Korean J Physiol Pharmacol.
2015 Mar;19(2):141-149. 10.4196/kjpp.2015.19.2.141.
Differential Gene Expression in GPR40-Overexpressing Pancreatic beta-cells Treated with Linoleic Acid
- Affiliations
-
- 1Department of Pharmacology, Chungnam National University College of Pharmacy, Daejeon 305-764, Korea. cm8r@cnu.ac.kr
- 2Institute of Drug Research & Development, Chungnam National University, Daejeon 305-764, Korea.
- KMID: 2285566
- DOI: http://doi.org/10.4196/kjpp.2015.19.2.141
Abstract
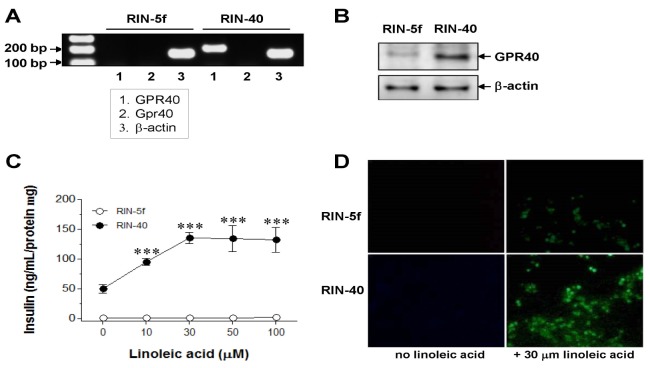
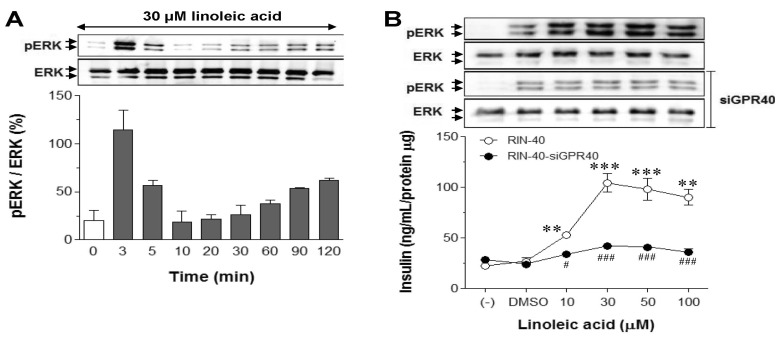
- "G protein-coupled receptor 40" (GPR40), a receptor for long-chain fatty acids, mediates the stimulation of glucose-induced insulin secretion. We examined the profiles of differential gene expression in GPR40-activated cells treated with linoleic acid, and finally predicted the integral pathways of the cellular mechanism of GPR40-mediated insulinotropic effects. After constructing a GPR40-overexpressing stable cell line (RIN-40) from the rat pancreatic beta-cell line RIN-5f, we determined the gene expression profiles of RIN-5f and RIN-40. In total, 1004 genes, the expression of which was altered at least twofold, were selected in RIN-5f versus RIN-40. Moreover, the differential genetic profiles were investigated in RIN-40 cells treated with 30 microM linoleic acid, which resulted in selection of 93 genes in RIN-40 versus RIN-40 treated with linoleic acid. Based on the Kyoto Encyclopedia of Genes and Genomes Pathway (KEGG, http://www.genome.jp/kegg/), sets of genes induced differentially by treatment with linoleic acid in RIN-40 cells were found to be related to mitogen-activated protein (MAP) kinase- and neuroactive ligand-receptor interaction pathways. A gene ontology (GO) study revealed that more than 30% of the genes were associated with signal transduction and cell proliferation. Thus, this study elucidated a gene expression pattern relevant to the signal pathways that are regulated by GPR40 activation during the acute period. Together, these findings increase our mechanistic understanding of endogenous molecules associated with GPR40 function, and provide information useful for identification of a target for the management of type 2 diabetes mellitus.
Keyword
MeSH Terms
Figure
Reference
-
1. Gerich JE. Is insulin resistance the principal cause of type 2 diabetes? Diabetes Obes Metab. 1999; 1:257–263. PMID: 11225637.
Article2. Wang J, Osei K. Proinsulin maturation disorder is a contributor to the defect of subsequent conversion to insulin in β-cells. Biochem Biophys Res Commun. 2011; 411:150–155. PMID: 21723250.
Article3. Hales CN, Barker DJ. Type 2 (non-insulin-dependent) diabetes mellitus: the thrifty phenotype hypothesis. Diabetologia. 1992; 35:595–601. PMID: 1644236.
Article4. Moller DE. New drug targets for type 2 diabetes and the metabolic syndrome. Nature. 2001; 414:821–827. PMID: 11742415.
Article5. Briscoe CP, Peat AJ, McKeown SC, Corbett DF, Goetz AS, Littleton TR, McCoy DC, Kenakin TP, Andrews JL, Ammala C, Fornwald JA, Ignar DM, Jenkinson S. Pharmacological regulation of insulin secretion in MIN6 cells through the fatty acid receptor GPR40: identification of agonist and antagonist small molecules. Br J Pharmacol. 2006; 148:619–628. PMID: 16702987.
Article6. Itoh Y, Kawamata Y, Harada M, Kobayashi M, Fujii R, Fukusumi S, Ogi K, Hosoya M, Tanaka Y, Uejima H, Tanaka H, Maruyama M, Satoh R, Okubo S, Kizawa H, Komatsu H, Matsumura F, Noguchi Y, Shinohara T, Hinuma S, Fujisawa Y, Fujino M. Free fatty acids regulate insulin secretion from pancreatic beta cells through GPR40. Nature. 2003; 422:173–176. PMID: 12629551.7. Salehi A, Flodgren E, Nilsson NE, Jimenez-Feltstrom J, Miyazaki J, Owman C, Olde B. Free fatty acid receptor 1 (FFA1R/GPR40) and its involvement in fatty-acid-stimulated insulin secretion. Cell Tissue Res. 2005; 322:207–215. PMID: 16044321.8. Steneberg P, Rubins N, Bartoov-Shifman R, Walker MD, Edlund H. The FFA receptor GPR40 links hyperinsulinemia, hepatic steatosis, and impaired glucose homeostasis in mouse. Cell Metab. 2005; 1:245–258. PMID: 16054069.
Article9. Nagasumi K, Esaki R, Iwachidow K, Yasuhara Y, Ogi K, Tanaka H, Nakata M, Yano T, Shimakawa K, Taketomi S, Takeuchi K, Odaka H, Kaisho Y. Overexpression of GPR40 in pancreatic beta-cells augments glucose-stimulated insulin secretion and improves glucose tolerance in normal and diabetic mice. Diabetes. 2009; 58:1067–1076. PMID: 19401434.10. Haber EP, Ximenes HM, Procopio J, Carvalho CR, Curi R, Carpinelli AR. Pleiotropic effects of fatty acids on pancreatic beta-cells. J Cell Physiol. 2003; 194:1–12. PMID: 12447984.11. Houze JB, Zhu L, Sun Y, Akerman M, Qiu W, Zhang AJ, Sharma R, Schmitt M, Wang Y, Liu J, Liu J, Medina JC, Reagan JD, Luo J, Tonn G, Zhang J, Lu JY, Chen M, Lopez E, Nguyen K, Yang L, Tang L, Tian H, Shuttleworth SJ, Lin DC. AMG 837: a potent, orally bioavailable GPR40 agonist. Bioorg Med Chem Lett. 2012; 22:1267–1270. PMID: 22217876.
Article12. Luo J, Swaminath G, Brown SP, Zhang J, Guo Q, Chen M, Nguyen K, Tran T, Miao L, Dransfield PJ, Vimolratana M, Houze JB, Wong S, Toteva M, Shan B, Li F, Zhuang R, Lin DC. A potent class of GPR40 full agonists engages the enteroinsular axis to promote glucose control in rodents. PLoS One. 2012; 7:e46300. PMID: 23056280.
Article13. Morgan NG. Fatty acids and beta-cell toxicity. Curr Opin Clin Nutr Metab Care. 2009; 12:117–122. PMID: 19202382.14. Rayasam GV, Tulasi VK, Sundaram S, Singh W, Kant R, Davis JA, Saini KS, Ray A. Identification of berberine as a novel agonist of fatty acid receptor GPR40. Phytother Res. 2010; 24:1260–1263. PMID: 20658575.
Article15. Araki T, Hirayama M, Hiroi S, Kaku K. GPR40-induced insulin secretion by the novel agonist TAK-875: first clinical findings in patients with type 2 diabetes. Diabetes Obes Metab. 2012; 14:271–278. PMID: 22051148.
Article16. Gromada J. The free fatty acid receptor GPR40 generates excitement in pancreatic beta-cells. Endocrinology. 2006; 147:672–673. PMID: 16418431.17. Feng DD, Luo Z, Roh SG, Hernandez M, Tawadros N, Keating DJ, Chen C. Reduction in voltage-gated K+ currents in primary cultured rat pancreatic beta-cells by linoleic acids. Endocrinology. 2006; 147:674–682. PMID: 16254037.18. Fujiwara K, Maekawa F, Yada T. Oleic acid interacts with GPR40 to induce Ca2+ signaling in rat islet beta-cells: mediation by PLC and L-type Ca2+ channel and link to insulin release. Am J Physiol Endocrinol Metab. 2005; 289:E670–E677. PMID: 15914509.19. Shapiro H, Shachar S, Sekler I, Hershfinkel M, Walker MD. Role of GPR40 in fatty acid action on the beta cell line INS-1E. Biochem Biophys Res Commun. 2005; 335:97–104. PMID: 16081037.20. Katsuma S, Hatae N, Yano T, Ruike Y, Kimura M, Hirasawa A, Tsujimoto G. Free fatty acids inhibit serum deprivationinduced apoptosis through GPR120 in a murine enteroendocrine cell line STC-1. J Biol Chem. 2005; 280:19507–19515. PMID: 15774482.
Article21. Seljeset S, Siehler S. Receptor-specific regulation of ERK1/2 activation by members of the "free fatty acid receptor" family. J Recept Signal Transduct Res. 2012; 32:196–201. PMID: 22712802.
Article22. Okubo Y, Blakesley VA, Stannard B, Gutkind S, Le Roith D. Insulin-like growth factor-I inhibits the stress-activated protein kinase/c-Jun N-terminal kinase. J Biol Chem. 1998; 273:25961–25966. PMID: 9748273.
Article23. Fritsche-Guenther R, Witzel F, Sieber A, Herr R, Schmidt N, Braun S, Brummer T, Sers C, Blüthgen N. Strong negative feedback from Erk to Raf confers robustness to MAPK signalling. Mol Syst Biol. 2011; 7:489. PMID: 21613978.
Article24. Mayer SI, Thiel G. Calcium influx into MIN6 insulinoma cells induces expression of Egr-1 involving extracellular signalregulated protein kinase and the transcription factors Elk-1 and CREB. Eur J Cell Biol. 2009; 88:19–33. PMID: 18783846.
Article25. Nolan CJ, Madiraju MS, Delghingaro-Augusto V, Peyot ML, Prentki M. Fatty acid signaling in the beta-cell and insulin secretion. Diabetes. 2006; 55(Suppl 2):S16–S23. PMID: 17130640.26. Taneera J, Lang S, Sharma A, Fadista J, Zhou Y, Ahlqvist E, Jonsson A, Lyssenko V, Vikman P, Hansson O, Parikh H, Korsgren O, Soni A, Krus U, Zhang E, Jing XJ, Esguerra JL, Wollheim CB, Salehi A, Rosengren A, Renström E, Groop L. A systems genetics approach identifies genes and pathways for type 2 diabetes in human islets. Cell Metab. 2012; 16:122–134. PMID: 22768844.
Article27. Yashiro H, Tsujihata Y, Takeuchi K, Hazama M, Johnson PR, Rorsman P. The effects of TAK-875, a selective G protein-coupled receptor 40/free fatty acid 1 agonist, on insulin and glucagon secretion in isolated rat and human islets. J Pharmacol Exp Ther. 2012; 340:483–489. PMID: 22106100.
Article28. Thanabalasingham G, Owen KR. Diagnosis and management of maturity onset diabetes of the young (MODY). BMJ. 2011; 343:d6044. PMID: 22012810.
Article29. Rosengren AH, Braun M, Mahdi T, Andersson SA, Travers ME, Shigeto M, Zhang E, Almgren P, Ladenvall C, Axelsson AS, Edlund A, Pedersen MG, Jonsson A, Ramracheya R, Tang Y, Walker JN, Barrett A, Johnson PR, Lyssenko V, McCarthy MI, Groop L, Salehi A, Gloyn AL, Renström E, Rorsman P, Eliasson L. Reduced insulin exocytosis in human pancreatic β-cells with gene variants linked to type 2 diabetes. Diabetes. 2012; 61:1726–1733. PMID: 22492527.
Article30. Yuan Q, Tang W, Zhang X, Hinson JA, Liu C, Osei K, Wang J. Proinsulin atypical maturation and disposal induces extensive defects in mouse Ins2+/Akita β-cells. PLoS One. 2012; 7:e35098. PMID: 22509386.31. Akinci E, Banga A, Greder LV, Dutton JR, Slack JM. Reprogramming of pancreatic exocrine cells towards a beta (β) cell character using Pdχ1, Ngn3 and MafA. Biochem J. 2012; 442:539–550. PMID: 22150363.32. Gaillard I, Rouquier S, Giorgi D. Olfactory receptors. Cell Mol Life Sci. 2004; 61:456–469. PMID: 14999405.
Article33. Chalé-Rush A, Burgess JR, Mattes RD. Multiple routes of chemosensitivity to free fatty acids in humans. Am J Physiol Gastrointest Liver Physiol. 2007; 292:G1206–G1212. PMID: 17234892.
Article34. Guo Y, Traurig M, Ma L, Kobes S, Harper I, Infante AM, Bogardus C, Baier LJ, Prochazka M. CHRM3 gene variation is associated with decreased acute insulin secretion and increased risk for early-onset type 2 diabetes in Pima Indians. Diabetes. 2006; 55:3625–3629. PMID: 17130513.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The influence of linoleic acid and ursolic acid on mouse peritoneal macrophage activity
- Isolation of ethanol-induced genes in pancreatic beta-cells by representational difference analysis (RDA)
- Effects of C18 Fatty Acids on Intracellular Ca2+ Mobilization and Histamine Release in RBL-2H3 Cells
- Antitumorigenic activities of linoleic acid detected by in situ hybridization on transplanted tumors in mice
- GPR40 Agonism Modulates Inflammatory Reactions in Vascular Endothelial Cells