Korean J Physiol Pharmacol.
2009 Feb;13(1):1-7. 10.4196/kjpp.2009.13.1.1.
Changes of Atrial Natriuretic Peptide System in Rats with Puromycin Aminonucleoside-Induced Nephrotic Syndrome
- Affiliations
-
- 1Department of Internal Medicine, Chonnam National University Medical School, Gwangju 501-757, Korea. skimw@chonnam.ac.kr
- 2Department of Physiology, Chonnam National University Medical School, Gwangju 501-757, Korea.
- KMID: 2285364
- DOI: http://doi.org/10.4196/kjpp.2009.13.1.1
Abstract
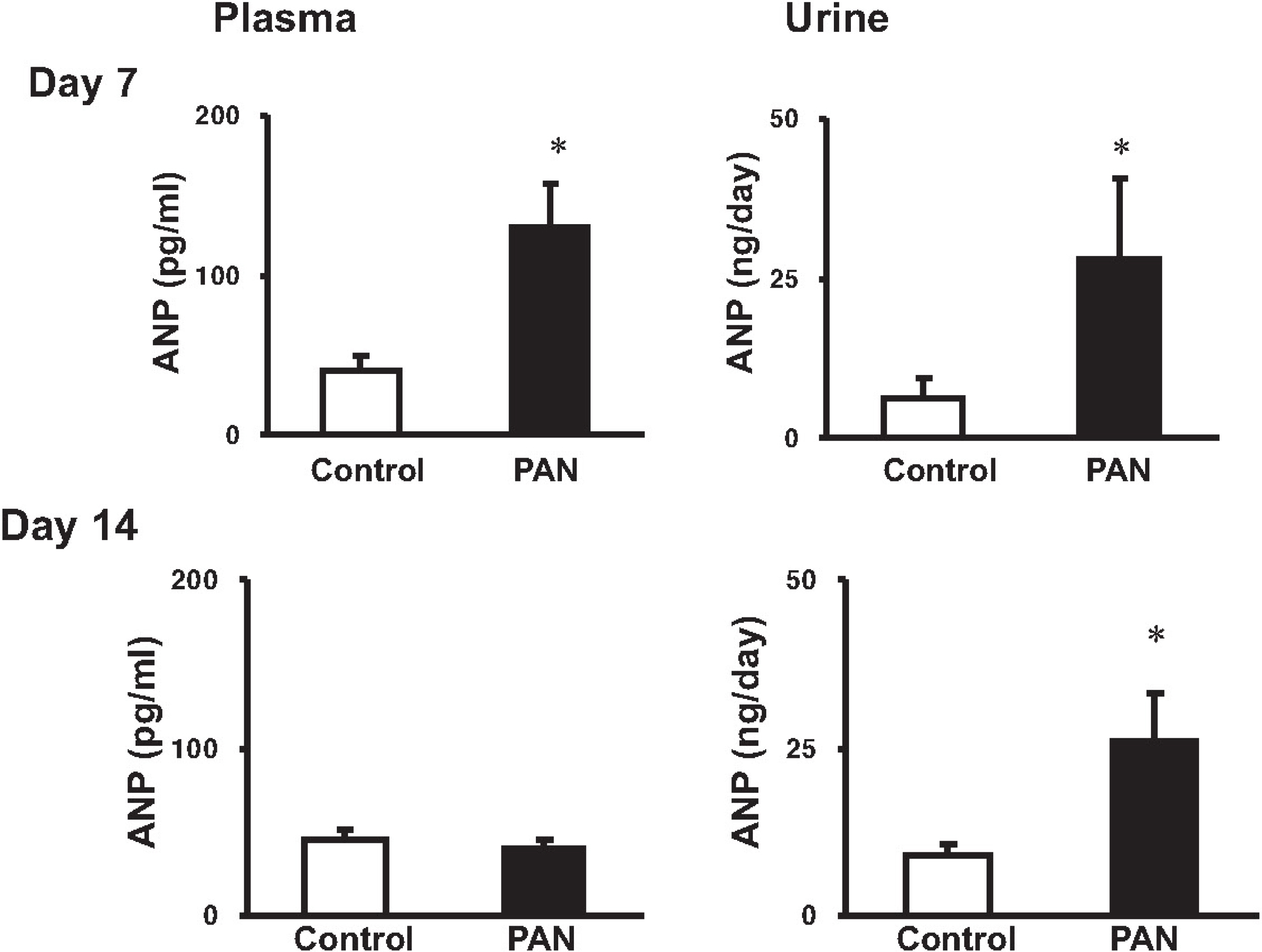
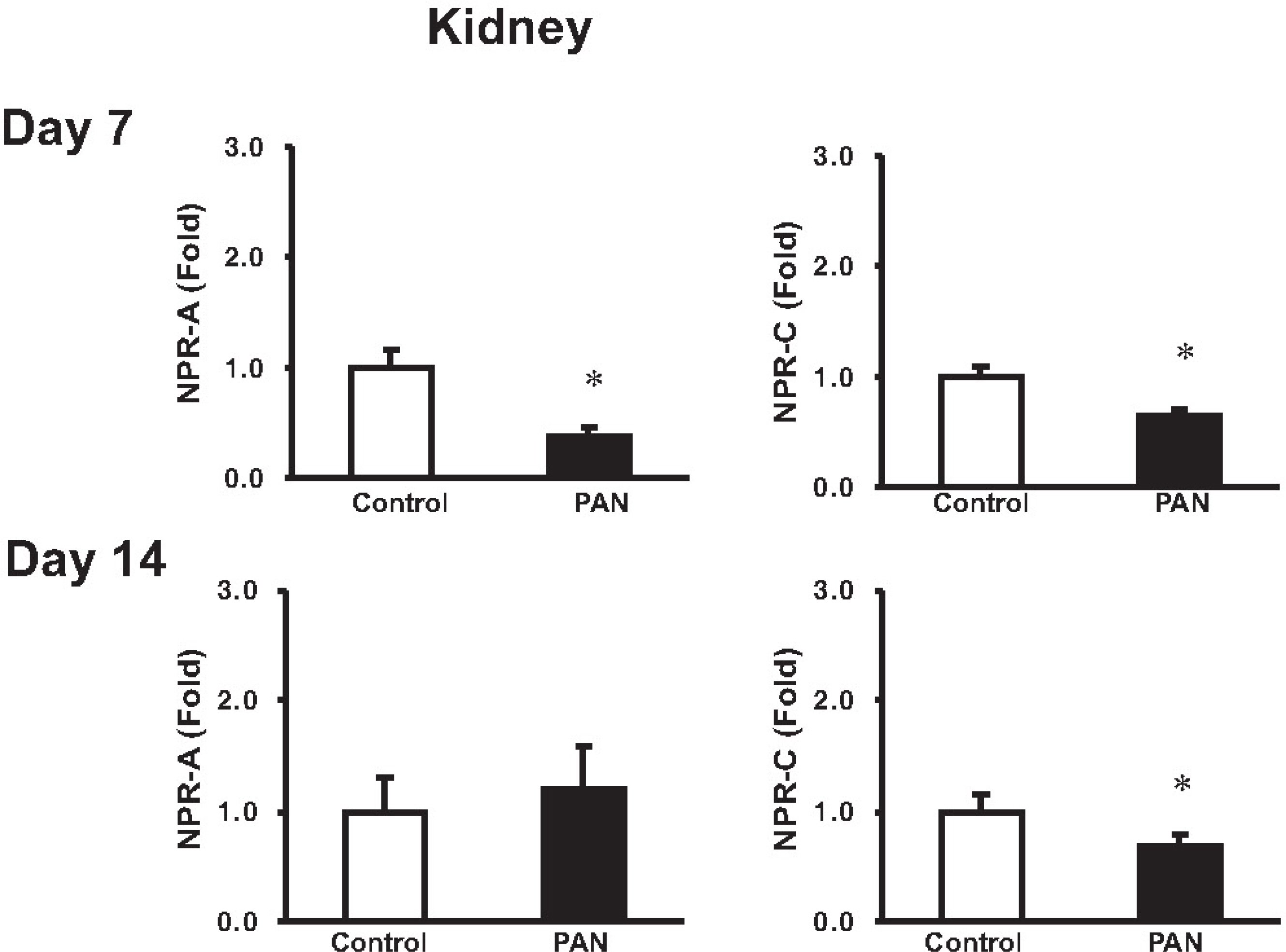
- Sodium retention is a hallmark of nephrotic syndrome. We investigated whether sodium retention is associated with changes of natriuretic peptide system at different stages (i.e., a sodium retaining stage and a compensatory stage) of nephrotic syndrome. At day 7 after PAN (puromycin aminonucleoside) injection, the urinary excretion of sodium was decreased, along with the development of ascites and positive sodium balance. The plasma and urinary ANP (atrial natriuretic peptide) immunoreactivities were increased. ANP mRNA expression was increased in the heart and kidney, whereas that of NPR (natriuretic peptide receptor)-A and NPR-C mRNA was decreased in the kidney. The expression of NEP was decreased in the kidney. At day 14, urinary excretion of sodium did not differ from the control. The plasma ANP level and heart ANP mRNA expression returned to their control values. The expression of ANP mRNA in the kidney was increased in association with increased urinary ANP immunoreactivities. The expression of NPR-A in the kidney became normal, whereas that of NPR-C kept decreased. The expression of NEP (neutral endopeptidase) remained decreased. These findings suggest that the increased renal ANP synthesis in association with decreased metabolism via NEP and NPR-C may play a compensatory role against the development of sodium retention in nephrotic syndrome. The decreased of NPR-A expression in the kidney may contribute to the ANP resistance at day 7. The subsequent recovery of NPR-A expression may play a role in promoting sodium excretion in later stage (at day 14).
MeSH Terms
Figure
Reference
-
Deschenes G., Doucet A. Collecting duct Na,K-ATPase activity correlates with urinary sodium excretion in rat nephrotic syndrome. J Am Soc Nephrol. 11:604–615. 2000.Deschenes G., Gonin S., Zolty E., Cheval L., Rousselot M., Martin PY., Verbavatz JM., Feraille E., Doucet A. Increased synthesis and avp unresponsiveness of Na,K-ATPase in collecting duct from nephrotic rats. J Am Soc Nephrol. 12:2241–2252. 2001.Drewett JG., Garbers DL. The family of guanylyl cyclase receptors and their ligands. Endocr Rev. 15:135–162. 1994.
ArticleGoetz KL., Wang BC., Geer PG., Leadley RJ Jr. Reinhardt HW. Atrial stretch increases sodium excretion independently of release of atrial peptides. Am J Physiol. 250:R946–R950. 1986.Ichikawa I., Rennke HG., Hoyer JR., Badr KF., Schor N., Troy JL., Lechene CP., Brenner BM. Role for intrarenal mechanisms in the impaired salt excretion of experimental nephritic syndrome. J Clin Invest. 71:91–103. 1983.Kim SW., Schou UK., Peters CD., de Seigneuxs S., Kwon TH., Knepper MA., Jonassen TE., Frokiaer J., Nielsen S. Increased apical targeting of renal epithelial sodium channel subunits and decreased expression of type 2 11beta-hydroxysteroid dehydrogenase in rats with CCl4-induced decompensated liver cirrhosis. J Am Soc Nephrol. 16:3196–3210. 2005.Kim SW., Wang W., Nielsen J., Praetorius J., Kwon TH., Knepper MA., Frokiaer J., Nielsen S. Increased expression and apical targeting of renal ENaC subunits in puromycin aminonucleoside-induced nephrotic syndrome in rats. Am J Physiol Renal Physiol. 286:F922–F935. 2004.
ArticleKim SW., Wang W., Sassen MC., Choi KC., Han JS., Knepper MA., Jonassen TE., Frokiaer J., Nielsen S. Biphasic changes of epithelial sodium channel abundance and trafficking in common bile duct ligation-induced liver cirrhosis. Kidney Int. 69:89–98. 2006.
ArticleLivak KJ., Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.Parkes DG., Coghlan JP., McDougall JG., Scoggins BA. Long-term hemodynamic actions of atrial natriuretic factor (99-126) in conscious sheep. Am J Physiol. 254:H811–H815. 1988.
ArticlePerico N., Delaini F., Lupini C., Benigni A., Galbusera M., Boccardo P., Remuzzi G. Blunted excretory response to atrial natriuretic peptide in experimental nephrosis. Kidney Int. 36:57–64. 1989.
ArticlePerico N., Remuzzi G. Edema of the nephrotic syndrome: the role of the atrial natriuretic peptide system. Am J Kidney Dis. 22:355–366. 1993.Peterson C., Madsen B., Perlman A., Chan AY., Myers BD. Atrial natriuretic peptide and the renal response to hypervolemia in nephrotic humans. Kidney Int. 34:825–831. 1988.
ArticleSinger DR., Shore AC., Markandu ND., Buckley MG., Sagnella GA., MacGregor GA. Dissociation between plasma atrial natriuretic peptide levels and urinary sodium excretion after intravenous saline infusion in normal man. Clin Sci. 73:285–289. 1987.
ArticleValentin JP., Qiu C., Muldowney WP., Ying WZ., Gardner DG., Humphreys NH. Cellular basis for blunted volume expansion natriuresis in experimental nephrotic syndrome. J Clin Invest. 90:1302–1312. 1992.
ArticleVande Walle JG., Donckerwolcke RA. Pathogenesis of edema formation in the nephrotic syndrome. Pediatr Nephrol. 16:283–293. 2001.
ArticleWilkins MR., Redondo J., Brown LA. The natriuretic-peptide family. Lancet. 349:1307–1310. 1997.
ArticleWoolf AS., Lyon TL., Hoffbrand BI., Cohen SL., Moult PJ. Effects of physiological infusion of atrial natriuretic factor on healthy subjects and patients with the nephrotic syndrome. Nephron. 52:244–250. 1989.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Changes in Endothelin Receptor Type B and Neuronal Nitric Oxide Synthase in Puromycin Aminonucleoside-Induced Nephrotic Syndrome
- Altered Expression of Renal AQP1, AQP2 and NHE3 in Puromycin Aminonucleoside Induced Nephrotic Syndrome: Intervention by Alpha-MSH Treatment
- The Effects of Selective Cyclooxygenase-2 Inhibition in Puromycin Aminonucleoside Nephropathy Rats
- The effects of lovastatin on puromycin aminonucleoside-induced focal segmental glomerulosclerosis in rats
- Effects of puromycin aminonucleoside on the cytoskeletal changes of glomerular epithelial cells