Cancer Res Treat.
2005 Jun;37(3):157-164.
Assessment of Tumor Regression by Consecutive Pelvic Magnetic Resonance Imaging and Dose Modification during High- Dose-Rate Brachytherapy for Carcinoma of the Uterine Cervix
- Affiliations
-
- 1Department of Radiation Oncology, Chonnam National University Medical School, Gwangju, Korea. tknam@chonnam.ac.kr
- 2Department of Obstetrics and Gynecology, Chonnam National University Medical School, Gwangju, Korea.
Abstract
- PURPOSE
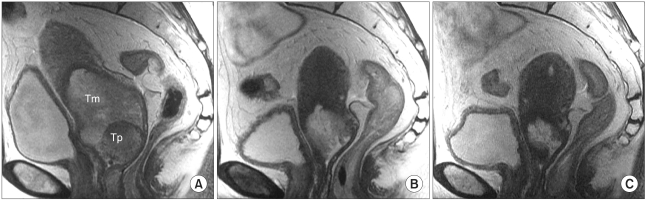
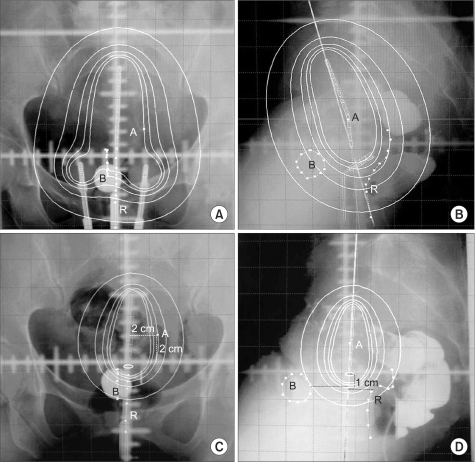
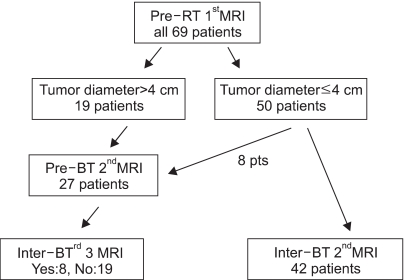
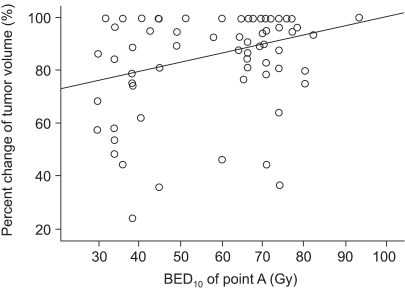
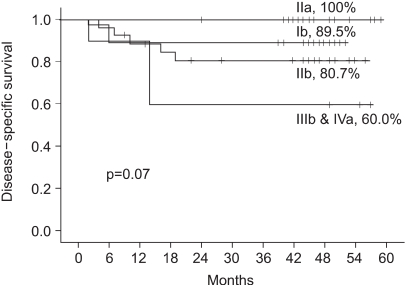
To assess tumor regression, as determined by pelvic magnetic resonance imaging (MRI), and evaluate the efficacies and toxicities of the interim brachytherapy (BT) modification method, according to tumor regression during multi-fractionated high-dose-rate (HDR) BT for uterine cervical cancer. MATERIALS AND METHODS: Consecutive MRI studies were performed pre-radiotherapy (RT), pre-BT and during interfraction of BT (inter-BT) in 69 patients with cervical cancer. External beam radiotherapy (EBRT) was performed, using a 10 MV X-ray, in daily fraction of 1.8 Gy with 4-fields, 5 d/wk. Radiation was delivered up to 50.4 Gy, with midline shielding at around 30.6 Gy. Of all 69 patients, 50 received modified interim BT after checking the inter-BT MRI. The BT was delivered in two sessions; the first was composed of several 5 Gy fractions to point A, twice weekly, using three channel applicators. According to the three measured orthogonal diameters of the regressed tumor, based on inter-BT MR images, the initial BT plan was modified, with the second session consisting of a few fractions of less than 5 Gy to point A, using a cervical cylinder applicator. RESULTS: The numbers of patients in FIGO stages Ib, IIa, IIb and IIIb+IVa were 19 (27.5%), 18 (26.1%), 27 (39.2%) and 5 (7.2%), respectively. Our treatment characteristics were comparable to those from the literatures with respect to the biologically effective dose (BED) to point A, rectum and bladder as reference points. In the regression analysis a significant correlation was observed between tumor regression and the cumulative dose to point A on the follow-up MRI. Nearly 80% regression of the initial tumor volume occurred after 30.6 Gy of EBRT, and this increased to 90% after an additional 25 Gy in 5 fractions of BT, which corresponds to 73.6 Gy of cumulative BED10 to point A. The median total fraction number, and those at the first and second sessions of BT were 8 (5~10), 5 (3~7) and 3 (1~5), respectively. The median follow-up time was 53 months (range, 9~66 months). The 4-year disease-free survival rate of all patients was 86.8%. Six (8.7%) patients developed pelvic failures, but major late complications developed in only two (2.9%). CONCLUSION: Our study shows that effective tumor control, equivalent survival and low rates of major complications can be achieved by modifying the fraction size during BT according to tumor regression, as determined by consecutive MR images. We recommend checking the follow-up MRI at a cumulative BED10 of around 65 Gy to point A, with the initial BT modified at a final booster BT session.
Keyword
MeSH Terms
Figure
Reference
-
1. Hricak H, Powell CB, Yu KK, Washington E, Subak LL, Stern JL, et al. Invasive cervical carcinoma: role of MR imaging in pretreatment work-up--cost minimization and diagnostic efficacy analysis. Radiology. 1996; 198:403–409. PMID: 8596840.
Article2. Krempien RC, Daeuber S, Hensley FW, Wannenmacher M, Harms W. Image fusion of CT and MRI data enables improved target volume definition in 3D-brachytherapy treatment planning. Brachytherapy. 2003; 2:164–171. PMID: 15062139.
Article3. Thomas L, Chacon B, Kind M, Lasbareilles O, Muyldermans P, Chemin A, et al. Magnetic resonance imaging in the treatment planning of radiation therapy in carcinoma of the cervix treated with the four-field pelvic technique. Int J Radiat Oncol Biol Phys. 1997; 37:827–832. PMID: 9128958.
Article4. Wachter-Gerstner N, Wachter S, Reinstadler E, Fellner C, Knocke TH, Potter R. The impact of sectional imaging on dose escalation in endocavitary HDR-brachytherapy of cervical cancer: results of a prospective comparative trial. Radiother Oncol. 2003; 68:51–59. PMID: 12885452.
Article5. Hatano K, Sekiya Y, Araki H, Sakai M, Togawa T, Narita Y, et al. Evaluation of the therapeutic effect of radiotherapy on cervical cancer using magnetic resonance imaging. Int J Radiat Oncol Biol Phys. 1999; 45:639–644. PMID: 10524417.
Article6. Gong QY, Tan LT, Romaniuk CS, Jones B, Brunt JN, Roberts N. Determination of tumour regression rates during radiotherapy for cervical carcinoma by serial MRI: comparison of two measurement techniques and examination of intraobserver and interobserver variability. Br J Radiol. 1999; 72:62–72. PMID: 10341691.
Article7. Flueckiger F, Ebner F, Poschauko H, Tamussino K, Einspieler R, Ranner G. Cervical cancer: serial MR imaging before and after primary radiation therapy--a 2-year follow-up study. Radiology. 1992; 184:89–93. PMID: 1609108.
Article8. Kodaira T, Fuwa N, Toita T, Nomoto Y, Kuzuya K, Tachibana K, et al. Clinical evaluation using magnetic resonance imaging for patients with stage III cervical carcinoma treated by radiation alone in multicenter analysis: its usefulness and limitations in clinical practice. Am J Clin Oncol. 2003; 26:574–583. PMID: 14663375.9. Wagenaar HC, Trimbos JB, Postema S, Anastasopoulou A, van der Geest RJ, Reiber JH, et al. Tumor diameter and volume assessed by magnetic resonance imaging in the prediction of outcome for invasive cervical cancer. Gynecol Oncol. 2001; 82:474–482. PMID: 11520143.
Article10. Huh SJ, Park W, Han Y. Interfractional variation in position of the uterus during radical radiotherapy for cervical cancer. Radiother Oncol. 2004; 71:73–79. PMID: 15066298.
Article11. ICRU 38. Dose and volume specification for reporting intracavitary therapy in gynecology. 1985. International commission on radiation units and measurements;p. 1–20.12. Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995; 31:1341–1346. PMID: 7713792.
Article13. Kim RY, Meyer JT, Plott WE, Spencer SA, Meredith RF, Jennelle RL, et al. Major geometric variations between multiple high-dose-rate applications of brachytherapy in cancer of the cervix: frequency and types of variation. Radiology. 1995; 195:419–422. PMID: 7724760.
Article14. Hoskin PJ, Cook M, Bouscale D, Cansdale J. Changes in applicator position with fractionated high dose rate gynaecological brachytherapy. Radiother Oncol. 1996; 40:59–62. PMID: 8844889.
Article15. Huh SJ. Current status of high dose rate brachytherapy in cervical cancer in Korea and optimal treatment schedule. J Korean Soc Ther Radiol Oncol. 1998; 16:357–366.16. Nag S, Cardenes H, Chang S, Das IJ, Erickson B, Ibbott GS, et al. Proposed guidelines for image-based intracavitary brachytherapy for cervical carcinoma: report from Image-Guided Brachytherapy Working Group. Int J Radiat Oncol Biol Phys. 2004; 60:1160–1172. PMID: 15519788.
Article17. Perez CA, Slessinger E, Grigsby PW. Design of an afterloading vaginal applicator (MIRALVA). Int J Radiat Oncol Biol Phys. 1990; 18:1503–1508. PMID: 2370200.
Article18. Wolfson AH, Wu X, Takita C, Shao H, Luo C, Watzich M, et al. A novel applicator for low-dose-rate brachytherapy of gynecological cancers. Int J Gynecol Cancer. 2003; 13:532–540. PMID: 12911734.
Article19. Mayr NA, Yuh WT, Zheng J, Ehrhardt JC, Sorosky JI, Magnotta VA, et al. Tumor size evaluated by pelvic examination compared with 3-D quantitative analysis in the prediction of outcome for cervical cancer. Int J Radiat Oncol Biol Phys. 1997; 39:395–404. PMID: 9308943.20. Lee SW, Suh CO, Chung EJ, Kim GE. Dose optimization of fractionated external adiation and high-dose-rate intracavitary brachytherapy for FIGO stage IB uterine cervical carcinoma. Int J Radiat Oncol Biol Phys. 2002; 52:1338–1344. PMID: 11955747.21. Clark BG, Souhami L, Roman TN, Chappell R, Evans MD, Fowler JF. The prediction of late rectal complications in patients treated with high dose-rate brachytherapy for carcinoma of the cervix. Int J Radiat Oncol Biol Phys. 1997; 38:989–993. PMID: 9276363.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pelvic MRI Application to the Dosimetric Analysis in Brachytherapy of Uterine Cervix Carcinoma
- Accuracy of Dose Estimation in High Dose Rate Intracavitary Radiotherapy of Carcinoma of the Uterine Cervix
- MRI Findings of Small Cell Neuroendocrine Carcinoma of the Uterine Cervix: A Case Report
- Use of transrectal ultrasound for high dose rate interstitial brachytherapy for patients of carcinoma of uterine cervix
- A Prospective Randomized Study on Two Dose Fractionation Regimens of High-Dose-Rate Brachytherapy for Carcinoma of the Uterine Cervix: Comparison of Efficacies and Toxicities Between Two Regimens