Allergy Asthma Respir Dis.
2013 Sep;1(3):221-226. 10.4168/aard.2013.1.3.221.
Effect of infosheet for topical tacrolimus 0.1% and its efficacy and compliance in the treatment of atopic dermatitis
- Affiliations
-
- 1Department of Dermatology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. chwon98@chol.com
- 2Department of Dermatology, Hanynag University College of Medicine, Seoul, Korea.
- 3Department of Dermatology, Kangdong Sacred Heart Hospital, Hallym University College of Medicine, Seoul, Korea.
- 4Department of Dermatology, Korea University College of Medicine, Seoul, Korea.
- 5Department of Dermatology, Kyung Hee University School of Medicine, Seoul, Korea.
- 6Department of Dermatology, Kangbuk Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 7Department of Dermatology, Yonsei University College of Medicine, Seoul, Korea.
- 8Department of Dermatology, Inje University Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Korea.
- 9Department of Dermatology, Dankook University College of Medicine, Cheonan, Korea.
- KMID: 2262964
- DOI: http://doi.org/10.4168/aard.2013.1.3.221
Abstract
- PURPOSE
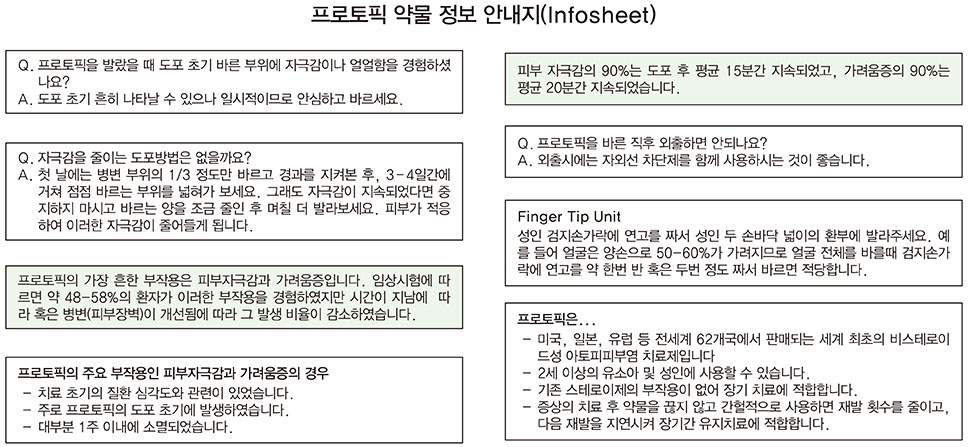
Topical calcineurin inhibitor is recently developed topical immunomodulator, and preliminary studies showed its effectiveness in the treatment of atopic dermatitis (AD). However, some side effects including transient irritation can influence the patient compliance. So, there are some needs to improve the patient compliance. The purpose of this study was to evaluate the efficacy, safety and patient compliance with using topical tacrolimus 0.1% to treat AD when the correct information about topical tacrolimus are properly given to patients.
METHODS
We examined the medical recordings, clinical severity scoring of total 194 AD patients at 9 general hospitals in Seoul, Korea from September 2010 to August 2011. We offered an infosheet of topical tacrolimus 0.1% and the patients applied it twice a day for 2 weeks. And we measured the efficacy of the topical tacrolimus 0.1% with SCORing atopic dermatitis (SCORAD) index, patient's global assessment (PGA), and investigator's global assessment (IGA).
RESULTS
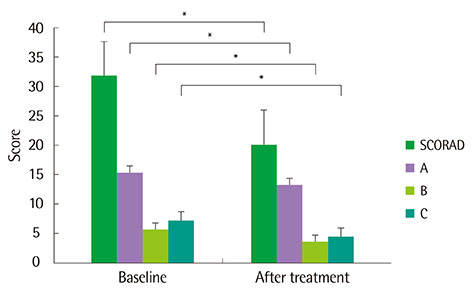
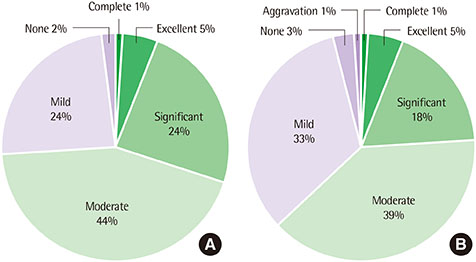
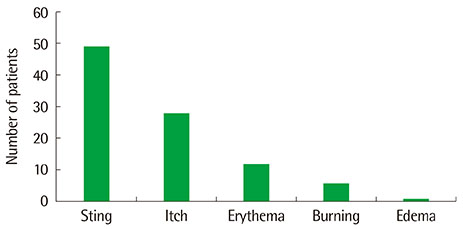
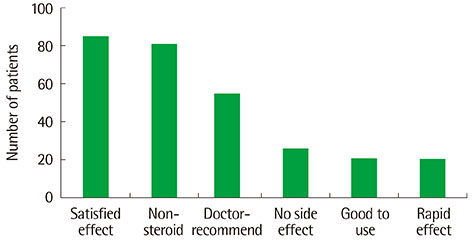
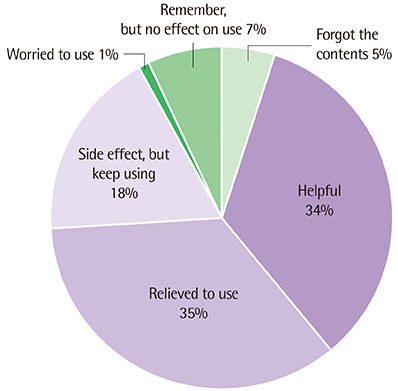
Topical tacrolimus 0.1% effectively controlled AD with a reduction of the SCORAD index from baseline 31.9 to 20.2 at 2 weeks of application. In IGA results showed 98% got improvement and in PGA, results showed 96% got improvement after treatment. Although 42.3% of the patients complained of adverse effects, these were all transient. The effect of information on topical tacrolimus 0.1% showed 34% patients could predict the side effect, 35% patients could feel safety to use, and 18% patients experienced side effect but could maintain topical calcineurin inhibitor.
CONCLUSION
Topical tacrolimus 0.1% may be an effective treatment modality for AD when patients show good compliance for applying the ointment. And properly given, the correct information may increase the patient compliance.
MeSH Terms
Figure
Reference
-
1. Kabashima K. New concept of the pathogenesis of atopic dermatitis: interplay among the barrier, allergy, and pruritus as a trinity. J Dermatol Sci. 2013; 70:3–11.
Article2. Guin JD. Complications of topical hydrocortisone. J Am Acad Dermatol. 1981; 4:417–422.
Article3. Graham-Brown R. Managing adults with atopic dermatitis. Dermatol Clin. 1996; 14:531–537.
Article4. Chase EP, Armstrong AW. Advances in management of atopic dermatitis: new therapies and novel uses of existing treatments. Semin Cutan Med Surg. 2012; 31:17–24.
Article5. Nasr IS. Topical tacrolimus in dermatology. Clin Exp Dermatol. 2000; 25:250–254.
Article6. Gupta AK, Adamiak A, Chow M. Tacrolimus: a review of its use for the management of dermatoses. J Eur Acad Dermatol Venereol. 2002; 16:100–114.
Article7. Katoh N, Hirano S, Yasuno H, Kishimoto S. Effects of tacrolimus ointment on facial eruption, itch, and scratching in patients with atopic dermatitis. J Dermatol. 2004; 31:194–199.
Article8. Hanifin JM, Ling MR, Langley R, Breneman D, Rafal E. Tacrolimus ointment for the treatment of atopic dermatitis in adult patients: part I, efficacy. J Am Acad Dermatol. 2001; 44:1 Suppl. S28–S38.
Article9. Soter NA, Fleischer AB Jr, Webster GF, Monroe E, Lawrence I. Tacrolimus ointment for the treatment of atopic dermatitis in adult patients: part II, safety. J Am Acad Dermatol. 2001; 44:1 Suppl. S39–S46.
Article10. Kapp A, Allen BR, Reitamo S. Atopic dermatitis management with tacrolimus ointment (Protopic). J Dermatolog Treat. 2003; 14:Suppl 1. 5–16.
Article11. Ruzicka T, Bieber T, Schopf E, Rubins A, Dobozy A, Bos JD, et al. European Tacrolimus Multicenter Atopic Dermatitis Study Group. A short-term trial of tacrolimus ointment for atopic dermatitis. N Engl J Med. 1997; 337:816–821.
Article12. Won CH, Seo PG, Park YM, Yang JM, Lee KH, Sung KJ, et al. A multicenter trial of the efficacy and safety of 0.03% tacrolimus ointment for atopic dermatitis in Korea. J Dermatolog Treat. 2004; 15:30–34.
Article13. Choi WW, Seo PG, Kim KH. Tacrolimus ointment; an open study for effects on severe facial atopic dermatitis in Korean. Ann Dermatol. 2002; 14:195–199.
Article14. Kim HO, Park CW, Lee CH, Lee JO. Efficacy of 0.1% Tacrolimus ment in Korean patients with atopic dermatitisoint. Korean J Dermatol. 2005; 43:312–318.15. Luoma R, Koivikko A, Viander M. Development of asthma, allergic rhinitis and atopic dermatitis by the age of five years: a prospective study of 543 newborns. Allergy. 1983; 38:339–346.
Article16. Park YM, Park HJ, Kim TY, Kim CW. The study on the hospital - based relative frequency, and clinical and laboratory findings of atopic dermatitis. Korean J Dermatol. 1997; 35:96–106.17. Hanifin JM, Rajka G. Diagnostic features of atopic dermatitis. Acta Derm Venereol Suppl (Stockh). 1980; Suppl 92. 44–47.18. Williams HC. On the definition and epidemiology of atopic dermatitis. Dermatol Clin. 1995; 13:649–657.
Article19. Lee HJ, Cho SH, Ha SJ, Ahn WK, Park YM, Byun DG, et al. Minor cutaneous features of atopic dermatitis in South Korea. Int J Dermatol. 2000; 39:337–342.
Article20. Kim BJ, Kim MN, Kim KH, Kim DW, Ro YS, Park CW, et al. Multicenter survey of the efficacy and compliance with usingtopical pimecrolimus by patients with atopic dermatitis. Korean J Dermatol. 2008; 46:1357–1361.21. Simpson EL, Hanifin JM. Atopic dermatitis. J Am Acad Dermatol. 2005; 53:115–128.
Article22. Rustin MH. The safety of tacrolimus ointment for the treatment of atopic dermatitis: a review. Br J Dermatol. 2007; 157:861–873.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Four Cases of Atopic Dermatitis with Topical Tacrolimus Therapy
- The Efficacy of Tapering Treatment with 0.1% Tacrolimus Ointment in Adult Patients with Atopic Dermatitis in Korea
- Efficacy of 0.1% Tacrolimus Ointment in Korean Patients with Atopic Dermatitis
- The Effect of Topical Tacrolimus in the Murine Contact Hypersensitivity and Dermatitis of Repeated Applications Induced by Diphenylcyclopropenone
- Tacrolimus ointment: An Open study for Effects on Severe Facial Atopic Dermatitis in Korean