH, Zeier M, Dohler B. Three-year outcomes following 1,420 ABO-incompatible living-donor kidney transplants performed after ABO antibody reduction: results from 101 centers. Transplantation. 2015; 99:400–404.16. de Weerd AE, van Agteren M, Leebeek FW, Ijzermans JN, Weimar W, Betjes MG. ABO-incompatible kidney transplant recipients have a higher bleeding risk after antigen-specific immunoadsorption. Transpl Int. 2015; 28:25–33.17. Asberg A, Jardine AG, Bignamini AA, Rollag H, Pescovitz MD, Gahlemann CC, et al. Effects of the intensity of immunosuppressive therapy on outcome of treatment for CMV disease in organ transplant recipients. Am J Transplant. 2010; 10:1881–1888.18. Bataille S, Moal V, Gaudart J, Indreies M, Purgus R, Dussol B, et al. Cytomegalovirus risk factors in renal transplantation with modern immunosuppression. Transpl Infect Dis. 2010; 12:480–488.19. Brennan DC, Legendre C, Patel D, Mange K, Wiland A, McCague K, et al. Cytomegalovirus incidence between everolimus versus mycophenolate in de novo renal transplants: pooled analysis of three clinical trials. Am J Transplant. 2011; 11:2453–2462.20. Ko GB, Kim T, Kim SH, Choi SH, Kim YS, Woo JH, et al. Increased incidence of herpes zoster in the setting of cytomegalovirus preemptive therapy after kidney transplantation. Transpl Infect Dis. 2013; 15:416–423.21. Lee JR, Bang H, Dadhania D, Hartono C, Aull MJ, Satlin M, et al. Independent risk factors for urinary tract infection and for subsequent bacteremia or acute cellular rejection: a single-center report of 1166 kidney allograft recipients. Transplantation. 2013; 96:732–738.22. Karim A, Farrugia D, Cheshire J, Mahboob S, Begaj I, Ray D, et al. Recipient age and risk for mortality after kidney transplantation in England. Transplantation. 2014; 97:832–838.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- ABO incompatibility is a risk factor for cytomegalovirus infection with living donor liver transplantation
- ABO-Incompatible Kidney Transplantation
- ABO-Incompatible Living Donor Liver Transplantation
- Case of ABO-Incompatible Living Donor Kidney Transplantation without Blood Products in a Jehovah's Witness
- Analysis of the Results of ABO-Incompatible Kidney Transplantation: In Comparison with ABO-Compatible Kidney Transplantation