Allergy Asthma Respir Dis.
2015 Jan;3(1):40-46. 10.4168/aard.2015.3.1.40.
Relationships of bronchodilator response with asthma control and fractional exhaled nitric oxide in children with atopic asthma
- Affiliations
-
- 1Department of Pediatrics, Chungbuk National University College of Medicine, Cheongju, Korea. yshahn@chungbuk.ac.kr
- 2Department of Preventive Medicine, Chungbuk National University College of Medicine, Cheongju, Korea.
- KMID: 2262275
- DOI: http://doi.org/10.4168/aard.2015.3.1.40
Abstract
- PURPOSE
Because bronchodilator response (BDR) is variable among asthmatic patients, there are practical limitations in using BDR to assess asthma control. We investigated the relationships of BDR with asthma control status and fractional exhaled nitric oxide (FeNO) in children with atopic asthma.
METHODS
One hundred ninety-one patients aged 8 to 16 years with atopic asthma were enrolled. Pulmonary function tests including BDR and FeNO were serially measured 10 times or more over 2 years when subjects were not receiving controller medications. During the last year of follow-up, the loss of asthma control was assessed in all participants.
RESULTS
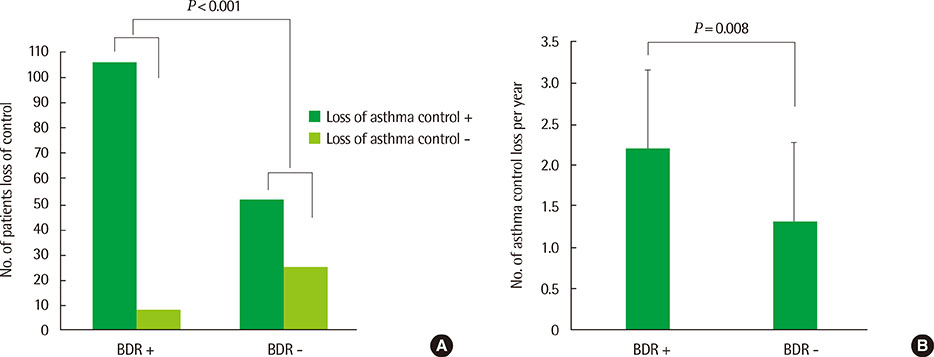
We identified 114 children (60%) with at least 1 positive BDR (> or =12%) over the 2-year observation period. Higher levels of BDRs and higher rates of positive BDRs were associated with lower lung function and lower methacholine PC20 (provocative concentration of methacholine causing a 20% fall in forced expiratory volume in one second). The loss of asthma control occurred in 106 of individuals (93%) who had positive BDRs, as compared to 52 of 77 (68%) with negative BDRs (P<0.001). There was no difference in FeNO levels between individuals with positive and negative BDRs. However, among children with negative BDRs, those developing the loss of asthma control had higher maximal FeNO levels and higher rates of FeNO>21 parts per billion than those who maintained asthma control (all P<0.001).
CONCLUSION
Positive BDRs are linked to a higher probability of asthma control loss in children with atopic asthma. In addition, high FeNO is associated with asthma control loss in asthmatic children with negative BDRs.
Keyword
MeSH Terms
Figure
Reference
-
1. Masoli M, Fabian D, Holt S, Beasley R. Global Initiative for Asthma (GINA) Program. The global burden of asthma: executive summary of the GINA Dissemination Committee report. Allergy. 2004; 59:469–478.
Article2. National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): guidelines for the diagnosis and management of asthma-summary report 2007. J Allergy Clin Immunol. 2007; 120:5 Suppl. S94–S138.3. American Thoracic Society. Lung function testing: selection of reference values and interpretative strategies. Am Rev Respir Dis. 1991; 144:1202–1218.4. Kerstjens HA, Brand PL, Quanjer PH, van der Bruggen-Bogaarts BA, Koeter GH, Postma DS. Dutch CNSLD Study Group. Variability of bronchodilator response and effects of inhaled corticosteroid treatment in obstructive airways disease. Thorax. 1993; 48:722–729.
Article5. Crapo RO. Pulmonary-function testing. N Engl J Med. 1994; 331:25–30.
Article6. Strunk RC, Szefler SJ, Phillips BR, Zeiger RS, Chinchilli VM, Larsen G, et al. Relationship of exhaled nitric oxide to clinical and inflammatory markers of persistent asthma in children. J Allergy Clin Immunol. 2003; 112:883–892.
Article7. Covar RA, Szefler SJ, Martin RJ, Sundstrom DA, Silkoff PE, Murphy J, et al. Relations between exhaled nitric oxide and measures of disease activity among children with mild-to-moderate asthma. J Pediatr. 2003; 142:469–475.
Article8. Faul JL, Demers EA, Burke CM, Poulter LW. Alterations in airway inflammation and lung function during corticosteroid therapy for atopic asthma. Chest. 2002; 121:1414–1420.
Article9. Sharma S, Litonjua AA, Tantisira KG, Fuhlbrigge AL, Szefler SJ, Strunk RC, et al. Clinical predictors and outcomes of consistent bronchodilator response in the childhood asthma management program. J Allergy Clin Immunol. 2008; 122:921–928.e4.
Article10. Postma DS, de Vries K, Koeter GH, Sluiter HJ. Independent influence of reversibility of air-flow obstruction and nonspecific hyperreactivity on the long-term course of lung function in chronic air-flow obstruction. Am Rev Respir Dis. 1986; 134:276–280.11. Tantisira KG, Fuhlbrigge AL, Tonascia J, Van Natta M, Zeiger RS, Strunk RC, et al. Bronchodilation and bronchoconstriction: predictors of future lung function in childhood asthma. J Allergy Clin Immunol. 2006; 117:1264–1271.
Article12. Fahy JV, Corry DB, Boushey HA. Airway inflammation and remodeling in asthma. Curr Opin Pulm Med. 2000; 6:15–20.
Article13. Goleva E, Hauk PJ, Boguniewicz J, Martin RJ, Leung DY. Airway remodeling and lack of bronchodilator response in steroid-resistant asthma. J Allergy Clin Immunol. 2007; 120:1065–1072.
Article14. National Heart, Lung and Blood Institute. National asthma education and prevention program. Expert panel report 3: guidelines for the diagnosis and management of asthma. Full report 2007 [Internet]. Bethesda: National Heart, Lung and Blood Institute;cited 2013 Jan 10. Available from: http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.pdf.15. The Global Initiative for Asthma. GINA Report, Global Strategy for Asthma Management and Prevention. Updated Dec. 2011 [Internet]. The Global Initiative For Asthma;2012. cited 2013 Jan 10. Available from: http://www.ginasthma.org/guidelines-gina-report-global-strategy-for-asthma.html.16. Hankinson JL, Odencrantz JR, Fedan KB. Spirometric reference values from a sample of the general U.S. population. Am J Respir Crit Care Med. 1999; 159:179–187.
Article17. Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, Coates A, et al. Standardisation of spirometry. Eur Respir J. 2005; 26:319–338.
Article18. Crapo RO, Casaburi R, Coates AL, Enright PL, Hankinson JL, Irvin CG, et al. Guidelines for methacholine and exercise challenge testing-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000; 161:309–329.19. Recommendations for standardized procedures for the on-line and off-line measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide in adults and children-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 1999; 160:2104–2117.20. Jones SL, Kittelson J, Cowan JO, Flannery EM, Hancox RJ, McLachlan CR, et al. The predictive value of exhaled nitric oxide measurements in assessing changes in asthma control. Am J Respir Crit Care Med. 2001; 164:738–743.
Article21. de Jongste JC, Carraro S, Hop WC, Baraldi E. CHARISM Study Group. Daily telemonitoring of exhaled nitric oxide and symptoms in the treatment of childhood asthma. Am J Respir Crit Care Med. 2009; 179:93–97.
Article22. Stern G, de Jongste J, van der Valk R, Baraldi E, Carraro S, Thamrin C, et al. Fluctuation phenotyping based on daily fraction of exhaled nitric oxide values in asthmatic children. J Allergy Clin Immunol. 2011; 128:293–300.
Article23. Woo SI, Lee JH, Kim H, Kang JW, Sun YH, Hahn YS. Utility of fractional exhaled nitric oxide (F(E)NO) measurements in diagnosing asthma. Respir Med. 2012; 106:1103–1109.
Article24. Kharitonov SA, Yates DH, Barnes PJ. Inhaled glucocorticoids decrease nitric oxide in exhaled air of asthmatic patients. Am J Respir Crit Care Med. 1996; 153:454–457.
Article25. Silkoff PE, McClean P, Spino M, Erlich L, Slutsky AS, Zamel N. Dose-response relationship and reproducibility of the fall in exhaled nitric oxide after inhaled beclomethasone dipropionate therapy in asthma patients. Chest. 2001; 119:1322–1328.
Article26. Suh DI, Lee EJ, Lee JW, Lee JK, Koh YY. Atopy as a risk factor for an elevated bronchodilator response in children with asthma. Pediatr Allergy Respir Dis. 2011; 21:39–46.
Article27. Jackson DJ, Virnig CM, Gangnon RE, Evans MD, Roberg KA, Anderson EL, et al. Fractional exhaled nitric oxide measurements are most closely associated with allergic sensitization in school-age children. J Allergy Clin Immunol. 2009; 124:949–953.
Article28. Spanier AJ, Hornung R, Lierl M, Lanphear BP. Environmental exposures and exhaled nitric oxide in children with asthma. J Pediatr. 2006; 149:220–226.
Article29. Colon-Semidey AJ, Marshik P, Crowley M, Katz R, Kelly HW. Correlation between reversibility of airway obstruction and exhaled nitric oxide levels in children with stable bronchial asthma. Pediatr Pulmonol. 2000; 30:385–392.
Article30. Kitch BT, Paltiel AD, Kuntz KM, Dockery DW, Schouten JP, Weiss ST, et al. A single measure of FEV1 is associated with risk of asthma attacks in long-term follow-up. Chest. 2004; 126:1875–1882.
Article31. Dundas I, Chan EY, Bridge PD, McKenzie SA. Diagnostic accuracy of bronchodilator responsiveness in wheezy children. Thorax. 2005; 60:13–16.
Article32. Galant SP, Morphew T, Newcomb RL, Hioe K, Guijon O, Liao O. The relationship of the bronchodilator response phenotype to poor asthma control in children with normal spirometry. J Pediatr. 2011; 158:953–959.el.
Article33. Bossios A, Gourgiotis D, Skevaki CL, Saxoni-Papageorgiou P, Lotvall J, Psarras S, et al. Rhinovirus infection and house dust mite exposure synergize in inducing bronchial epithelial cell interleukin-8 release. Clin Exp Allergy. 2008; 38:1615–1626.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Utility of Fractional Exhaled Nitric Oxide in the Diagnosis of Asthma and the Assessment of Asthma Control
- Measurements of fractional exhaled nitric oxide in pediatric asthma
- Usefulness of bronchodilator response as an index of asthma control in children
- Relationship between methacholine PC20 level and asthma control status among pediatric patients with atopic asthma
- Relevance of Exhaled Nitric Oxide Levels to Asthma Control Test Scores and Spirometry Values in Children with Atopic Asthma