Korean J Urol.
2013 Oct;54(10):697-702.
Effects of Low-Dose Tamsulosin on Sexual Function in Patients With Lower Urinary Tract Symptoms Suggestive of Benign Prostatic Hyperplasia
- Affiliations
-
- 1Department of Urology, Hallym University College of Medicine, Chuncheon, Korea. yang1408@hallym.or.kr
- 2Department of Urology, Kyung Hee University School of Medicine, Seoul, Korea.
- 3Department of Urology, Korea University College of Medicine, Seoul, Korea.
Abstract
- PURPOSE
The aim of the present study was to evaluate the effects of low-dose tamsulosin on sexual function in patients with lower urinary tract symptoms (LUTS) suggestive of benign prostatic hyperplasia.
MATERIALS AND METHODS
A total of 138 male LUTS patients aged more than 50 years with an International Prostate Symptom Score (IPSS) > or =8 were enrolled in this open-label, multicenter, prospective, noncomparative observational study. Clinical assessments included IPSS, quality of life (QoL) index, International Index of Erectile Function (IIEF), Danish Prostate Symptom Score (DAN-PSS), and an early morning erection questionnaire. The data were recorded at baseline and at 1 and 3 months after treatment with tamsulosin 0.2 mg/d. Adverse events were analyzed in all patients.
RESULTS
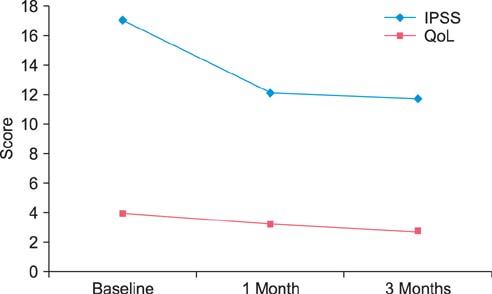
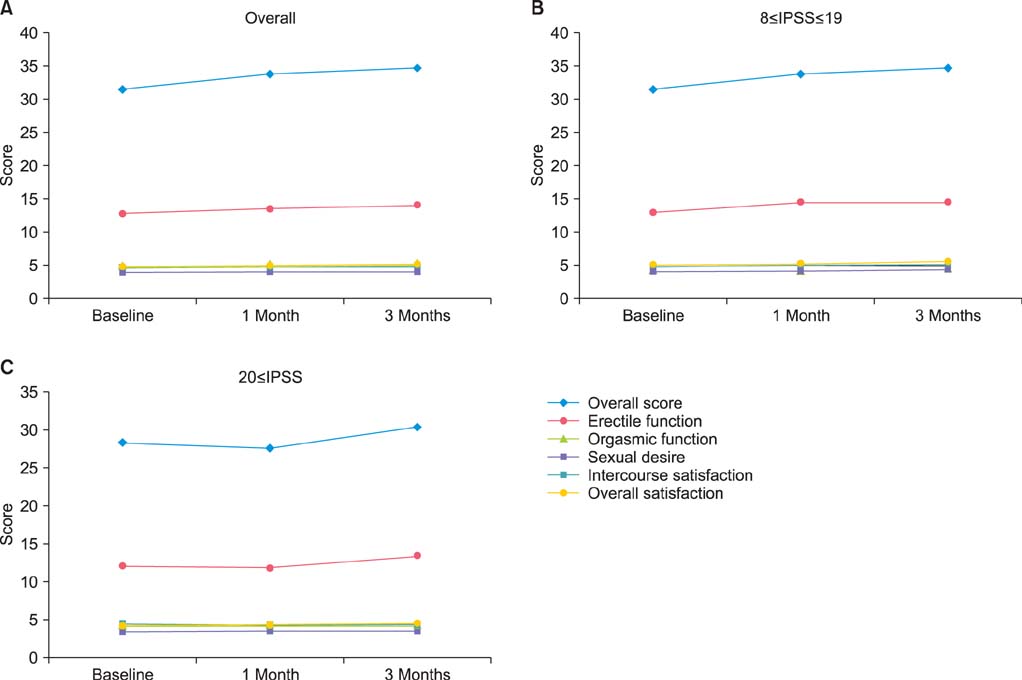
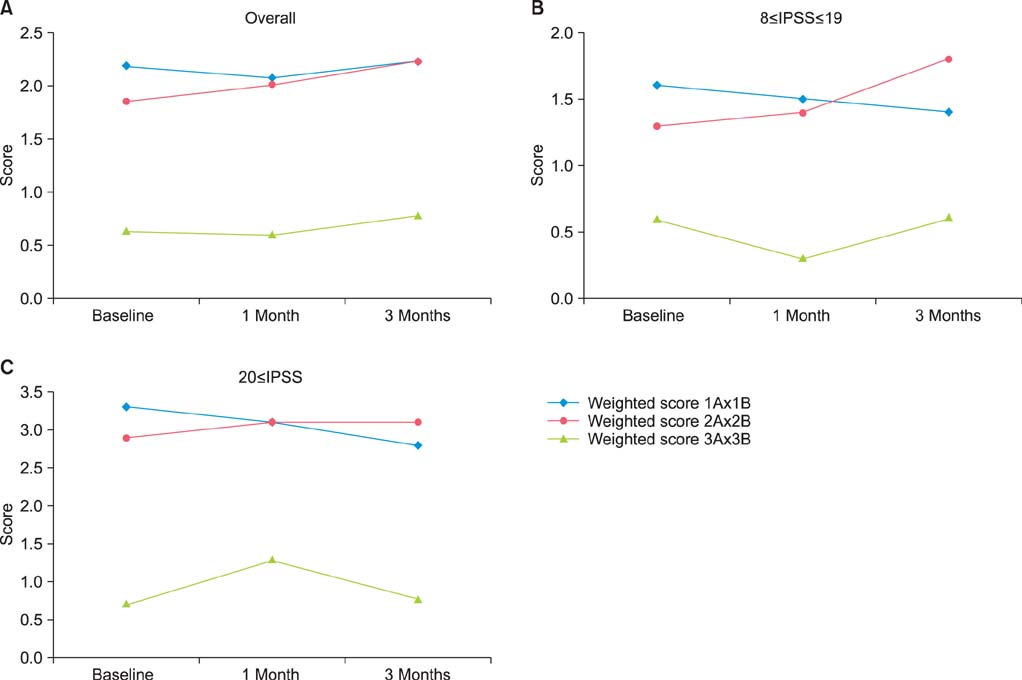
During the study period of 3 months, the IPSS and QoL index significantly improved from baseline by -11.40+/-9.40 and -1.11+/-1.36, respectively (p<0.001). However, there were no clinically relevant changes in total IIEF score (mean difference, 1.63+/-15.50; p=0.406) or the 5 subdomains (p>0.05). Furthermore, DAN-PSS weighted scores (AxB) showed no clinically relevant changes (mean difference on Q1, Q2, and Q3: -0.45+/-2.94, 0.27+/-2.50, and -1.27+/-2.27, p>0.05). In addition, there were no clinically significant changes in responses on the early morning erection questionnaire.
CONCLUSIONS
Tamsulosin at the dose of 0.2 mg significantly improved the IPSS and the QoL index compared with baseline. However, tamsulosin did not exhibit any significant impact on sexual function or any negative impact on ejaculatory function.
Keyword
MeSH Terms
Figure
Reference
-
1. Berry SJ, Coffey DS, Walsh PC, Ewing LL. The development of human benign prostatic hyperplasia with age. J Urol. 1984; 132:474–479.2. Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol. 1994; 151:54–61.3. Braun M, Wassmer G, Klotz T, Reifenrath B, Mathers M, Engelmann U. Epidemiology of erectile dysfunction: results of the Cologne Male Survey. Int J Impot Res. 2000; 12:305–311.4. Rosen R, Altwein J, Boyle P, Kirby RS, Lukacs B, Meuleman E, et al. Lower urinary tract symptoms and male sexual dysfunction: the multinational survey of the aging male (MSAM-7). Eur Urol. 2003; 44:637–649.5. Martin-Morales A, Sanchez-Cruz JJ, Saenz de, Rodriguez-Vela L, Jimenez-Cruz JF, Burgos-Rodriguez R. Prevalence and independent risk factors for erectile dysfunction in Spain: results of the Epidemiologia de la Disfuncion Erectil Masculina Study. J Urol. 2001; 166:569–574.6. Boyle P, Robertson C, Mazzetta C, Keech M, Hobbs R, Fourcade R, et al. The association between lower urinary tract symptoms and erectile dysfunction in four centres: the UrEpik study. BJU Int. 2003; 92:719–725.7. Braun MH, Sommer F, Haupt G, Mathers MJ, Reifenrath B, Engelmann UH. Lower urinary tract symptoms and erectile dysfunction: co-morbidity or typical "Aging Male" symptoms? Results of the "Cologne Male Survey". Eur Urol. 2003; 44:588–594.8. Kim SW. Prostatic disease and sexual dysfunction. Korean J Urol. 2011; 52:373–378.9. Andersson KE. Alpha-adrenoceptors and benign prostatic hyperplasia: basic principles for treatment with alpha-adrenoceptor antagonists. World J Urol. 2002; 19:390–396.10. Medina JJ, Parra RO, Moore RG. Benign prostatic hyperplasia (the aging prostate). Med Clin North Am. 1999; 83:1213–1229.11. Reilly CM, Stopper VS, Mills TM. Androgens modulate the alpha-adrenergic responsiveness of vascular smooth muscle in the corpus cavernosum. J Androl. 1997; 18:26–31.12. Chapple CR, Wyndaele JJ, Nordling J, Boeminghaus F, Ypma AF, Abrams P. Tamsulosin, the first prostate-selective alpha 1Aadrenoceptor antagonist: a meta-analysis of two randomized, placebo-controlled, multicentre studies in patients with benign prostatic obstruction (symptomatic BPH): European Tamsulosin Study Group. Eur Urol. 1996; 29:155–167.13. AUA Practice Guidelines Committee. AUA guideline on management of benign prostatic hyperplasia (2003): chapter 1. diagnosis and treatment recommendations. J Urol. 2003; 170(2 Pt 1):530–547.14. Choi HR, Chung WS, Shim BS, Kwon SW, Hong SJ, Chung BH, et al. Translation validity and reliability of I-PSS Korean version. Korean J Urol. 1996; 37:659–665.15. Chung TG, Lee TK, Chung S, Lee MS, Kim YS, Ahn TY. The Korean version of the International Index of Erectile Function (IIEF): reliability and validation study. Korean J Urol. 1999; 40:1334–1343.16. Kim DS, Chung BH. Translation validity and reliability of the Korean version of the DAN-PSS. Korean J Urol. 2003; 44:871–875.17. Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension. 2003; 42:1206–1252.18. Madersbacher S, Alivizatos G, Nordling J, Sanz CR, Emberton M, de la Rosette JJ. EAU 2004 guidelines on assessment, therapy and follow-up of men with lower urinary tract symptoms suggestive of benign prostatic obstruction (BPH guidelines). Eur Urol. 2004; 46:547–554.19. Cho IR. Benign prostatic hyperplasia and sexual dysfunction. Korean J Androl. 2010; 28:145–153.20. Lepor H. Phase III multicenter placebo-controlled study of tamsulosin in benign prostatic hyperplasia: Tamsulosin Investigator Group. Urology. 1998; 51:892–900.21. Lepor H. Long-term evaluation of tamsulosin in benign prostatic hyperplasia: placebo-controlled, double-blind extension of phase III trial: Tamsulosin Investigator Group. Urology. 1998; 51:901–906.22. Narayan P, Lepor H. Long-term, open-label, phase III multicenter study of tamsulosin in benign prostatic hyperplasia. Urology. 2001; 57:466–470.23. Yokoyama T, Hara R, Fukumoto K, Fujii T, Jo Y, Miyaji Y, et al. Effects of three types of alpha-1 adrenoreceptor blocker on lower urinary tract symptoms and sexual function in males with benign prostatic hyperplasia. Int J Urol. 2011; 18:225–230.24. Traish AM, Gupta S, Toselli P, de Tejada IS, Goldstein I, Moreland RB. Identification of alpha 1-adrenergic receptor subtypes in human corpus cavernosum tissue and in cultured trabecular smooth muscle cells. Receptor. 1995; 5:145–157.25. Nordling J. Efficacy and safety of two doses (10 and 15 mg) of alfuzosin or tamsulosin (0.4 mg) once daily for treating symptomatic benign prostatic hyperplasia. BJU Int. 2005; 95:1006–1012.26. Hofner K, Claes H, De Reijke TM, Folkestad B, Speakman MJ. Tamsulosin 0.4 mg once daily: effect on sexual function in patients with lower urinary tract symptoms suggestive of benign prostatic obstruction. Eur Urol. 1999; 36:335–341.27. Buzelin JM, Fonteyne E, Kontturi M, Witjes WP, Khan A. Comparison of tamsulosin with alfuzosin in the treatment of patients with lower urinary tract symptoms suggestive of bladder outlet obstruction (symptomatic benign prostatic hyperplasia): the European Tamsulosin Study Group. Br J Urol. 1997; 80:597–605.28. Kim JH, Park JY, Oh MM, Lee JG, Kwon SS, Bae JH. Treatment satisfaction with low-dose tamsulosin for symptomatic benign prostatic hyperplasia: results from a multicentre cross-sectional survey. Int J Clin Pract. 2012; 66:1209–1215.29. Lee E. Comparison of tamsulosin and finasteride for lower urinary tract symptoms associated with benign prostatic hyperplasia in Korean patients. J Int Med Res. 2002; 30:584–590.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Benign Prostatic Hyperplasia and Sexual Dysfunction
- Impact of Lower Urinary Tract Symptoms/Benign Prostatic Hyperplasia Treatment with Tamsulosin and Solifenacin Combination Therapy on Erectile Function
- Effect of Tamsulosin, a Selective alpha1A-adrenoreceptor Antagonist, in Benign Prostatic Hyperplasia
- The Experience with Combination of Finasteride and Tamsulosin on Benign Prostatic Hyperplasia
- Efficacy and Tolerability of Tamsulosin 0.4 mg in Patients with Symptomatic Benign Prostatic Hyperplasia