Allergy Asthma Immunol Res.
2013 Nov;5(6):357-364. 10.4168/aair.2013.5.6.357.
Lost to Follow-Up in Asthmatics Does Not Mean Treatment Failure: Causes and Clinical Outcomes of Non-Adherence To Outpatient Treatment In Adult Asthma
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea. helenmed@snu.ac.kr
- 2Institute of Allergy and Clinical Immunology, Seoul National University Medical Research Center, Seoul, Korea.
- KMID: 2260278
- DOI: http://doi.org/10.4168/aair.2013.5.6.357
Abstract
- PURPOSE
Long-term asthma management is recommended to asthmatics; however, many patients do not adhere to follow-up treatment. It is unclear why many asthmatics do not adhere to follow-up treatment and long-term clinical course after discontinuation of asthma management. This study investigates the factors associated with loss to follow-up and observes the clinical course in asthmatics who discontinued asthma treatment.
METHODS
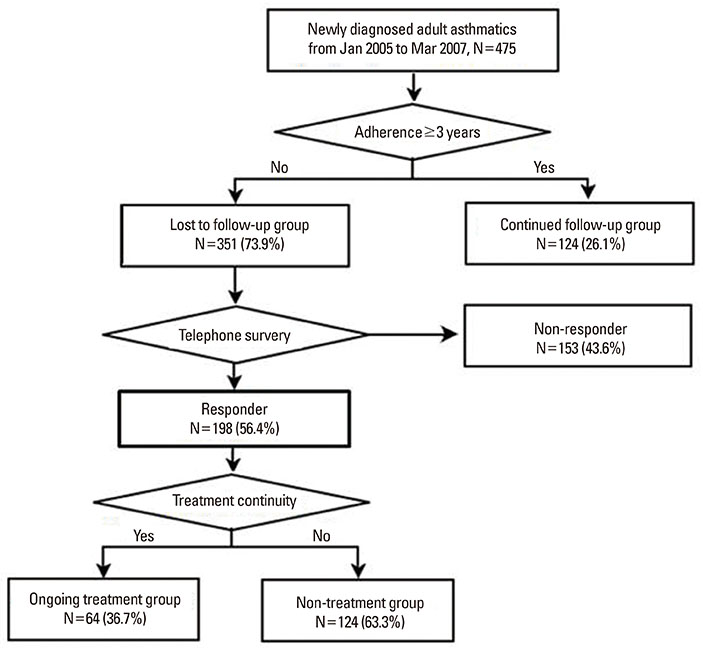
A retrospective investigation was conducted after reviewing medical records of adult patients who were newly diagnosed with asthma at a university hospital in Seoul, South Korea from January 2005 to March 2007. We compared baseline demographics and the clinical and laboratory profiles of patients to see if they successfully adhered to the treatment at an outpatient clinic for at least 3 years. The clinical course and asthma control status were surveyed by telephone for patients who were lost to follow-up within 3 years.
RESULTS
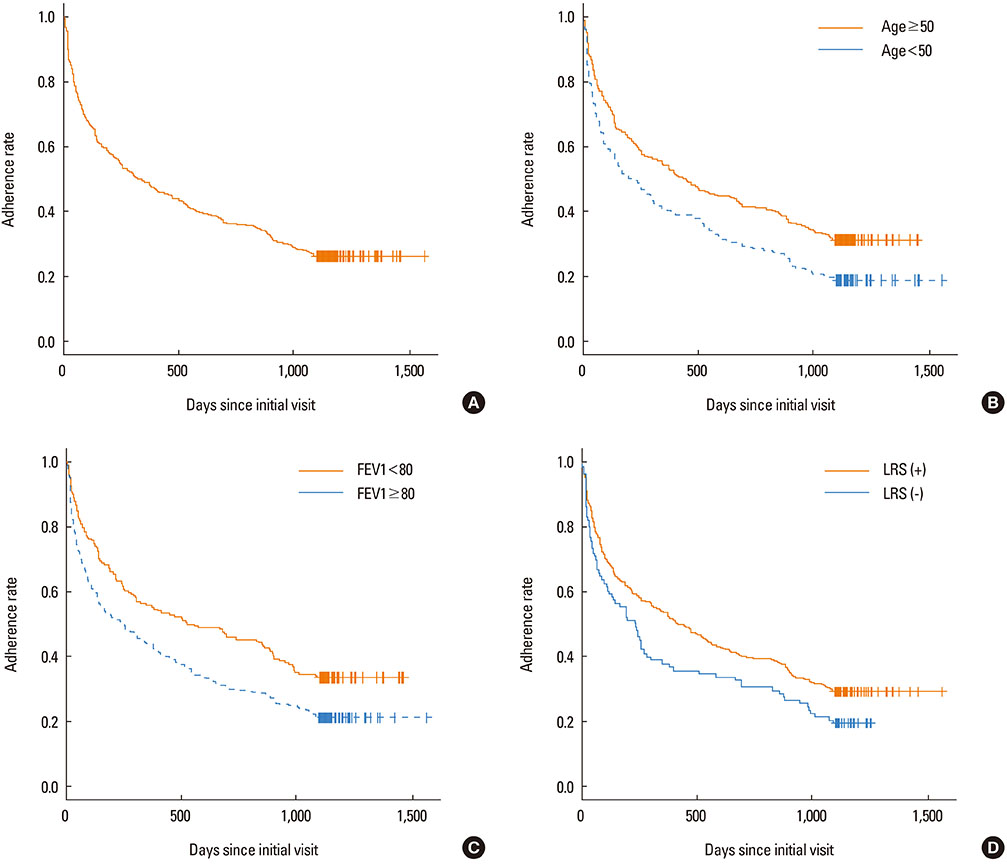
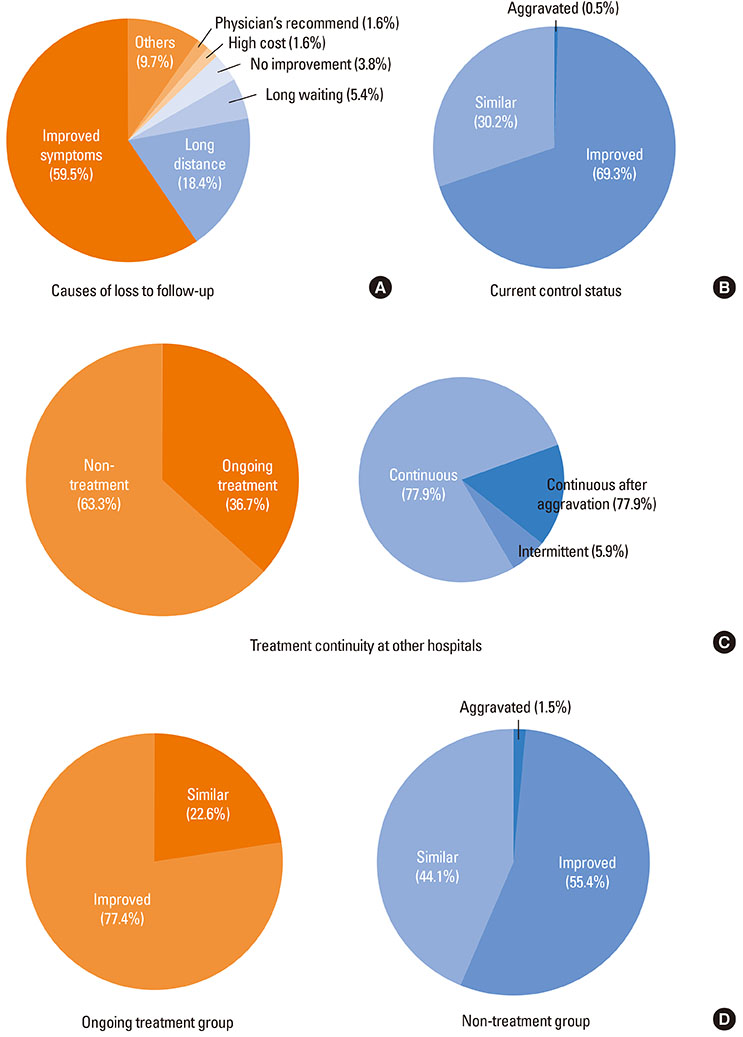
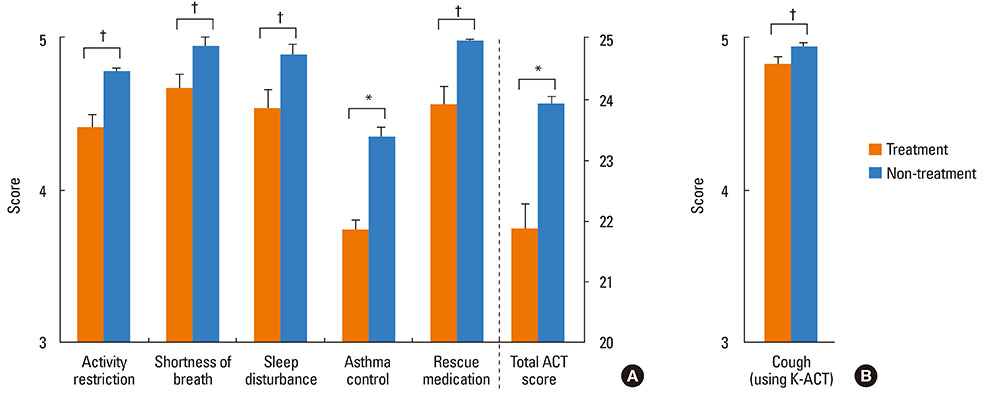
A total of 351 (73.9%) out of 475 patients were lost to follow-up within 3 years of asthma diagnosis. Patients lost to follow-up were younger and had clinical features of less severe asthma at time of diagnosis (higher FEV1 and PC20, and lower grade treatments) compared to patients who adhered to the follow-up for longer than 3 years (all P<0.05). Among the 198 responders to the telephone survey, 124 responders (62.6%) answered that they eventually discontinued asthma medication. A significantly higher proportion of the 124 responders who discontinued asthma treatment maintained symptom improvement compared to the 74 responders who continued asthma medication (77.4% vs. 55.4%, P=0.003).
CONCLUSIONS
Almost three quarters of newly diagnosed asthmatics discontinued asthma medication within 3 years despite a medical recommendation. There are considerable numbers of asthmatics who can maintain long-term asthma control status without medication.
MeSH Terms
Figure
Reference
-
1. Bateman ED, Hurd SS, Barnes PJ, Bousquet J, Drazen JM, FitzGerald M, Gibson P, Ohta K, O'Byrne P, Pedersen SE, Pizzichini E, Sullivan SD, Wenzel SE, Zar HJ. Global strategy for asthma management and prevention: GINA executive summary. Eur Respir J. 2008; 31:143–178.2. Lange P, Scharling H, Ulrik CS, Vestbo J. Inhaled corticosteroids and decline of lung function in community residents with asthma. Thorax. 2006; 61:100–104.3. Beckett PA, Howarth PH. Pharmacotherapy and airway remodelling in asthma? Thorax. 2003; 58:163–174.4. Cochrane MG, Bala MV, Downs KE, Mauskopf J, Ben-Joseph RH. Inhaled corticosteroids for asthma therapy: patient compliance, devices, and inhalation technique. Chest. 2000; 117:542–550.5. Yoo KH, Jeong JW, Yoon HJ, Chang SI, Moon HB, Choi BH, Kim YY, Cho SH. Customized Asthma Control Test with reflection on sociocultural differences. J Korean Med Sci. 2010; 25:1134–1139.6. National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): Guidelines for the diagnosis and management of asthma-summary report 2007. J Allergy Clin Immunol. 2007; 120:S94–S138.7. Barnett SB, Nurmagambetov TA. Costs of asthma in the United States: 2002-2007. J Allergy Clin Immunol. 2011; 127:145–152.8. Kim CY, Park HW, Ko SK, Chang SI, Moon HB, Kim YY, Cho SH. The financial burden of asthma: a nationwide comprehensive survey conducted in the republic of Korea. Allergy Asthma Immunol Res. 2011; 3:34–38.9. Gillisen A. Patient's adherence in asthma. J Physiol Pharmacol. 2007; 58 Suppl 5:205–222.10. Gamble J, Stevenson M, McClean E, Heaney LG. The prevalence of nonadherence in difficult asthma. Am J Respir Crit Care Med. 2009; 180:817–822.11. Coutts JA, Gibson NA, Paton JY. Measuring compliance with inhaled medication in asthma. Arch Dis Child. 1992; 67:332–333.12. Sheffer AL, LaForce C, Chervinsky P, Pearlman D, Schaberg A. Fluticasone Propionate Asthma Study Group. Fluticasone propionate aerosol: efficacy in patients with mild to moderate asthma. J Fam Pract. 1996; 42:369–375.13. Chervinsky P, van As A, Bronsky EA, Dockhorn R, Noonan M, La-Force C, Pleskow W. The Fluticasone Propionate Asthma Study Group. Fluticasone propionate aerosol for the treatment of adults with mild to moderate asthma. J Allergy Clin Immunol. 1994; 94:676–683.14. Boushey HA, Sorkness CA, King TS, Sullivan SD, Fahy JV, Lazarus SC, Chinchilli VM, Craig TJ, Dimango EA, Deykin A, Fagan JK, Fish JE, Ford JG, Kraft M, Lemanske RF Jr, Leone FT, Martin RJ, Mauger EA, Pesola GR, Peters SP, Rollings NJ, Szefler SJ, Wechsler ME, Israel E. National Heart, Lung, and Blood Institute's Asthma Clinical Research Network. Daily versus as-needed corticosteroids for mild persistent asthma. N Engl J Med. 2005; 352:1519–1528.15. Bai TR, Vonk JM, Postma DS, Boezen HM. Severe exacerbations predict excess lung function decline in asthma. Eur Respir J. 2007; 30:452–456.16. Boulet LP, Turcotte H, Laviolette M, Naud F, Bernier MC, Martel S, Chakir J. Airway hyperresponsiveness, inflammation, and subepithelial collagen deposition in recently diagnosed versus long-standing mild asthma. Influence of inhaled corticosteroids. Am J Respir Crit Care Med. 2000; 162:1308–1313.17. Bergeron C, Hauber HP, Gotfried M, Newman K, Dhanda R, Servi RJ, Ludwig MS, Hamid Q. Evidence of remodeling in peripheral airways of patients with mild to moderate asthma: effect of hydrofluoroalkane-flunisolide. J Allergy Clin Immunol. 2005; 116:983–989.18. Papi A, Canonica GW, Maestrelli P, Paggiaro P, Olivieri D, Pozzi E, Crimi N, Vignola AM, Morelli P, Nicolini G, Fabbri LM. BEST Study Group. Rescue use of beclomethasone and albuterol in a single inhaler for mild asthma. N Engl J Med. 2007; 356:2040–2052.19. Price D, Musgrave SD, Shepstone L, Hillyer EV, Sims EJ, Gilbert RF, Juniper EF, Ayres JG, Kemp L, Blyth A, Wilson EC, Wolfe S, Freeman D, Mugford HM, Murdoch J, Harvey I. Leukotriene antagonists as first-line or add-on asthma-controller therapy. N Engl J Med. 2011; 364:1695–1707.20. Szefler SJ, Martin RJ, King TS, Boushey HA, Cherniack RM, Chinchilli VM, Craig TJ, Dolovich M, Drazen JM, Fagan JK, Fahy JV, Fish JE, Ford JG, Israel E, Kiley J, Kraft M, Lazarus SC, Lemanske RF Jr, Mauger E, Peters SP, Sorkness CA. Asthma Clinical Research Network of the National Heart Lung, and Blood Institute. Significant variability in response to inhaled corticosteroids for persistent asthma. J Allergy Clin Immunol. 2002; 109:410–418.21. Cumming RG, Mitchell P, Leeder SR. Use of inhaled corticosteroids and the risk of cataracts. N Engl J Med. 1997; 337:8–14.22. Sharek PJ, Bergman DA. The effect of inhaled steroids on the linear growth of children with asthma: a meta-analysis. Pediatrics. 2000; 106:E8.23. Agertoft L, Pedersen S. Effect of long-term treatment with inhaled budesonide on adult height in children with asthma. N Engl J Med. 2000; 343:1064–1069.24. Deykin A, Lazarus SC, Fahy JV, Wechsler ME, Boushey HA, Chinchilli VM, Craig TJ, Dimango E, Kraft M, Leone F, Lemanske RF, Martin RJ, Pesola GR, Peters SP, Sorkness CA, Szefler SJ, Israel E. Asthma Clinical Research Network, National Heart, Lung, and Blood Institute/NIH. Sputum eosinophil counts predict asthma control after discontinuation of inhaled corticosteroids. J Allergy Clin Immunol. 2005; 115:720–727.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Asthma in the elderly
- Longitudinal Changes in Lung Functions in Adult Asthmatics Treated with Medications
- Comparison of the Patient's Global Self-Assessment Scoring Method with the Quality of Life Questionnaire for Adult Korean Asthmatics
- Pulmonary Function and Clinical Characteristics Influenced by Cigarette Smoking among Adult Asthmatics
- Predictors of Psychiatric Outpatient Adherence after an Emergency Room Visit for a Suicide Attempt