Korean J Obstet Gynecol.
2010 Jun;53(6):475-488. 10.5468/kjog.2010.53.6.475.
Fetal origins of adult disease
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Soonchunhyang University College of Medicine, Seoul, Korea. kychoi@hosp.sch.ac.kr
- KMID: 2273887
- DOI: http://doi.org/10.5468/kjog.2010.53.6.475
Abstract
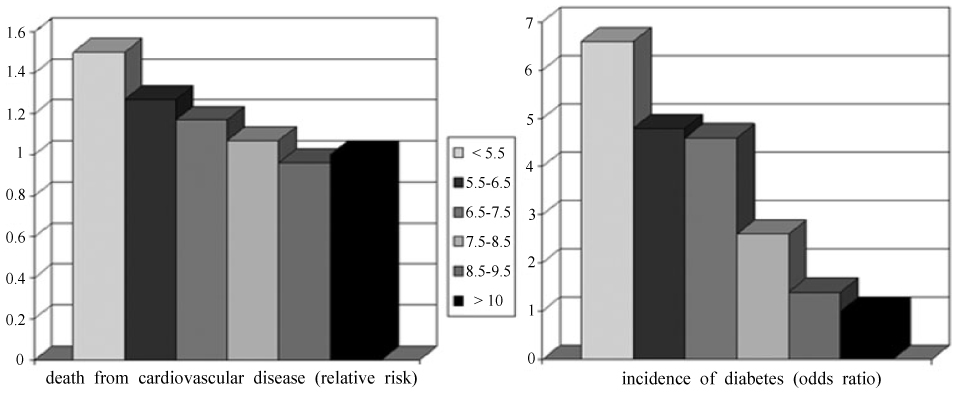
- Human epidemiological and animal studies show that many chronic adult disease have their antecedents in compromised fetal and early postnatal development. Developmental programming is defined as the response by the developing mammalian organism to a specific challenge during a critical time window that alters the trajectory of development with resulting persistent effects on phenotype. Each individual's phenotype is influenced by the developmental environment as much as their genes. Also, the term 'fetal origins of adult disease' was coined on the basis of the inverse association between low birth weight and blood pressure, adult-onset diabetes, coronary heart disease, and stroke seen in numerous epidemiological studies. However, it seems unlikely that birth weight is involved in causal pathways underlying theses observations, and if it were then the significance to public health of these findings is very limited because of our inability to modify birth weight to a relevant extent in humans. There has been a major focus on maternal nutrition. Important targets for future research include distinction between the effects of different pregnancy conditions, such as maternal malnutrition, preeclampsia, and maternal infection, which may have dissimilar late-life consequences. This will be a crucial step when the associations that are currently emerging will be translated into disease prevention.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Health Promoting Behavior in Pregnant Couples: Actor-Partner Interdependence Model Analysis
Ju Hee Kim, Young-A Song
J Korean Acad Nurs. 2017;47(4):467-475. doi: 10.4040/jkan.2017.47.4.467.
Reference
-
1. Dorner G. Stumpf WE, Grant LD, editors. Perinatal hormone levels and brain organization. Anatomical neuroendocrinology. 1975. Basel: S. Karger;245–252.2. Forsdahl A. Are poor living conditions in childhood and adolescence an important risk factor for arteriosclerotic heart disease? Br J Prev Soc Med. 1977. 31:91–95.3. Barker DJP. Mothers, babies and health in later life. 1998. 2nd ed. Edinburgh: Churchill Livingstone.4. Hales CN, Barker DJ. The thrifty phenotype hypothesis. Br Med Bull. 2001. 60:5–20.5. Nijland MJ, Ford SP, Nathanielsz PW. Prenatal origins of adult disease. Curr Opin Obstet Gynecol. 2008. 20:132–138.6. Freathy RM, Weedon MN, Bennett A, Hypponen E, Relton CL, Knight B, et al. Type 2 diabetes TCF7L2 risk genotypes alter birth weight: a study of 24,053 individuals. Am J Hum Genet. 2007. 80:1150–1161.7. Schubeler D, Lorincz MC, Cimbora DM, Telling A, Feng YQ, Bouhassira EE, et al. Genomic targeting of methylated DNA: influence of methylation on transcription, replication, chromatin structure, and histone acetylation. Mol Cell Biol. 2000. 20:9103–9112.8. Hanson MA, Gluckman PD. Developmental origins of health and disease: new insights. Basic Clin Pharmacol Toxicol. 2008. 102:90–93.9. Rinaudo PF, Lamb J. Fetal origins of perinatal morbidity and/or adult disease. Semin Reprod Med. 2008. 26:436–445.10. Godfrey KM, Barker DJ. Fetal programming and adult health. Public Health Nutr. 2001. 4:611–624.11. Gluckman PD, Hanson MA. Developmental plasticity and human disease: research directions. J Intern Med. 2007. 261:461–471.12. Barker DJ, Winter PD, Osmond C, Margetts B, Simmonds SJ. Weight in infancy and death from ischaemic heart disease. Lancet. 1989. 2:577–580.13. Osmond C, Barker DJ, Winter PD, Fall CH, Simmonds SJ. Early growth and death from cardiovascular disease in women. BMJ. 1993. 307:1519–1524.14. Hales CN, Barker DJ, Clark PM, Cox LJ, Fall C, Osmond C, et al. Fetal and infant growth and impaired glucose tolerance at age 64. BMJ. 1991. 303:1019–1022.15. Newsome CA, Shiell AW, Fall CH, Phillips DI, Shier R, Law CM. Is birth weight related to later glucose and insulin metabolism?-A systematic review. Diabet Med. 2003. 20:339–348.16. Barker DJ, Eriksson JG, Forsen T, Osmond C. Fetal origins of adult disease: strength of effects and biological basis. Int J Epidemiol. 2002. 31:1235–1239.17. Keller G, Zimmer G, Mall G, Ritz E, Amann K. Nephron number in patients with primary hypertension. N Engl J Med. 2003. 348:101–108.18. Phillips DI. Insulin resistance as a programmed response to fetal undernutrition. Diabetologia. 1996. 39:1119–1122.19. Barker DJ, Forsen T, Uutela A, Osmond C, Eriksson JG. Size at birth and resilience to effects of poor living conditions in adult life: longitudinal study. BMJ. 2001. 323:1273–1276.20. Marmot M, Wilkinson RG. Psychosocial and material pathways in the relation between income and health: a response to Lynch et al. BMJ. 2001. 322:1233–1236.21. Phillips DI, Walker BR, Reynolds RM, Flanagan DE, Wood PJ, Osmond C, et al. Low birth weight predicts elevated plasma cortisol concentrations in adults from 3 populations. Hypertension. 2000. 35:1301–1306.22. Painter RC, Roseboom TJ, Bleker OP. Prenatal exposure to the Dutch famine and disease in later life: an overview. Reprod Toxicol. 2005. 20:345–352.23. Vonnahme KA, Hess BW, Hansen TR, McCormick RJ, Rule DC, Moss GE, et al. Maternal undernutrition from early-to mid-gestation leads to growth retardation, cardiac ventricular hypertrophy, and increased liver weight in the fetal sheep. Biol Reprod. 2003. 69:133–140.24. Snoeck A, Remacle C, Reusens B, Hoet JJ. Effect of a low protein diet during pregnancy on the fetal rat endocrine pancreas. Biol Neonate. 1990. 57:107–118.25. Latini G, De Mitri B, Del Vecchio A, Chitano G, De Felice C, Zetterstrom R. Foetal growth of kidneys, liver and spleen in intrauterine growth restriction:"programming" causing "metabolic syndrome" in adult age. Acta Paediatr. 2004. 93:1635–1639.26. Woods LL, Ingelfinger JR, Nyengaard JR, Rasch R. Maternal protein restriction suppresses the newborn renin-angiotensin system and programs adult hypertension in rats. Pediatr Res. 2001. 49:460–467.27. Welham SJ, Riley PR, Wade A, Hubank M, Woolf AS. Maternal diet programs embryonic kidney gene expression. Physiol Genomics. 2005. 22:48–56.28. Dong F, Ford SP, Fang CX, Nijland MJ, Nathanielsz PW, Ren J. Maternal nutrient restriction during early to mid gestation up-regulates cardiac insulin-like growth factor (IGF) receptors associated with enlarged ventricular size in fetal sheep. Growth Horm IGF Res. 2005. 15:291–299.29. Zhu MJ, Ford SP, Nathanielsz PW, Du M. Effect of maternal nutrient restriction in sheep on the development of fetal skeletal muscle. Biol Reprod. 2004. 71:1968–1973.30. Hales CN, Desai M, Ozanne SE, Crowther NJ. Fishing in the stream of diabetes: from measuring insulin to the control of fetal organogenesis. Biochem Soc Trans. 1996. 24:341–350.31. Gluckman PD, Hanson MA. Living with the past: evolution, development, and patterns of disease. Science. 2004. 305:1733–1736.32. Gluckman PD, Hanson MA, Beedle AS. Non-genomic transgenerational inheritance of disease risk. Bioessays. 2007. 29:145–154.33. Rinn JL, Rozowsky JS, Laurenzi IJ, Petersen PH, Zou K, Zhong W, et al. Major molecular differences between mammalian sexes areinvolved in drug metabolism and renal function. Dev Cell. 2004. 6:791–800.34. Ahluwalia A, Clodfelter KH, Waxman DJ. Sexual dimorphism of rat liver gene expression: regulatory role of growth hormone revealed by deoxyribonucleic Acid microarray analysis. Mol Endocrinol. 2004. 18:747–760.35. Gilbert JS, Ford SP, Lang AL, Pahl LR, Drumhiller MC, Babcock SA, et al. Nutrient restriction impairs nephrogenesis in a gender-specific manner in the ovine fetus. Pediatr Res. 2007. 61:42–47.36. West-Eberhard M. Phenotypic plasticity and the origins of diversity. Annu Rev Ecol Syst. 1989. 20:249–278.37. Bateson PPG, Martin PR. Design for a life: how behaviour develops. 1999. London: Jonatha Cape.38. Brooks AA, Johnson MR, Steer PJ, Pawson ME, Abdalla HI. Birth weight: nature or nurture? Early Hum Dev. 1995. 42:29–35.39. McCance RA. Food, growth, and time. Lancet. 1962. 2:621–626.40. Vaag A, Henriksen JE, Madsbad S, Holm N, Beck-Nielsen H. Insulin secretion, insulin action, and hepatic glucose production in identical twins discordant for non-insulin-dependent diabetes mellitus. J Clin Invest. 1995. 95:690–698.41. Froguel P, Zouali H, Vionnet N, Velho G, Vaxillaire M, Sun F, et al. Familial hyperglycemia due to mutations in glucokinase. Definition of a subtype of diabetes mellitus. N Engl J Med. 1993. 328:697–702.42. Hattersley AT, Beards F, Ballantyne E, Appleton M, Harvey R, Ellard S. Mutations in the glucokinase gene of the fetus result in reduced birth weight. Nat Genet. 1998. 19:268–270.43. Munoz J, Lok KH, Gower BA, Fernandez JR, Hunter GR, Lara-Castro C, et al. Polymorphism in the transcription factor 7-like 2 (TCF7L2) gene is associated with reduced insulin secretion in nondiabetic women. Diabetes. 2006. 55:3630–3634.44. Eriksson JG, Lindi V, Uusitupa M, Forsen TJ, Laakso M, Osmond C, et al. The effects of the Pro12Ala polymorphism of the peroxisome proliferator-activated receptor-gamma2 gene on insulin sensitivity and insulin metabolism interact with size at birth. Diabetes. 2002. 51:2321–2324.45. Eriksson JG, Forsen T, Tuomilehto J, Osmond C, Barker DJ. Early growth and coronary heart disease in later life: longitudinal study. BMJ. 2001. 322:949–953.46. Barker DJ. The developmental origins of adult disease. J Am Coll Nutr. 2004. 23:588S–595S.47. Barker DJ, Forsen T, Eriksson JG, Osmond C. Growth and living conditions in childhood and hypertension in adult life: a longitudinal study. J Hypertens. 2002. 20:1951–1956.48. Ravelli GP, Stein ZA, Susser MW. Obesity in young men after famine exposure in utero and early infancy. N Engl J Med. 1976. 295:349–353.49. Clausen JO, Borch-Johnsen K, Pedersen O. Relation between birth weight and the insulin sensitivity index in a population sample of 331 young, healthy Caucasians. Am J Epidemiol. 1997. 146:23–31.50. Flanagan DE, Moore VM, Godsland IF, Cockington RA, Robinson JS, Phillips DI. Fetal growth and the physiological control of glucose tolerance in adults: a minimal model analysis. Am J Physiol Endocrinol Metab. 2000. 278:E700–E706.51. Gluckman PD, Hanson MA. Gluckman P, Hanson M, editors. The coneptual basis for the developmental origins of health and disease. Developmental origins of health and disease. 2006. Cambridge: Cambridge University Press;33–50.52. Gluckman PD, Hanson MA, Spencer HG, Bateson P. Environmental influences during development and their later consequences for health and disease: implications for the interpretation of empirical studies. Proc Biol Sci. 2005. 272:671–677.53. Law CM, Shiell AW. Is blood pressure inversely related to birth weight? The strength of evidence from a systematic review of the literature. J Hypertens. 1996. 14:935–941.54. Leon DA, Koupilova I, Lithell HO, Berglund L, Mohsen R, Vagero D, et al. Failure to realise growth potential in utero and adult obesity in relation to blood pressure in 50 year old Swedish men. BMJ. 1996. 312:401–406.55. Rich-Edwards JW, Stampfer MJ, Manson JE, Rosner B, Hankinson SE, Colditz GA, et al. Birth weight and risk of cardiovascular disease in a cohort of women followed up since 1976. BMJ. 1997. 315:396–400.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Fetal Programming and Adult Disease
- What is fetal programming?: A lifetime health is under the control of in-utero health
- What is fetal programming?: a lifetime health is under the control of in utero health
- A Case of Fetal Supraventricular Tachycardia with Fetal Ascites Treated by Antiarrhythmic Agent
- Clinical study on fetal heart rate and fetal movement reponse to music sound stimulation