Nutr Res Pract.
2012 Dec;6(6):491-498.
Antioxidant activities of licorice-derived prenylflavonoids
- Affiliations
-
- 1School of Applied Biosciences and Food Science and Biotechnology, BK21 research Team for Developing Functional Health Food Materials, Kyungpook National University, 80 Daehak-ro, Buk-gu, Daegu 702-701, Korea. vision@knu.ac.kr
- 2R&D Team, Gyeongbuk Natural Color Institute, Yeongcheon, Gyeongbuk 770-906, Korea.
- 3Department of Food Science and Nutrition, Hallym University, Chuncheon, Gangwon 200-702, Korea.
Abstract
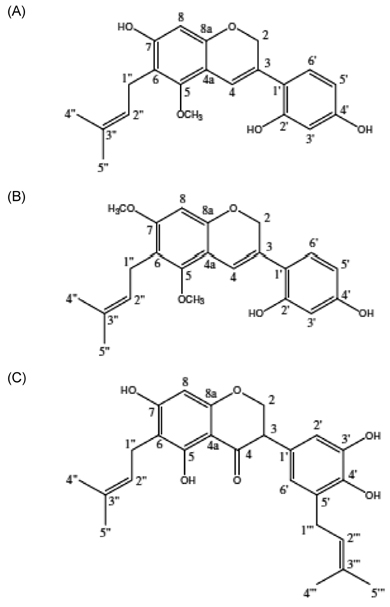
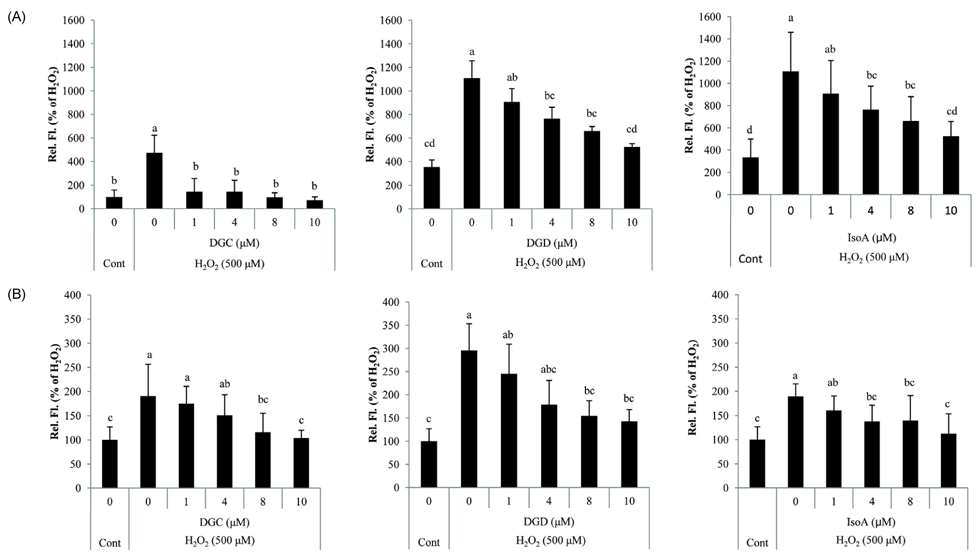
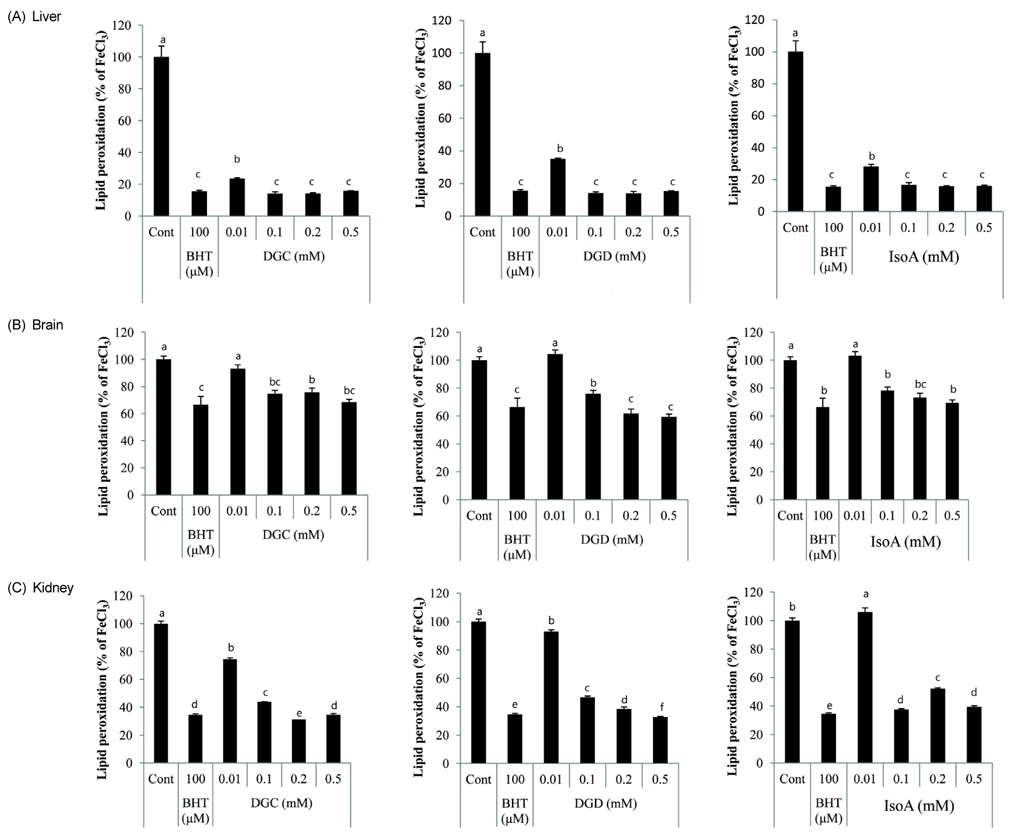
- Glycyrrhiza uralensis (or licorice) is a widely used Oriental herbal medicine from which the phenylflavonoids dehydroglyasperin C (DGC), dehydroglyasperin D (DGD), and isoangustone A (IsoA) are derived. The purpose of the present study was to evaluate the antioxidant properties of DGC, DGD, and IsoA. The three compounds showed strong ferric reducing activities and effectively scavenged DPPH, ABTS+, and singlet oxygen radicals. Among the three compounds tested, DGC showed the highest free radical scavenging capacity in human hepatoma HepG2 cells as assessed by oxidant-sensitive fluorescent dyes dichlorofluorescein diacetate and dihydroethidium bromide. In addition, all three compounds effectively suppressed lipid peroxidation in rat tissues as well as H2O2-induced ROS production in hepatoma cells. This study demonstrates that among the three phenylflavonoids isolated from licorice, DGC possesses the most potent antioxidant activity, suggesting it has protective effects against chronic diseases caused by reactive oxygen species as well as potential as an antioxidant food additive.
MeSH Terms
-
Animals
Benzopyrans
Carcinoma, Hepatocellular
Chronic Disease
Ethidium
Flavonoids
Fluorescent Dyes
Food Additives
Glycyrrhiza
Glycyrrhiza uralensis
Hep G2 Cells
Herbal Medicine
Humans
Isoflavones
Lipid Peroxidation
Rats
Reactive Oxygen Species
Singlet Oxygen
Benzopyrans
Ethidium
Flavonoids
Fluorescent Dyes
Food Additives
Isoflavones
Reactive Oxygen Species
Singlet Oxygen
Figure
Reference
-
1. Lobo V, Patil A, Phatak A, Chandra N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn Rev. 2010. 4:118–126.
Article2. Hazra B, Biswas S, Mandal N. Antioxidant and free radical scavenging activity of Spondias pinnata. BMC Complement Altern Med. 2008. 8:63.
Article3. Halliwell B, Aeschbach R, Loliger J, Aruoma OI. The characterization of antioxidants. Food Chem Toxicol. 1995. 33:601–617.
Article4. Lopez-Varela S, Gonzalez-Gross M, Marcos A. Functional foods and the immune system: a review. Eur J Clin Nutr. 2002. 56:Suppl 3. S29–S33.
Article5. Veerapur VP, Prabhakar KR, Parihar VK, Kandadi MR, Ramakrishana S, Mishra B, Satish Rao BS, Srinivasan KK, Priyadarsini KI, Unnikrishnan MK. Ficus racemosa stem bark extract: apotent antioxidant and a probable natural radioprotector. Evid Based Complement Alternat Med. 2009. 6:317–324.
Article6. Mae T, Kishida H, Nishiyama T, Tsukagawa M, Konishi E, Kuroda M, Mimaki Y, Sashida Y, Takahashi K, Kawada T, Nakagawa K, Kitahara M. A licorice ethanolic extract with peroxisome proliferator-activated receptor-gamma ligand-binding activity affects diabetes in KK-Ay mice, abdominal obesity in diet-induced obese C57BL mice and hypertension in spontaneously hypertensive rats. J Nutr. 2003. 133:3369–3377.
Article7. Fukai T, Marumo A, Kaitou K, Kanda T, Terada S, Nomura T. Anti-Helicobacter pylori flavonoids from licorice extract. Life Sci. 2002. 71:1449–1463.
Article8. Kondo K, Shiba M, Yamaji H, Morota T, Zhengmin C, Huixia P, Shoyama Y. Species identification of licorice using nrDNA and cpDNA genetic markers. Biol Pharm Bull. 2007. 30:1497–1502.
Article9. Kuroda M, Mimaki Y, Sashida Y, Mae T, Kishida H, Nishiyama T, Tsukagawa M, Konishi E, Takahashi K, Kawada T, Nakagawa K, Kitahara M. Phenolics with PPAR-gamma ligand-binding activity obtained from licorice (Glycyrrhiza uralensis roots) and ameliorative effects of glycyrin on genetically diabetic KK-A(y) mice. Bioorg Med Chem Lett. 2003. 13:4267–4272.
Article10. Hatano T, Shintani Y, Aga Y, Shiota S, Tsuchiya T, Yoshida T. Phenolic constituents of licorice. VIII. Structures of glicophenone and glicoisoflavanone, and effects of licorice phenolics on methicillin-resistant Staphylococcus aureus. Chem Pharm Bull (Tokyo). 2000. 48:1286–1292.
Article11. Li J, Lim SS, Lee ES, Gong JH, Shin D, Kang IJ, Kang YH. Isoangustone A suppresses mesangial fibrosis and inflammation in human renal mesangial cells. Exp Biol Med (Maywood). 2011. 236:435–444.
Article12. Seon MR, Park SY, Kwon SJ, Lim SS, Choi HJ, Park H, Lim DY, Kim JS, Lee CH, Kim J, Park JH. Hexane/ethanol extract of Glycyrrhiza uralensis and its active compound isoangustone A induce G1 cycle arrest in DU145 human prostate and 4T1 murine mammary cancer cells. J Nutr Biochem. 2012. 23:85–92.
Article13. Seon MR, Lim SS, Choi HJ, Park SY, Cho HJ, Kim JK, Kim J, Kwon DY, Park JH. Isoangustone A present in hexane/ethanol extract of Glycyrrhiza uralensis induces apoptosis in DU145 human prostate cancer cells via the activation of DR4 and intrinsic apoptosis pathway. Mol Nutr Food Res. 2010. 54:1329–1339.
Article14. Seo JY, Park J, Kim HJ, Lee IA, Lim JS, Lim SS, Choi SJ, Park JH, Kang HJ, Kim JS. Isoalantolactone from Inula helenium caused Nrf2-mediated induction of detoxifying enzymes. J Med Food. 2009. 12:1038–1045.
Article15. Lee YS, Kim SH, Kim JK, Shin HK, Kang YH, Park JH, Lim SS. Rapid identification and preparative isolation of antioxidant components in licorice. J Sep Sci. 2010. 33:664–671.
Article16. Benzie IF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the FRAP assay. Anal Biochem. 1996. 239:70–76.
Article17. Hatano T, Kagawa H, Yasuhara T, Okuda T. Two new flavonoids and other constituents in licorice root: their relative astringency and radical scavenging effects. Chem Pharm Bull (Tokyo). 1988. 36:2090–2097.
Article18. Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999. 26:1231–1237.
Article19. Kim HJ, Suh HJ, Kim JH, Park S, Joo YC, Kim JS. Antioxidant activity of glyceollins derived from soybean elicited with Aspergillus sojae. J Agric Food Chem. 2010. 58:11633–11638.
Article20. Wang H, Joseph JA. Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med. 1999. 27:612–616.
Article21. Li WG, Miller FJ Jr, Brown MR, Chatterjee P, Aylsworth GR, Shao J, Spector AA, Oberley LW, Weintraub NL. Enhanced H(2)O(2)-induced cytotoxicity in "epithelioid" smooth muscle cells: implications for neointimal regression. Arterioscler Thromb Vasc Biol. 2000. 20:1473–1479.22. Rao AR, Sarada R, Baskaran V, Ravishankar GA. Antioxidant activity of Botryococcus braunii extract elucidated in vitro models. J Agric Food Chem. 2006. 54:4593–4599.
Article23. Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol. 1978. 52:302–310.24. Jadhav HR, Bhutani KK. Antioxidant properties of Indian medicinal plants. Phytother Res. 2002. 16:771–773.
Article25. Shoemaker M, Hamilton B, Dairkee SH, Cohen I, Campbell MJ. In vitro anticancer activity of twelve Chinese medicinal herbs. Phytother Res. 2005. 19:649–651.
Article26. Amagase H, Petesch BL, Matsuura H, Kasuga S, Itakura Y. Intake of garlic and its bioactive components. J Nutr. 2001. 131:955S–962S.
Article27. Singh BN, Shankar S, Srivastava RK. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochem Pharmacol. 2011. 82:1807–1821.
Article28. Gibellini L, Pinti M, Nasi M, Montagna JP, De Biasi S, Roat E, Bertoncelli L, Cooper EL, Cossarizza A. Quercetin and cancer chemoprevention. Evid Based Complement Alternat Med. 2011. 2011:591356.
Article29. Higdon JV, Frei B. Tea catechins and polyphenols: health effects, metabolism, and antioxidant functions. Crit Rev Food Sci Nutr. 2003. 43:89–143.
Article30. Davis JM, Murphy EA, Carmichael MD. Effects of the dietary flavonoid quercetin upon performance and health. Curr Sports Med Rep. 2009. 8:206–213.
Article31. Cuendet M, Guo J, Luo Y, Chen S, Oteham CP, Moon RC, van Breemen RB, Marler LE, Pezzuto JM. Cancer chemopreventive activity and metabolism of isoliquiritigenin, a compound found in licorice. Cancer Prev Res (Phila). 2010. 3:221–232.
Article32. Fu Y, Hsieh TC, Guo J, Kunicki J, Lee MY, Darzynkiewicz Z, Wu JM. Licochalcone-A, a novel flavonoid isolated from licorice root (Glycyrrhiza glabra), causes G2 and late-G1 arrests in androgen-independent PC-3 prostate cancer cells. Biochem Biophys Res Commun. 2004. 322:263–270.
Article33. Heikens J, Fliers E, Endert E, Ackermans M, van Montfrans G. Liquorice-induced hypertension--a new understanding of an old disease: case report and brief review. Neth J Med. 1995. 47:230–234.
Article34. Sardi A, Geda C, Nerici L, Bertello P. Rhabdomyolysis and arterial hypertension caused by apparent excess of mineralocorticoids: a case report. Ann Ital Med Int. 2002. 17:126–129.35. Asl MN, Hosseinzadeh H. Review of pharmacological effects of Glycyrrhiza sp. and its bioactive compounds. Phytother Res. 2008. 22:709–724.
Article36. Harborne JB. Nature, distribution and function of plant flavonoids. Prog Clin Biol Res. 1986. 213:15–24.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Indigenization of Licorice and Its Meaning During the Early Days of the Joseon Dynasty
- Licorice-nduced Hypokalemia and Myopathy
- Licorice Induced Hypokalemia
- Electrical storm induced by hypokalemia associated with herbal medicines containing licorice
- A Case of Licorice-Induced Hypokalemic Rhabdomyolysis in a Patient Using a Diuretic Drug