J Korean Surg Soc.
2012 May;82(5):281-287.
Clinical features and prognostic factors in papillary thyroid microcarcinoma depends on age
- Affiliations
-
- 1Department of Surgery, Gyeongsang National University Hospital, Jinju, Korea. drjcy@hanmail.net
Abstract
- PURPOSE
Clinical outcomes of papillary thyroid microcarcinoma (PTMC) vary. In general, age at diagnosis is an independent prognostic factor in conventional papillary thyroid carcinoma. However, it is unclear in patients of PTMC. The purpose of this study was to identify clinicopathologic features and prognostic factors of PTMC according to patients' age.
METHODS
Five hundred twenty-seven patients who received thyroid surgery and diagnosed as having PTC between January 2001 and December 2009 were included. The clinical data were retrospectively analyzed.
RESULTS
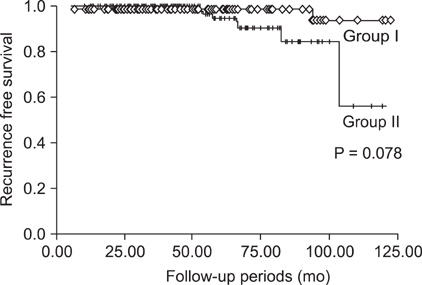
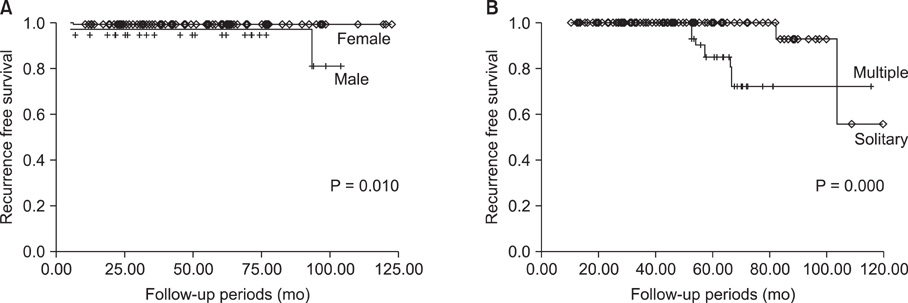
We divided the patients into two groups; group I who were younger than 45 years, and group II who were 45 years old or older. The mean tumor size and incidences of neck lymph nodes involvement of group I was larger than group II. In group II, however, there were more patients who had multiple cancer foci and were body mass index > or = 25 kg/m2. The overall incidence of recurrent disease was 3.2%. The incidence of recurrence was higher in group II (2.0% vs. 4.0%), without a statistical difference. In multivariate analysis, the significant risk factors of recurrence were male gender and multifocality in group I, and lymph node metastasis and multifocality in group II. In particular, the male gender and multifocality showed the highest odds ratio (OR) on each group (OR, 4.721 and 6.177).
CONCLUSION
The patients with PTMCs had different clinical features and prognostic factors according to age. Hence, clinicians should consider a different strategy for therapy and plan for follow-up according to age.
Keyword
MeSH Terms
Figure
Reference
-
1. DeLellis RA, Lloyd RV, Heitz PU, Eng C. Pathology and genetics of tumours of endocrine organs. 2004. Lyon: IARC Press;(World Health Organization classification of tumours; v. 8).2. Ito Y, Uruno T, Nakano K, Takamura Y, Miya A, Kobayashi K, et al. An observation trial without surgical treatment in patients with papillary microcarcinoma of the thyroid. Thyroid. 2003. 13:381–387.3. Chow SM, Law SC, Chan JK, Au SK, Yau S, Lau WH. Papillary microcarcinoma of the thyroid-Prognostic significance of lymph node metastasis and multifocality. Cancer. 2003. 98:31–40.4. Bramley MD, Harrison BJ. Papillary microcarcinoma of the thyroid gland. Br J Surg. 1996. 83:1674–1683.5. Franssila KO, Harach HR. Occult papillary carcinoma of the thyroid in children and young adults. A systemic autopsy study in Finland. Cancer. 1986. 58:715–719.6. Noguchi S, Yamashita H, Uchino S, Watanabe S. Papillary microcarcinoma. World J Surg. 2008. 32:747–753.7. Cappelli C, Castellano M, Braga M, Gandossi E, Pirola I, De Martino E, et al. Aggressiveness and outcome of papillary thyroid carcinoma (PTC) versus microcarcinoma (PMC): a mono-institutional experience. J Surg Oncol. 2007. 95:555–560.8. Liou MJ, Lin JD, Chung MH, Liau CT, Hsueh C. Renal metastasis from papillary thyroid microcarcinoma. Acta Otolaryngol. 2005. 125:438–442.9. Lee J, Rhee Y, Lee S, Ahn CW, Cha BS, Kim KR, et al. Frequent, aggressive behaviors of thyroid microcarcinomas in Korean patients. Endocr J. 2006. 53:627–632.10. Arora N, Turbendian HK, Kato MA, Moo TA, Zarnegar R, Fahey TJ 3rd. Papillary thyroid carcinoma and microcarcinoma: is there a need to distinguish the two? Thyroid. 2009. 19:473–477.11. Mercante G, Frasoldati A, Pedroni C, Formisano D, Renna L, Piana S, et al. Prognostic factors affecting neck lymph node recurrence and distant metastasis in papillary microcarcinoma of the thyroid: results of a study in 445 patients. Thyroid. 2009. 19:707–716.12. Orsenigo E, Beretta E, Fiacco E, Scaltrini F, Veronesi P, Invernizzi L, et al. Management of papillary microcarcinoma of the thyroid gland. Eur J Surg Oncol. 2004. 30:1104–1106.13. Lin KL, Wang OC, Zhang XH, Dai XX, Hu XQ, Qu JM. The BRAF mutation is predictive of aggressive clinicopathological characteristics in papillary thyroid microcarcinoma. Ann Surg Oncol. 2010. 17:3294–3300.14. Giordano D, Gradoni P, Oretti G, Molina E, Ferri T. Treatment and prognostic factors of papillary thyroid microcarcinoma. Clin Otolaryngol. 2010. 35:118–124.15. Lee J, Yun JS, Nam KH, Chung WY, Soh EY, Park CS. Papillary thyroid microcarcinoma: clinicopathologic characteristics and treatment strategy. J Korean Surg Soc. 2007. 72:276–282.16. Lee KJ, Cho YJ, Kim SJ, Lee SC, Kim JG, Ahn CJ, et al. Analysis of the clinicopathologic features of papillary thyroid microcarcinoma based on 7-mm tumor size. World J Surg. 2011. 35:318–323.17. Lombardi CP, Bellantone R, De Crea C, Paladino NC, Fadda G, Salvatori M, et al. Papillary thyroid microcarcinoma: extrathyroidal extension, lymph node metastases, and risk factors for recurrence in a high prevalence of goiter area. World J Surg. 2010. 34:1214–1221.18. Moon HJ, Kim EK, Chung WY, Yoon JH, Kwak JY. Minimal extrathyroidal extension in patients with papillary thyroid microcarcinoma: is it a real prognostic factor? Ann Surg Oncol. 2011. 18:1916–1923.19. Ito Y, Miyauchi A, Inoue H, Fukushima M, Kihara M, Higashiyama T, et al. An observational trial for papillary thyroid microcarcinoma in Japanese patients. World J Surg. 2010. 34:28–35.20. Grodski S, Delbridge L. An update on papillary microcarcinoma. Curr Opin Oncol. 2009. 21:1–4.21. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010. 17:1471–1474.22. Yu XM, Wan Y, Sippel RS, Chen H. Should all papillary thyroid microcarcinomas be aggressively treated? An analysis of 18,445 cases. Ann Surg. 2011. 254:653–660.23. Wada N, Duh QY, Sugino K, Iwasaki H, Kameyama K, Mimura T, et al. Lymph node metastasis from 259 papillary thyroid microcarcinomas: frequency, pattern of occurrence and recurrence, and optimal strategy for neck dissection. Ann Surg. 2003. 237:399–407.24. Ito Y, Tomoda C, Uruno T, Takamura Y, Miya A, Kobayashi K, et al. Clinical significance of metastasis to the central compartment from papillary microcarcinoma of the thyroid. World J Surg. 2006. 30:91–99.25. So YK, Son YI, Hong SD, Seo MY, Baek CH, Jeong HS, et al. Subclinical lymph node metastasis in papillary thyroid microcarcinoma: a study of 551 resections. Surgery. 2010. 148:526–531.26. Roh JL, Kim JM, Park CI. Central cervical nodal metastasis from papillary thyroid microcarcinoma: pattern and factors predictive of nodal metastasis. Ann Surg Oncol. 2008. 15:2482–2486.27. Besic N, Zgajnar J, Hocevar M, Petric R. Extent of thyroidectomy and lymphadenectomy in 254 patients with papillary thyroid microcarcinoma: a single-institution experience. Ann Surg Oncol. 2009. 16:920–928.28. Paes JE, Hua K, Nagy R, Kloos RT, Jarjoura D, Ringel MD. The relationship between body mass index and thyroid cancer pathology features and outcomes: a clinicopathological cohort study. J Clin Endocrinol Metab. 2010. 95:4244–4250.29. Goodman MT, Kolonel LN, Wilkens LR. The association of body size, reproductive factors and thyroid cancer. Br J Cancer. 1992. 66:1180–1184.30. Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet. 2008. 371:569–578.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Ectopic Thyroid Papillary Carcinoma with Incidental Papillary Thyroid Microcarcinoma
- Analysis of Relationship between Prognostic Factors and Number of Fine-Needle Aspiration Cytology (FNAC) Prior to Surgery for Papillary Thyroid Microcarcinoma According to Size
- A Case of Cystic Lymph Node Metastasis from Thyroid Papillary Microcarcinoma
- The Prognostic Factors and Therapeutic Strategy for Papillary Thyroid Microcarcinoma
- A Review of Active Surveillance of Papillary Thyroid Microcarcinoma