Nat Prod Sci.
2016 Mar;22(1):35-40. 10.20307/nps.2016.22.1.35.
Chemical Composition of Clausena lansium (Lour.) Skeels Leaves and Antifungal Activity
- Affiliations
-
- 1National Institute of Medicinal Materials, Hanoi 100000, Vietnam. khoi_nguyenminh@yahoo.co.uk
- 2R&D Biological Science Research, KAO Corporation, Tochigi 321-3497, Japan.
- 3KAO Consumer Products (Southeast Asia) Co., Ltd. Bangkok 10330, Thailand.
- KMID: 2312919
- DOI: http://doi.org/10.20307/nps.2016.22.1.35
Abstract
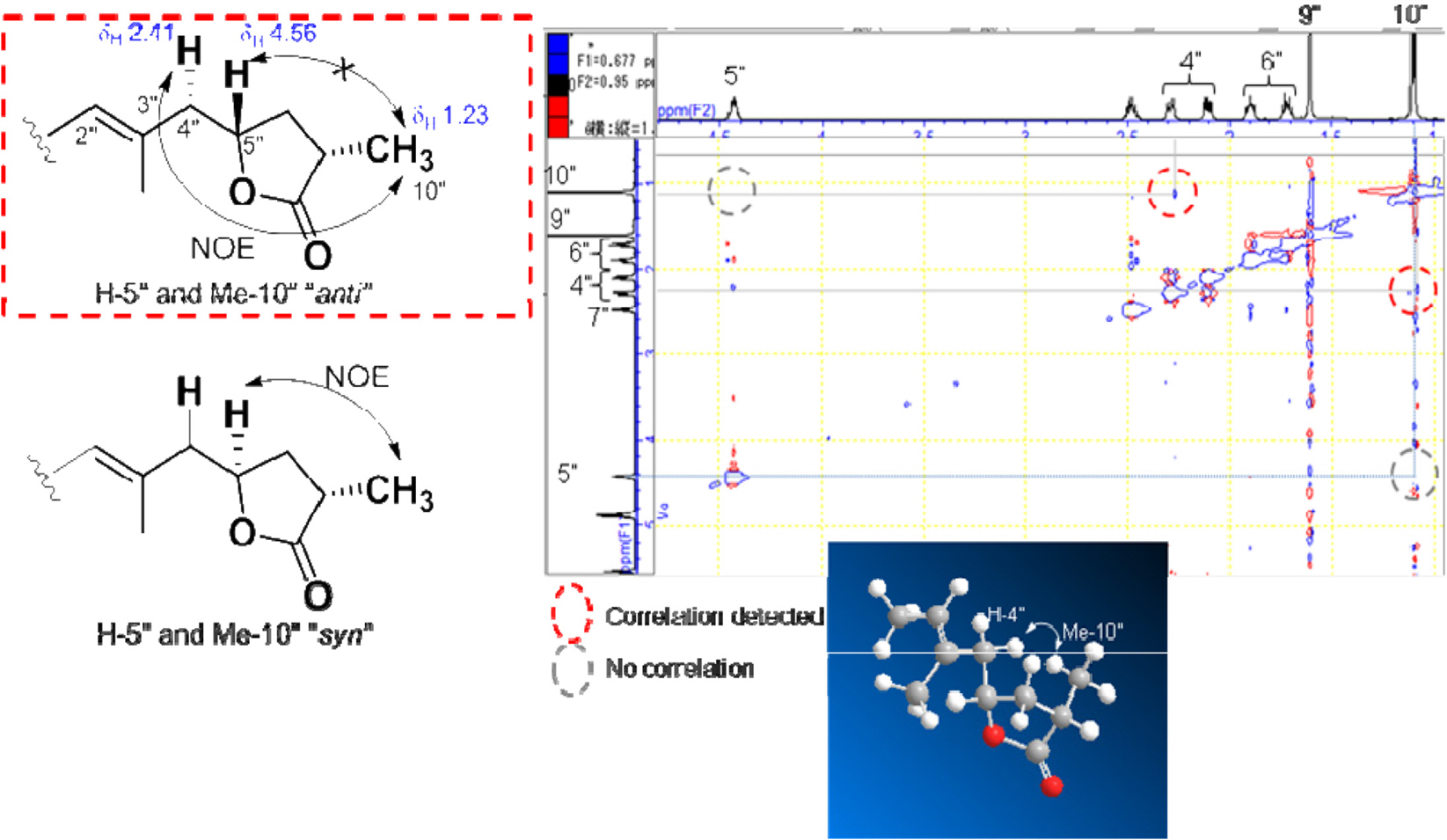
- The first study on chemical constituents and biological activities of Clausena lansium (Lour.) Skeels (Rutaceae) growing in Vietnam has been done. Phytochemical investigation of n-hexane extract led to the isolation of five compounds: dihydroindicolactone (1), 8-geranyloxy psoralen (2), imperatorin (3), heraclenol (4) and indicolactone (5), in which this is the first report on the presence of dihydroindicolactone (1). Their structures were elucidated based on LC/MS/NMR hyphenated techniques as well as comparison with those of literature data. The n-hexane extract and its subfractions, ethanol 95% extract and several isolated compounds were evaluated for antifungal activity.
Figure
Reference
-
(1). Chi V. V.The dictionary of Vietnamese medicinal Plants; Medicine Publishing House : Vietnam. 1999; 1148.(2). Adebajo A. C., Iwalewa E. O., Obuotor E. M., Ibikunle G. F., Omisore N. O., Adewunmi C. O., Obaparusi O. O., Klaes M., Adetogun G. E., Schmidt T. J., Verspohl E. J. J.Ethnopharmacol. 2009; 122:10–19.(3). Prasad K. N., Xie H., Hao J., Yang B., Qiu S., Wei X., Chen F., Jiang Y. J.Food Chem. 2010; 118:62–66.(4). Chokeprasert P., Charles A. L., Sue K. H., Huang T. C. J.Food Comp. Anal. 2007; 20:52–56.(5). Dincel D., Hatipoglu S. D., Goren A. C., Topcu G. Turk. J.Chem. 2013; 37:675–683.(6). Lakshmi V., Prakash D., Raj K., Kapil R. S., Popli S. P.Phytochemistry. 1984; 23:2629–2631.(7). Maneerat W., Prawat U., Saewan N., Laphookhieo S. J.Braz. Chem. Soc. 2010; 21:665–668.(8). Takamasa K., Koichi M., Michiko A., Ryoko S., Yuka M., Rui K., Atsuhiko H., Takashi S., Shuichi S., Shinichi W., Hideyo Y., Shigeru A., Noboru O. J.Clin. Microbiol. 2007; 45:3737–3742.(9). Shen D. Y., Chan Y. Y., Hwang T. L., Juang S. H., Huang S. C., Kuo P. C., Thang T. D., Lee E. J., Damu A. G., Wu T. S. J.Nat. Prod. 2014; 77:1215–1223.(10). Adams M., Ettl, S.;Kunert, O.;Wube A. A., Haslinger E., Bucar, F.;Bauer R.Planta Med. 2006; 72:1132–1135.(11). Singh S., Singh P., Singh S. K., Trivedi M., Dixit R. K., Shanker P. Int. Res. J.Pharm. App. Sci. 2013; 3:1–11.(12). Adikaram N. K. B., Abhayawardhane Y., Leslie Gunatilaka A. A., Ratnayake Bandara B. M., Kithsiri Wijeratne E. M.Plant Pathology. 1989; 38:258–265.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diversity and Antifungal Activity of Endophytic Fungi Associated with Camellia oleifera
- Antifungal Activity of 4',7-Dimethoxyisoflavone Against Some Fungi
- Chemical Constituents of the Fruiting Bodies of Clitocybe nebularis and Their Antifungal Activity
- Antifungal Activity of 2-Hydroxy 4,4'6'Trimethoxy Chalcone
- Essential Oil of Marrubium vulgare: Chemical Composition and Biological Activities. A Review