Tuberc Respir Dis.
2014 Jan;76(1):23-29.
Interferon-gamma Enzyme-Linked Immunospot Assay in Patients with Tuberculosis and Healthy Adults
- Affiliations
-
- 1Department of Internal Medicine, Hallym University College of Medicine, Chuncheon, Korea. ighyun@hallym.or.kr
- 2Department of Laboratory Medicine, Hallym University College of Medicine, Chuncheon, Korea. kimhyun@hallym.or.kr
Abstract
- BACKGROUND
Interferon-gamma assays based on tuberculosis (TB)-specific antigens have been utilized for diagnosing and ruling out latent TB and active TB, but their utility is still limited for TB incidence countries. The aim of this study is to understand the clinical utility of enzyme-linked immunospot (ELISpot) assays among patients with clinically suspected TB and healthy adults in clinical practices and community-based settings.
METHODS
The ELISpot assays (T SPOT.TB, Oxford Immunotec, UK) were prospectively performed in 202 patients. After excluding those with indeterminate results, 196 were included for analysis: 41 were TB patients, 93 were non-TB patients, and 62 were healthy adults.
RESULTS
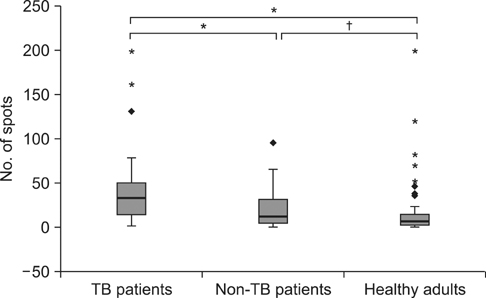
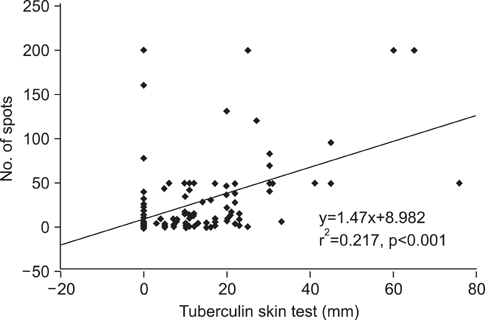
The sensitivity and negative predictive values of the T SPOT.TB assays for the diagnosis of TB were 87.8% and 89.1%, respectively, among patients with suspected TB. The agreement between the tuberculin skin test (10-mm cutoff) and the T SPOT.TB assay was 66.1% (kappa=0.335) in all participants and 80.0% (kappa=0.412) in TB patients. Among those without TB (n=155), a past history of TB and fibrotic TB scar on chest X-rays were significant factors that yielded positive T SPOT.TB results. There was a significant difference in the magnitude of T SPOT.TB spot counts between TB patients and non-TB patients or healthy adults.
CONCLUSION
The T SPOT.TB assay appeared to be a useful test for the diagnostic exclusion of TB. A positive result, however, should be cautiously interpreted for potential positives among those without active TB in intermediate TB incidence areas.
MeSH Terms
Figure
Reference
-
1. Dye C, Scheele S, Dolin P, Pathania V, Raviglione MC. Consensus statement. Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. WHO Global Surveillance and Monitoring Project. JAMA. 1999; 282:677–686.2. Aber VR, Allen BW, Mitchison DA, Ayuma P, Edwards EA, Keyes AB. Quality control in tuberculosis bacteriology. 1. Laboratory studies on isolated positive cultures and the efficiency of direct smear examination. Tubercle. 1980; 61:123–133.3. Pai M, Riley LW, Colford JM Jr. Interferon-gamma assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect Dis. 2004; 4:761–776.4. Gooding S, Chowdhury O, Hinks T, Richeldi L, Losi M, Ewer K, et al. Impact of a T cell-based blood test for tuberculosis infection on clinical decision-making in routine practice. J Infect. 2007; 54:e169–e174.5. Jasmer RM, Nahid P, Hopewell PC. Clinical practice: latent tuberculosis infection. N Engl J Med. 2002; 347:1860–1866.6. Mazurek GH, Jereb J, Vernon A, LoBue P, Goldberg S, Castro K, et al. Updated guidelines for using interferon gamma release assays to detect Mycobacterium tuberculosis infection: United States, 2010. MMWR Recomm Rep. 2010; 59:1–25.7. Sokal JE. Editorial: measurement of delayed skin-test responses. N Engl J Med. 1975; 293:501–502.8. Kang YA, Lee HW, Hwang SS, Um SW, Han SK, Shim YS, et al. Usefulness of whole-blood interferon-gamma assay and interferon-gamma enzyme-linked immunospot assay in the diagnosis of active pulmonary tuberculosis. Chest. 2007; 132:959–965.9. Kang YA, Lee HW, Yoon HI, Cho B, Han SK, Shim YS, et al. Discrepancy between the tuberculin skin test and the whole-blood interferon gamma assay for the diagnosis of latent tuberculosis infection in an intermediate tuberculosis-burden country. JAMA. 2005; 293:2756–2761.10. Lee JY, Choi HJ, Park IN, Hong SB, Oh YM, Lim CM, et al. Comparison of two commercial interferon-gamma assays for diagnosing Mycobacterium tuberculosis infection. Eur Respir J. 2006; 28:24–30.11. Huebner RE, Schein MF, Bass JB Jr. The tuberculin skin test. Clin Infect Dis. 1993; 17:968–975.12. Hong YP, Kim SJ, Lew WJ, Lee EK, Han YC. The seventh nationwide tuberculosis prevalence survey in Korea, 1995. Int J Tuberc Lung Dis. 1998; 2:27–36.13. Lalvani A, Pathan AA, Durkan H, Wilkinson KA, Whelan A, Deeks JJ, et al. Enhanced contact tracing and spatial tracking of Mycobacterium tuberculosis infection by enumeration of antigen-specific T cells. Lancet. 2001; 357:2017–2021.14. Richeldi L, Ewer K, Losi M, Bergamini BM, Roversi P, Deeks J, et al. T cell-based tracking of multidrug resistant tuberculosis infection after brief exposure. Am J Respir Crit Care Med. 2004; 170:288–295.15. Ewer K, Deeks J, Alvarez L, Bryant G, Waller S, Andersen P, et al. Comparison of T-cell-based assay with tuberculin skin test for diagnosis of Mycobacterium tuberculosis infection in a school tuberculosis outbreak. Lancet. 2003; 361:1168–1173.16. Janssens JP, Roux-Lombard P, Perneger T, Metzger M, Vivien R, Rochat T. Quantitative scoring of an interferon-gamma assay for differentiating active from latent tuberculosis. Eur Respir J. 2007; 30:722–728.17. McKay A, Kraut A, Murdzak C, Yassi A. Determinants of tuberculin reactivity among health care workers: Interpretation of positivity following BCG vaccination. Can J Infect Dis. 1999; 10:134–139.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Accuracy of an Interferon-gamma Release Assay to Detect Active Tuberculosis in Children: A Pilot Study
- Safety and Effectiveness of Cytomegalovirus Specific Antigen Induced Interferon-Gamma ELISPOT/ELISA: A Systematic Review
- Canges of Peripheral IFN-r-Producing CD4(+) and CD4(-) Cell Frequencies in Kawasaki Disease
- A Case of Sulfasalazine-Induced Hypersensitivity Syndrome Confirmed by Enzyme-Linked Immunospot Assay
- Diagnosis and treatment of latent tuberculosis infection