Clin Nutr Res.
2013 Jan;2(1):67-75. 10.7762/cnr.2013.2.1.67.
The Association of Metabolic Syndrome and Serum gamma-Glutamyl Transpeptidase: A 4-Year Cohort Study of 3,698 Korean Male Workers
- Affiliations
-
- 1Department of Medical Nutrition, Graduate School of East-West Medical Science, Kyung Hee University, Yongin 446-791, Korea. ypark@khu.ac.kr
- 2Institute of Medical Nutrition, Kyung Hee University, Seoul 137-701, Korea.
- KMID: 2279717
- DOI: http://doi.org/10.7762/cnr.2013.2.1.67
Abstract
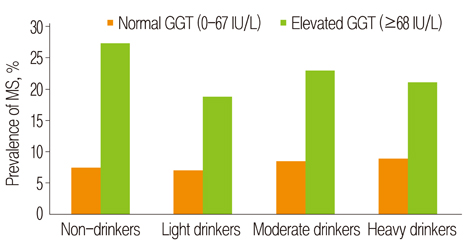
- The aim of the present study was to examine the causal-effect of baseline (year 2004) serum gamma-glutamyl transpeptidase (GGT) level with the prevalence of metabolic syndrome (MS) in year 2008. The study was comprised of male workers who underwent a regular health check-up in 2004 and 2008. MS was diagnosed according to the American Association of Clinical Endocrinologists (AACE) criteria. In the subgroup analysis according to serum GGT level, triglyceride (TG), low density lipoprotein cholesterol (LDL-C), and total cholesterol (TC) showed a significant increasing tendency (p < 0.001). In addition, unexpectedly results were consistent in non-drinkers (p < 0.001). GGT level was significantly associated with risk factors of MS (waist circumference [WC]: r = 0.18, p < 0.001; fasting blood glucose [FBG]: r = 0.16, p < 0.001; TG: r = 0.29, p < 0.001). As the secondary biomarker, homeostasis model assessment of insulin sensitivity (HOMA-S) and TC had significant correlations with GGT level (HOMA-S: r = -0.14, p < 0.001; TC: r = 0.21, p < 0.001). In the 4-year prospective analysis, the predictive effect of baseline GGT concentrations on change in MS status was evaluated using Cox proportional model. Elevated GGT concentrations measured in 2004 were associated with the risk of MS incidence after 4 years (GGT: HR 1.7 [95% CI: 1.2-2.3]) (p < 0.01). This observation indicates that an elevated GGT level could be suggested as a subsidiary marker for MS and partially reflects dyslipidemia as a component of MS.
MeSH Terms
Figure
Cited by 1 articles
-
Association between Serum Gamma-Glutamyltransferase and Prevalence of Metabolic Syndrome Using Data from the Korean Genome and Epidemiology Study
Mi Young Lee, Dae Sung Hyon, Ji Hye Huh, Hae Kyung Kim, Sul Ki Han, Jang Young Kim, Sang Baek Koh
Endocrinol Metab. 2019;34(4):390-397. doi: 10.3803/EnM.2019.34.4.390.
Reference
-
1. Mozumdar A, Liguori G. Persistent increase of prevalence of metabolic syndrome among U.S. adults: NHANES III to NHANES 1999-2006. Diabetes Care. 2011; 34:216–219.
Article2. Statistics Korea. The annual report on the cause of death statistics. 2000. cited 2012 August 28. Available from http://kosis.kr/metadata/main.jsp?surv_id=19&curYear=2010.3. Aso Y, Wakabayashi S, Yamamoto R, Matsutomo R, Takebayashi K, Inukai T. Metabolic syndrome accompanied by hypercholesterolemia is strongly associated with proinflammatory state and impairment of fibrinolysis in patients with type 2 diabetes: synergistic effects of plasminogen activator inhibitor-1 and thrombin-activatable fibrinolysis inhibitor. Diabetes Care. 2005; 28:2211–2216.
Article4. Freiberg MS, Cabral HJ, Heeren TC, Vasan RS, Curtis Ellison R. Third National Health and Nutrition Examination Survey. Alcohol consumption and the prevalence of the Metabolic Syndrome in the US.: a cross-sectional analysis of data from the Third National Health and Nutrition Examination Survey. Diabetes Care. 2004; 27:2954–2959.
Article5. Yoon YS, Oh SW, Baik HW, Park HS, Kim WY. Alcohol consumption and the metabolic syndrome in Korean adults: the 1998 Korean National Health and Nutrition Examination Survey. Am J Clin Nutr. 2004; 80:217–224.
Article6. Fraser A, Harris R, Sattar N, Ebrahim S, Smith GD, Lawlor DA. Gamma-glutamyltransferase is associated with incident vascular events independently of alcohol intake: analysis of the British Women's Heart and Health Study and Meta-Analysis. Arterioscler Thromb Vasc Biol. 2007; 27:2729–2735.
Article7. Miranda PJ, DeFronzo RA, Califf RM, Guyton JR. Metabolic syndrome: definition, pathophysiology, and mechanisms. Am Heart J. 2005; 149:33–45.
Article8. Wannamethee SG, Shaper AG, Lennon L, Whincup PH. Hepatic enzymes, the metabolic syndrome, and the risk of type 2 diabetes in older men. Diabetes Care. 2005; 28:2913–2918.
Article9. André P, Balkau B, Born C, Royer B, Wilpart E, Charles MA, Eschwège E. Hepatic markers and development of type 2 diabetes in middle aged men and women: a three-year follow-up study. The D.E.S.I.R. Study (Data from an Epidemiological Study on the Insulin Resistance syndrome). Diabetes Metab. 2005; 31:542–550.
Article10. Gigleux I, Gagnon J, St-Pierre A, Cantin B, Dagenais GR, Meyer F, Després JP, Lamarche B. Moderate alcohol consumption is more cardioprotective in men with the metabolic syndrome. J Nutr. 2006; 136:3027–3032.
Article11. Lieber CS. Alcohol and the liver: 1984 update. Hepatology. 1984; 4:1243–1260.
Article12. Miller PM, Anton RF, Egan BM, Basile J, Nguyen SA. Excessive alcohol consumption and hypertension: clinical implications of current research. J Clin Hypertens (Greenwich). 2005; 7:346–351.
Article13. Nyblom H, Berggren U, Balldin J, Olsson R. High AST/ALT ratio may indicate advanced alcoholic liver disease rather than heavy drinking. Alcohol Alcohol. 2004; 39:336–339.
Article14. Westin J, Lagging LM, Spak F, Aires N, Svensson E, Lindh M, Dhillon AP, Norkrans G, Wejstål R. Moderate alcohol intake increases fibrosis progression in untreated patients with hepatitis C virus infection. J Viral Hepat. 2002; 9:235–241.
Article15. Alatalo PI, Koivisto HM, Hietala JP, Puukka KS, Bloigu R, Niemelä OJ. Effect of moderate alcohol consumption on liver enzymes increases with increasing body mass index. Am J Clin Nutr. 2008; 88:1097–1103.
Article16. Anton RF, Stout RL, Roberts JS, Allen JP. The effect of drinking intensity and frequency on serum carbohydrate-deficient transferrin and γ-glutamyl transferase levels in outpatient alcoholics. Alcohol Clin Exp Res. 1998; 22:1456–1462.
Article17. Nam SM, Yu HY, Lee MY, Koh JH, Shin JY, Shin YG, Chung CH. Alcohol consumption, liver enzymes, and prevalence of metabolic syndrome in Korean adult men. J Korean Diabetes Assoc. 2007; 31:253–260.
Article18. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; 18:499–502.
Article19. Yates AP, Laing I. Age-related increase in haemoglobin A1c and fasting plasma glucose is accompanied by a decrease in β cell function without change in insulin sensitivity: evidence from a cross-sectional study of hospital personnel. Diabet Med. 2002; 19:254–258.
Article20. Razak F, Anand SS, Shannon H, Vuksan V, Davis B, Jacobs R, Teo KK, McQueen M, Yusuf S. Defining obesity cut points in a multiethnic population. Circulation. 2007; 115:2111–2118.
Article21. Bhushan V, Le TT, Ozturk A, Rao D. First aid for the USMLE step 1 2007: a student to student guide. New York: McGraw-Hill;2006.22. Sato KK, Hayashi T, Nakamura Y, Harita N, Yoneda T, Endo G, Kambe H. Liver enzymes compared with alcohol consumption in predicting the risk of type 2 diabetes: the Kansai Healthcare Study. Diabetes Care. 2008; 31:1230–1236.
Article23. Lee DH, Ha MH, Kim JH, Christiani DC, Gross MD, Steffes M, Blomhoff R, Jacobs DR Jr. Gamma-glutamyltransferase and diabetes--a 4 year follow-up study. Diabetologia. 2003; 46:359–364.
Article24. Lee DH, Silventoinen K, Jacobs DR Jr, Jousilahti P, Tuomileto J. γ-glutamyltransferase, obesity, and the risk of type 2 diabetes: observational cohort study among 20,158 middle-aged men and women. J Clin Endocrinol Metab. 2004; 89:5410–5414.
Article25. Poikolainen K, Vartiainen E. Determinants of γ-glutamyltransferase: positive interaction with alcohol and body mass index, negative association with coffee. Am J Epidemiol. 1997; 146:1019–1024.
Article26. Mukamal KJ, Conigrave KM, Mittleman MA, Camargo CA Jr, Stampfer MJ, Willett WC, Rimm EB. Roles of drinking pattern and type of alcohol consumed in coronary heart disease in men. N Engl J Med. 2003; 348:109–118.
Article27. Lee DH, Jacobs DR Jr, Gross M, Kiefe CI, Roseman J, Lewis CE, Steffes M. γ-glutamyltransferase is a predictor of incident diabetes and hypertension: the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Clin Chem. 2003; 49:1358–1366.
Article28. Nakanishi N, Suzuki K, Tatara K. Serum γ-glutamyltransferase and risk of metabolic syndrome and type 2 diabetes in middle-aged Japanese men. Diabetes Care. 2004; 27:1427–1432.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Association between Serum Gamma-Glutamyltransferase within Normal Levels and Metabolic Syndrome in Office Workers: A 4-Year Follow-up Study
- The health effects of low blood lead level in oxidative stress as a marker, serum gamma-glutamyl transpeptidase level, in male steelworkers
- The Change of Glutathione Content in the Placenta of Patients with Pregnancy-induced Hypertension
- The Association between Gamma-glutamyltransferase and Metabolic Syndrome in Korean Male Workers
- Induction of Rat Liver gamma-Glutamyl Transpeptidase by Bile Acid Load