Allergy Asthma Immunol Res.
2014 Nov;6(6):525-534. 10.4168/aair.2014.6.6.525.
The Importance of Allergen Avoidance in High Risk Infants and Sensitized Patients: A Meta-analysis Study
- Affiliations
-
- 1Guangdong Provincial Key Laboratory of Allergy & Clinical Immunology, the State Key Laboratory of Respiratory Disease, the Second Affiliated Hospital of Guangzhou Medical University, Guangdong 510260, China. taoailin@gzhmu.edu.cn
- 2Respiratory Department, Southern Building, General Hospital of Chinese People's Liberation Army, Beijing 100853, China. 13801151778@163.com
- KMID: 2260175
- DOI: http://doi.org/10.4168/aair.2014.6.6.525
Abstract
- PURPOSE
At this time, there is uncertainty regarding whether allergen avoidance is the most appropriate strategy for managing or preventing allergies. The purpose of this study was to evaluate the effectiveness of allergen avoidance in the prevention of allergic symptoms in previously sensitized patients and newborns that have the potential to develop allergies.
METHODS
We performed online searches of articles published from January 1980 to December 2012 in PubMed and The Cochrane Central Register of Controlled Trials, and selected articles involving randomized controlled trials (RCTs) and allergen avoidance. The parameters used to determine allergenic potential in newborns included the risk ratio (RR) of eczema, asthma, rhinitis, wheeze, and cough. The methods employed to evaluate previously sensitized patients were the standardized mean difference (SMD) of forced expiratory volume in 1 second (FEV1) and peak expiratory flow rate (PEFR). Data quality was assessed using the Jadad scale.
RESULTS
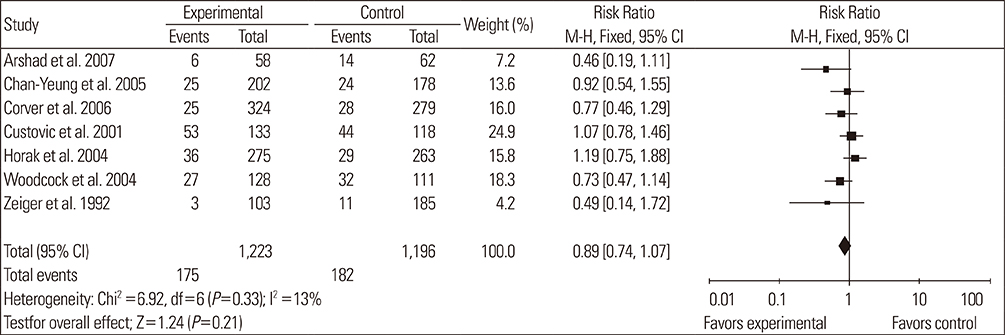
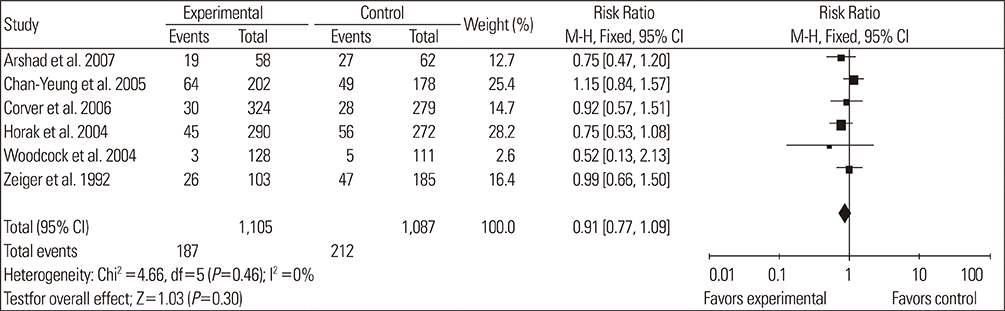
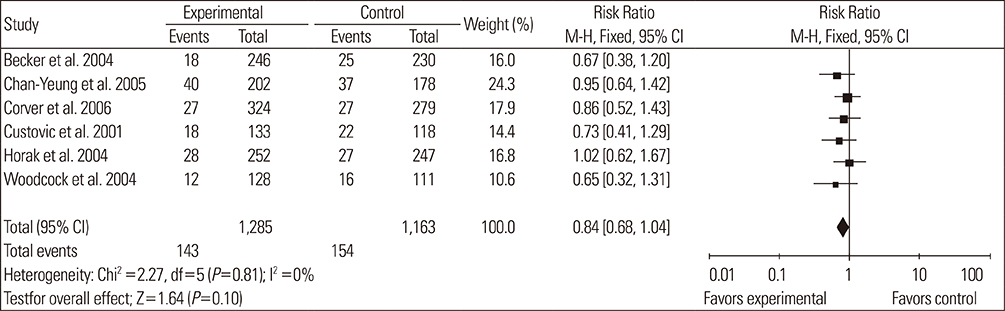
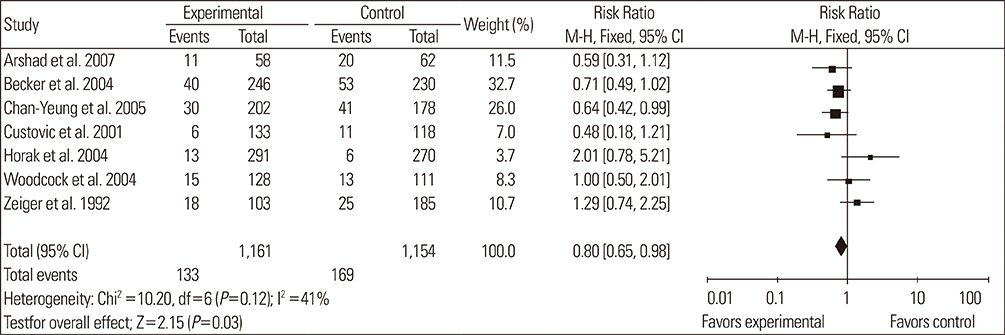
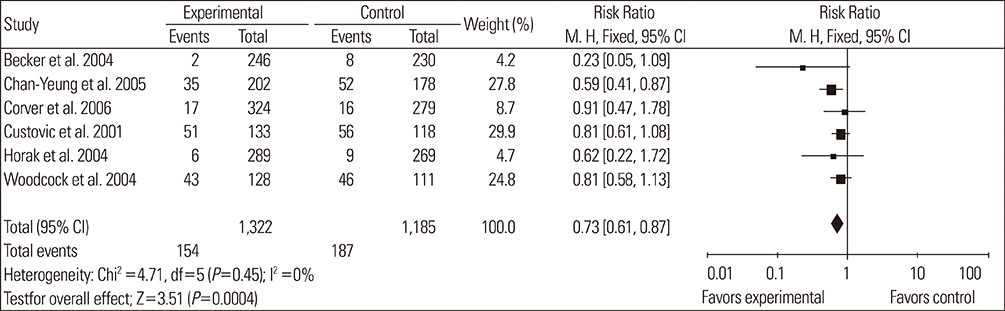
A total of 14 RCTs were identified. Meta-analysis demonstrated that allergen avoidance for newborns did not reduce the subsequent incidence of allergic diseases (eczema, P=0.21; rhinitis, P=0.3; cough, P=0.1) but significantly reduced the incidence of asthma and wheezing in high-risk infants (asthma, P=0.03; wheeze, P=0.0004). However, previously sensitized patients who reduced their exposure to known allergens did not show improvement in their lung functions (FEV1, P=0.3; PEFR morning, P=0.53; PEFR evening, P=0.2; PEFR, P=0.29).
CONCLUSIONS
Allergen avoidance may not always be successful in preventing allergic symptoms. However, rigorous methodological studies are required to confirm this hypothesis.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Gene-Environment Interactions in Asthma: Genetic and Epigenetic Effects
Jong-Uk Lee, Jeong Dong Kim, Choon-Sik Park
Yonsei Med J. 2015;56(4):877-886. doi: 10.3349/ymj.2015.56.4.877.Clinical Implications of Single Nucleotide Polymorphisms in Diagnosis of Asthma and its Subtypes
Jong-Sook Park, Ji-Hye Son, Choon-Sik Park, Hun Soo Chang
Yonsei Med J. 2019;60(1):1-9. doi: 10.3349/ymj.2019.60.1.1.
Reference
-
1. Ring J, Akdis C, Behrendt H, Lauener RP, Schäppi G, Akdis M, Ammann W, de Beaumont O, Bieber T, Bienenstock J, Blaser K, Bochner B, Bousquet J, Crameri R, Custovic A, Czerkinsky C, Darsow U, Denburg J, Drazen J, de Villiers EM, Fire A, Galli S, Haahtela T, zur Hausen H, Hildemann S, Holgate S, Holt P, Jakob T, Jung A, Kemeny M, Koren H, Leung D, Lockey R, Marone G, Mempel M, Menné B, Menz G, Mueller U, von Mutius E, Ollert M, O'Mahony L, Pawankar R, Renz H, Platts-Mills T, Roduit C, Schmidt-Weber C, Traidl-Hoffmann C, Wahn U, Rietschel E. Davos declaration: allergy as a global problem. Allergy. 2012; 67:141–143.2. Prescott SL, Bouygue GR, Videky D, Fiocchi A. Avoidance or exposure to foods in prevention and treatment of food allergy? Curr Opin Allergy Clin Immunol. 2010; 10:258–266.3. Eder W, Ege MJ, von Mutius E. The asthma epidemic. N Engl J Med. 2006; 355:2226–2235.4. Bieber T. Atopic dermatitis. N Engl J Med. 2008; 358:1483–1494.5. de Vries MP, van den Bemt L, Aretz K, Thoonen BP, Muris JW, Kester AD, Cloosterman S, van Schayck CP. House dust mite allergen avoidance and self-management in allergic patients with asthma: randomised controlled trial. Br J Gen Pract. 2007; 57:184–190.6. Flinterman AE, Knulst AC, Meijer Y, Bruijnzeel-Koomen CA, Pasmans SG. Acute allergic reactions in children with AEDS after prolonged cow's milk elimination diets. Allergy. 2006; 61:370–374.7. Du Toit G, Katz Y, Sasieni P, Mesher D, Maleki SJ, Fisher HR, Fox AT, Turcanu V, Amir T, Zadik-Mnuhin G, Cohen A, Livne I, Lack G. Early consumption of peanuts in infancy is associated with a low prevalence of peanut allergy. J Allergy Clin Immunol. 2008; 122:984–991.8. Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996; 17:1–12.9. Schachter HM, Pham B, King J, Langford S, Moher D. How efficacious and safe is short-acting methylphenidate for the treatment of attention-deficit disorder in children and adolescents? A meta-analysis. CMAJ. 2001; 165:1475–1488.10. Schulz KF, Chalmers I, Hayes RJ, Altman DG. Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials. JAMA. 1995; 273:408–412.11. Arshad SH, Bateman B, Sadeghnejad A, Gant C, Matthews SM. Prevention of allergic disease during childhood by allergen avoidance: the Isle of Wight prevention study. J Allergy Clin Immunol. 2007; 119:307–313.12. Becker A, Watson W, Ferguson A, Dimich-Ward H, Chan-Yeung M. The Canadian asthma primary prevention study: outcomes at 2 years of age. J Allergy Clin Immunol. 2004; 113:650–656.13. Chan-Yeung M, Ferguson A, Watson W, Dimich-Ward H, Rousseau R, Lilley M, Dybuncio A, Becker A. The Canadian Childhood Asthma Primary Prevention Study: outcomes at 7 years of age. J Allergy Clin Immunol. 2005; 116:49–55.14. Corver K, Kerkhof M, Brussee JE, Brunekreef B, van Strien RT, Vos AP, Smit HA, Gerritsen J, Neijens HJ, de Jongste JC. House dust mite allergen reduction and allergy at 4 yr: follow up of the PIAMA-study. Pediatr Allergy Immunol. 2006; 17:329–336.15. Custovic A, Simpson BM, Simpson A, Kissen P, Woodcock A. NAC Manchester Asthma and Allergy Study Group. Effect of environmental manipulation in pregnancy and early life on respiratory symptoms and atopy during first year of life: a randomised trial. Lancet. 2001; 358:188–193.16. Eggleston PA, Butz A, Rand C, Curtin-Brosnan J, Kanchanaraksa S, Swartz L, Breysse P, Buckley T, Diette G, Merriman B, Krishnan JA. Home environmental intervention in inner-city asthma: a randomized controlled clinical trial. Ann Allergy Asthma Immunol. 2005; 95:518–524.17. Hayden ML, Perzanowski M, Matheson L, Scott P, Call RS, Platts-Mills TA. Dust mite allergen avoidance in the treatment of hospitalized children with asthma. Ann Allergy Asthma Immunol. 1997; 79:437–442.18. Horak F Jr, Matthews S, Ihorst G, Arshad SH, Frischer T, Kuehr J, Schwieger A, Forster J. SPACE study group. Effect of mite-impermeable mattress encasings and an educational package on the development of allergies in a multinational randomized, controlled birth-cohort study -- 24 months results of the Study of Prevention of Allergy in Children in Europe. Clin Exp Allergy. 2004; 34:1220–1225.19. Morgan WJ, Crain EF, Gruchalla RS, O'Connor GT, Kattan M, Evans R 3rd, Stout J, Malindzak G, Smartt E, Plaut M, Walter M, Vaughn B, Mitchell H. Inner-City Asthma Study Group. Results of a home-based environmental intervention among urban children with asthma. N Engl J Med. 2004; 351:1068–1080.20. Rijssenbeek-Nouwens LH, Oosting AJ, de Bruin-Weller MS, Bregman I, de Monchy JG, Postma DS. Clinical evaluation of the effect of anti-allergic mattress covers in patients with moderate to severe asthma and house dust mite allergy: a randomised double blind placebo controlled study. Thorax. 2002; 57:784–790.21. Sheikh A, Hurwitz B, Sibbald B, Barnes G, Howe M, Durham S. House dust mite barrier bedding for childhood asthma: randomised placebo controlled trial in primary care [ISRCTN 63308372]. BMC Fam Pract. 2002; 3:12.22. Woodcock A, Lowe LA, Murray CS, Simpson BM, Pipis SD, Kissen P, Simpson A, Custovic A. NAC Manchester Asthma and Allergy Study Group. Early life environmental control: effect on symptoms, sensitization, and lung function at age 3 years. Am J Respir Crit Care Med. 2004; 170:433–439.23. Wright GR, Howieson S, McSharry C, McMahon AD, Chaudhuri R, Thompson J, Donnelly I, Brooks RG, Lawson A, Jolly L, McAlpine L, King EM, Chapman MD, Wood S, Thomson NC. Effect of improved home ventilation on asthma control and house dust mite allergen levels. Allergy. 2009; 64:1671–1680.24. Zeiger RS, Heller S, Mellon MH, Halsey JF, Hamburger RN, Sampson HA. Genetic and environmental factors affecting the development of atopy through age 4 in children of atopic parents: a prospective randomized study of food allergen avoidance. Pediatr Allergy Immunol. 2007; 3:110–127.25. Ihre E, Zetterström O. Increase in non-specific bronchial responsiveness after repeated inhalation of low doses of allergen. Clin Exp Allergy. 1993; 23:298–305.26. Rijssenbeek-Nouwens LH, Oosting AJ, De Monchy JG, Bregman I, Postma DS, De Bruin-Weller MS. The effect of anti-allergic mattress encasings on house dust mite-induced early- and late-airway reactions in asthmatic patients. A double-blind, placebo-controlled study. Clin Exp Allergy. 2002; 32:117–125.27. Luczynska C, Tredwell E, Smeeton N, Burney P. A randomized controlled trial of mite allergen-impermeable bed covers in adult mite-sensitized asthmatics. Clin Exp Allergy. 2003; 33:1648–1653.28. Marks GB, Tovey ER, Green W, Shearer M, Salome CM, Woolcock AJ. House dust mite allergen avoidance: a randomized controlled trial of surface chemical treatment and encasement of bedding. Clin Exp Allergy. 1994; 24:1078–1083.29. Woodcock A, Forster L, Matthews E, Martin J, Letley L, Vickers M, Britton J, Strachan D, Howarth P, Altmann D, Frost C, Custovic A. Medical Research Council General Practice Research Framework. Control of exposure to mite allergen and allergen-impermeable bed covers for adults with asthma. N Engl J Med. 2003; 349:225–236.30. Jennings S, Prescott SL. Early dietary exposures and feeding practices: role in pathogenesis and prevention of allergic disease? Postgrad Med J. 2010; 86:94–99.31. Gøtzsche PC, Johansen HK, Schmidt LM, Burr ML. House dust mite control measures for asthma. Cochrane Database Syst Rev. 2004; CD001187.32. Liao EC, Lee MF, Tsaii JJ. The tropmyosin specific IgE and its roles of cross-reactivity between shrimp and dust mites. Clin Transl Allergy. 2011; 1:P70.33. Wang J, Calatroni A, Visness CM, Sampson HA. Correlation of specific IgE to shrimp with cockroach and dust mite exposure and sensitization in an inner-city population. J Allergy Clin Immunol. 2011; 128:834–837.34. Fernández-Rivas M, Bolhaar S, González-Mancebo E, Asero R, van Leeuwen A, Bohle B, Ma Y, Ebner C, Rigby N, Sancho AI, Miles S, Zuidmeer L, Knulst A, Breiteneder H, Mills C, Hoffmann-Sommergruber K, van Ree R. Apple allergy across Europe: how allergen sensitization profiles determine the clinical expression of allergies to plant foods. J Allergy Clin Immunol. 2006; 118:481–488.35. Vieths S, Scheurer S, Ballmer-Weber B. Current understanding of cross-reactivity of food allergens and pollen. Ann N Y Acad Sci. 2002; 964:47–68.36. Scheurer S, Wangorsch A, Haustein D, Vieths S. Cloning of the minor allergen Api g 4 profilin from celery (Apium graveolens) and its cross-reactivity with birch pollen profilin Bet v 2. Clin Exp Allergy. 2000; 30:962–971.37. Davies JM, Dang TD, Voskamp A, Drew AC, Biondo M, Phung M, Upham JW, Rolland JM, O'Hehir RE. Functional immunoglobulin E cross-reactivity between Pas n 1 of Bahia grass pollen and other group 1 grass pollen allergens. Clin Exp Allergy. 2011; 41:281–291.38. Kim JS, Sicherer SH. Living with food allergy: allergen avoidance. Pediatr Clin North Am. 2011; 58:459–470. xi39. Ling M, Long AA. Pet dander and difficult-to-control asthma: therapeutic options. Allergy Asthma Proc. 2010; 31:385–391.40. Braun-Fahrländer C, Riedler J, Herz U, Eder W, Waser M, Grize L, Maisch S, Carr D, Gerlach F, Bufe A, Lauener RP, Schierl R, Renz H, Nowak D, von Mutius E. Allergy and Endotoxin Study Team. Environmental exposure to endotoxin and its relation to asthma in school-age children. N Engl J Med. 2002; 347:869–877.41. Tomicić S, Fälth-Magnusson K, Böttcher MF. Dysregulated Th1 and Th2 responses in food-allergic children--does elimination diet contribute to the dysregulation? Pediatr Allergy Immunol. 2010; 21:649–655.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Indoor Allergen Avoidance and Asthma
- Family-Centered Care for High-Risk Infants and the Roles of Healthcare Professionals
- Blood eosinophil counts as a biomarker for allergen sensitization in childhood allergic diseases in comparison with total IgE
- The Investigation of Hypoproteinemia in Severe Infantile Atopic Dermatitis
- Current trends in treatment of allergic rhinitis