Korean J Urol.
2009 Jul;50(7):656-662.
Low-Dose-Rate Brachytherapy for Low- and Intermediate-Risk Groups of Localized Prostate Cancer
- Affiliations
-
- 1Department of Urology, CHA Bundang Medical Center, CHA University, Seongnam, Korea. dsparkmd@cha.ac.kr
- 2Department of Radiation Oncology, CHA Bundang Medical Center, CHA University, Seongnam, Korea.
Abstract
- PURPOSE
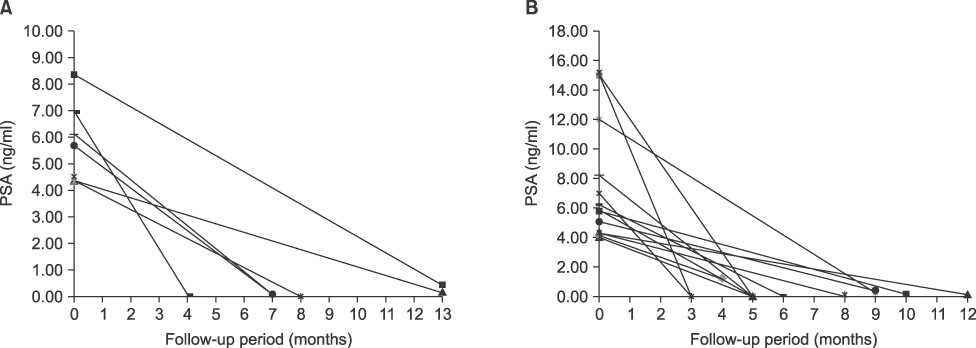
We applied low-dose-rate brachytherapy for low- and intermediate-risk groups of prostate cancer patients. Our initial experiences were analyzed to assess the result of low-dose-rate brachytherapy for low- and intermediate-risk groups of patients with localized prostate cancer. MATERIALS AND METHODS: A total of 50 consecutive patients have been treated with brachytherapy for 1 year since April 2007. Among them, a total of 24 patients in the low- or intermediate-risk groups were enrolled: 10 of the 24 patients were in the low-risk group (clinical T1a-T2b, Gleason score [GS] of 2-6, PSA<10 ng/ml), and 14 patients were in the intermediate-risk group (clinical T2b-T2c, Gleason score of 7, or PSA 10-20 ng/ml). Implantations were performed by practicing a real-time ultrasound-guided placement including prostatic capsular placement in the intermediate-risk group. All 24 patients were treated with 1 to 3 months of androgen-deprivation therapy. RESULTS: In the low- and intermediate-risk groups, the median patients' ages were 64 and 70 years, respectively. The numbers of patients in the low-risk group according to clinical T stage were 4 cases of T1c and 6 cases of T2a. The intermediate-risk group included 4 patients of stage T2a, 3 patients of stage T2b, and 7 patients of T2c. Five patients with a GS< or =6 and 9 patients with a GS of 7 were classified as being in the intermediate-risk group. Serum PSA levels in the intermediate-risk group were less than 10 ng/ml in 11 patients and 10-20 ng/ml in 3 patients. The median radiation doses delivered to 90% of the prostate in the low-risk and intermediate-risk groups were 257.5 Gy (range, 142.5-357.5 Gy) and 260.0 Gy (range, 147.5-357.5 Gy), respectively. Biochemical failure was not revealed in any case during follow-up. No patients experienced major complications. CONCLUSIONS: We can expect outstanding local control effect with low-dose-rate brachytherapy in low- and intermediate-risk prostate cancer. Our technique of modifying the insertion field in the intermediate-risk group is feasible and tolerable. However, long-term follow-up data are needed for this strategy.
Keyword
Figure
Reference
-
1. Jang TL, Han M, Roehl KA, Hawkins SA, Catalona WJ. More favorable tumor features and progression-free survival rates in a longitudinal prostate cancer screening study: PSA era and threshold-specific effects. Urology. 2006. 67:343–348.2. Blasko JC, Mate T, Sylvester JE, Grimm PD, Cavanagh W. Brachytherapy for carcinoma of the prostate: techniques, patient selection, and clinical outcomes. Semin Radiat Oncol. 2002. 12:81–94.3. Potters L, Morgenstern C, Calugaru E, Fearn P, Hassal A, Presser J, et al. 12-year outcomes following permanent prostate brachytherapy in patients with clinically localized prostate cancer. J Urol. 2005. 173:1562–1566.4. Oh MM, Bahk YW, Tropper SE. Preliminary report of clinical experience of iodine-125 seed implant for early prostatic cancer: the first case in Korea. Korean J Urol. 2001. 42:1235–1240.5. Cooperberg MR, Lubeck DP, Meng MV, Mehta SS, Carroll PR. The changing face of low-risk prostate cancer: trends in clinical presentation and primary management. J Clin Oncol. 2004. 22:2141–2149.6. Potters L, Fearn P, Kattan MW. External radiotherapy and permanent prostate brachytherapy in patients with localized prostate cancer. Brachytherapy. 2002. 1:36–41.7. Merrick GS, Butler WM, Galbreath RW, Lief JH. Five-year biochemical outcome following permanent interstitial brachytherapy for clinical T1-T3 prostate cancer. Int J Radiat Oncol Biol Phys. 2001. 51:41–48.8. Blasko JC, Ragde H, Grimm PD, Cavanagh W. Prostate brachytherapy: importance of technique. J Clin Oncol. 1996. 14:1965–1967.9. Ragde H, Blasko JC, Grimm PD, Kenny GM, Sylvester JE, Hoak DC, et al. Interstitial iodine-125 radiation without adjuvant therapy in the treatment of clinically localized prostate carcinoma. Cancer. 1997. 80:442–453.10. Merrick GS, Butler WM, Wallner KE, Galbreath RW, Anderson RL, Kurko BS, et al. Erectile function after prostate brachytherapy. Int J Radiat Oncol Biol Phys. 2005. 62:437–447.11. Shipley WU, Scardino PT, Kaufman DS, Katan MW. Vogelzang NJ, Scardino PT, Shipley WU, Debruyne FM, Linehan WM, editors. Treatment of early stage prostate cancer. Comprehensive textbook of genitourinary oncology. 2006. 3rd ed. Philadelphia: Lippincott Williams & Wilkins;153–157.12. Park DS, Jang WK, Oh JJ, Jee SH, Shin HS. Application of low dose rate brachytherapy in locally advanced prostate cancer. Korean J Urol Oncol. 2008. 6:127–132.13. Grimm PD, Blasko JC, Sylvester JE, Meier RM, Cavanagh W. 10-year biochemical (prostate-specific antigen) control of prostate cancer with (125)I brachytherapy. Int J Radiat Oncol Biol Phys. 2001. 51:31–40.14. Pollack A, Zagars GK, Starkschall G, Antolak JA, Lee JJ, Huang E, et al. Prostate cancer radiation dose response: results of the M.D. Anderson phase III randomized trial. Int J Radiat Oncol Biol Phys. 2002. 53:1097–1105.15. Frank SJ, Grimm PD, Sylvester JE, Merrick GS, Davis BJ, Zietman A, et al. Interstitial implant alone or in combination with external beam radiation therapy for intermediate-risk prostate cancer: a survey of practice patterns in the United States. Brachytherapy. 2007. 6:2–8.16. Lee I, Sandler H. Hormone therapy and radiotherapy for intermediate risk prostate cancer. Semin Radiat Oncol. 2008. 18:7–14.17. Guedea F, Aguilo F, Polo A, Langley S, Laing R, Henderson A, et al. Early biochemical outcomes following permanent interstitial brachytherapy as monotherapy in 1050 patients with clinical T1-T2 prostate cancer. Radiother Oncol. 2006. 80:57–61.18. Kwok Y, DiBiase SJ, Amin PP, Naslund M, Sklar G, Jacobs SC. Risk group stratification in patients undergoing permanent (125)I prostate brachytherapy as monotherapy. Int J Radiat Oncol Biol Phys. 2002. 53:588–594.19. Blasko JC, Grimm PD, Sylvester JE, Badiozamani KR, Hoak D, Cavanagh W. Palladium-103 brachytherapy for prostate carcinoma. Int J Radiat Oncol Biol Phys. 2000. 46:839–850.20. Stone NN, Stock RG. Long-term urinary, sexual, and rectal morbidity in patients treated with iodine-125 prostate brachytherapy followed up for a minimum of 5 years. Urology. 2007. 69:338–342.21. Mabjeesh NJ, Chen J, Stenger A, Matzkin H. Preimplant predictive factors of urinary retention after iodine 125 prostate brachytherapy. Urology. 2007. 70:548–553.22. Stock RG, Stone NN, Tabert A, Iannuzzi C, DeWyngaert JK. A dose-response study for I-125 prostate implants. Int J Radiat Oncol Biol Phys. 1998. 41:101–108.23. Kao J, Stone NN, Lavaf A, Dumane V, Cesaretti JA, Stock RG. (125)I monotherapy using D90 implant doses of 180Gy or greater. Int J Radiat Oncol Biol Phys. 2008. 70:96–101.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Permanent Brachytherapy of Localized Prostate Cancer: Preliminary Results
- Pulmonary Metastases After Low-Dose-Rate Brachytherapy for Localized Prostate Cancer
- Current Status of Brachytherapy for Prostate Cancer
- Hypofractionated stereotactic body radiotherapy in low- and intermediate-risk prostate carcinoma
- Low Dose versus High Dose Radioiodine Therapy