J Korean Ophthalmol Soc.
2009 Apr;50(4):594-602. 10.3341/jkos.2009.50.4.594.
Effect of Triamcinolone on Angiogenesis-related Factors of Cultured Retinal Pigment Epithelial Cells
- Affiliations
-
- 1Department of Ophthalmology, College of Medicine, Chosun University, Gwangju, Korea. eyelovehyun@hanmail.net
- 2I Clinic, Gwangju, Korea.
- KMID: 2212262
- DOI: http://doi.org/10.3341/jkos.2009.50.4.594
Abstract
-
PURPOSE: To examine the effects of triamcinolone on angiogenesis related factors in cultured human retinal pigment epithelial cells.
METHODS
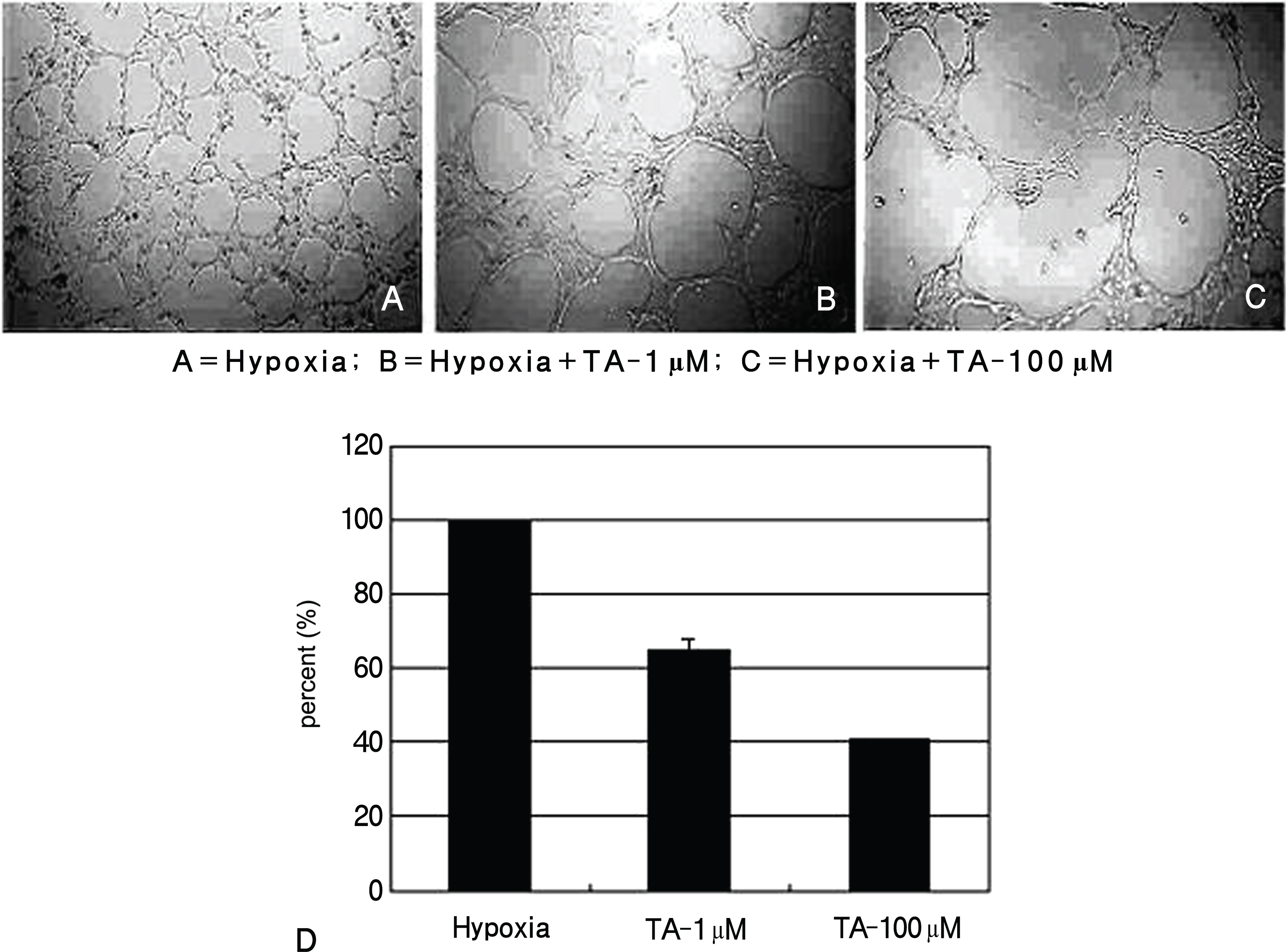
Human retinal pigment epithelial cells were exposed to triamcinolone, cultured in a hypoxic environment, and expression and production of VEGF and PEDF were subsequently tested by RT-PCR and Western blot. Angiogenesis was measured via a tube formation assay using ECV 304 cells and with a migration assay using human dermal microvascular endothelial cells.
RESULTS
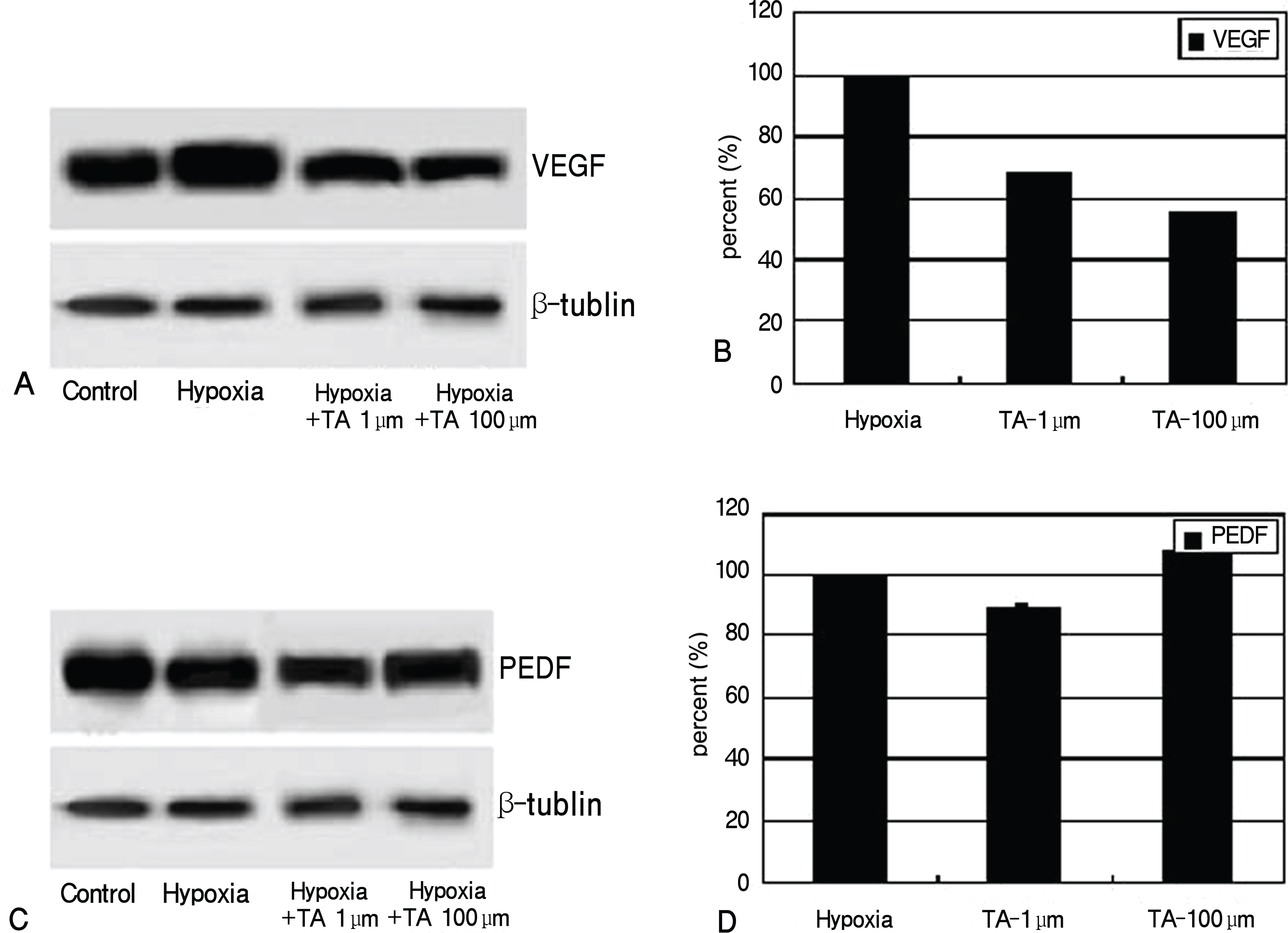
Expression and production of VEGF and PEDF were tested by RT-PCR and Western blot, respectively. VEGF abundance was reduced while that of PEDF was unchanged in triamcinolone exposed retinal pigment epithelial cells cultured in hypoxic environment compared with cells with no treatment in hypoxic environment (p<0.05). Tube formation and cell migration were reduced by triamcinolone (p<0.05).
CONCLUSIONS
These results suggest that triamcinolone affects the secretion of angiogenesis-related factors and suppresses neovascularization.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Kim JW, Kim HK, Kim HC. Photodynamic Therapy for Choroidal Neovascularization caused by age-related macular degeneration. J Korean Ophthalmol Soc. 2002; 43:1435–43.2. Kosano H, Okano T, Katsura Y, et al. ProMMP-9 (92 kDa gelatinase) in vitreous fluid of patients with proliferative diabetic retinopathy. Life Sci. 1999; 64:2307–15.
Article3. Vingerling JR, Dielemans I, Hofman A, et al. The prevalence of age-related maculopathy in Rotterdam Study. Ophthalmology. 1995; 102:205–10.4. Jin M, Kashiwangi K, Iizuka Y, et al. Matrix metalloproteinases in human diabetic and nondiabetic vitreous. Retina. 2001; 21:28–33.
Article5. Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996; 86:353–64.
Article6. Camochiaro PA. Retinal and choroidal neovascularization. J Cell Physiol. 2000; 184:301–10.7. Kim YD, Park YC, Choi GJ. Expression of angiogenesis-related factors in retinal pigment epithelial cells under hypoxia. J Korean Ophthalmol Soc. 2006; 47:629–36.8. Kim JM, Kim JY, Lee YH, Choi GJ. Angiogenesis according to expressive change of angiogenic related factor in human RPE under oxidative stress. J Korean Ophthalmol Soc. 2005; 46:366–76.9. Plate KH, Breier G, Welch HA, et al. Vascular endothelial growth factor is a potential tumor angiogenesis factor in human gliomas in vivo. Nature. 1992; 359:845–8.10. Behzadian MA, Wang XL, AL-Shabrawey M, Caldwell RB. Effects of hypoxia on glial cell expression of angiogenesis-regulating factors VEGF and TGF-beta. Glia. 1998; 24:216–25.11. Maione TE, Gray GS, Petro J, et al. Inhibition of angiogenesis by recombinant human platelet factor-4 and related peptides. Science. 1990; 247:77–9.
Article12. O'Reilly MS, Holmgren L, Shing Y, et al. Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis lung carcinoma. Cell. 1994; 79:315–28.13. O'Reilly MS, Boehm T, Shing Y, et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell. 1997; 88:277–85.14. Yao Y, Guan M, Zhao XQ, Huang YF. Downregulation of the pigment epithelium derived factor by hypoxia and elevated glucose concentration in cultured human retinal pigment epithelial cells. Zhonghua Yi Xue Za Zhi. 2003; 25:1989–92.15. Boyd SR, Zachary I, Chajravarthy U, et al. Correlation of increased vascular growth factor with neovascularization and permeability in ischemic central vein occlusion. Arch Ophthalmol. 2002; 120:1644–50.16. Ogata N, Nishikawa M, Nishimura T, et al. Unbalanced vitreous level of pigment epithelium-derived factor and vascular endothelial growth factor in diabetic retinopathy. Am J Ophthalmol. 2002; 134:348–53.17. Panfold PL, Gyory JF, Hunyor AB, Billson FA. Exudative macular degeneration and intravitreal triamcinolone: a pilot study. Aust N Z J Ophthalmol. 1995; 23:293–8.18. Jonas JB, Akkoyun I, Budde WM, et al. Intravitreal reinjection of triamcinolone for exudative age-related macular degeneration. Arch Ophthalmol. 2004; 122:218–22.19. Jonas JB, Kressig I, Hugger P, et al. Intravitreal triamcinolone acetonide for exudative age-related macular degeneration. Br J Ophthalmol. 2003; 87:462–8.20. Ciulla TA, Criwell MH, Danis RP, et al. Choroidal neovascular membrane inhibition in a laser treated rat model with intraocular sustained release triamcinolone acetonide microimplants. Br J Ophthalmol. 2003; 87:1032–7.
Article21. Pieters R., Huismans DR, Leyva A, Veerman AJ. Adaptation of the rapid automated tetrazolium dye based (MTT) assay for chemo-sensitivity testing in childhood leukemia. Cancer Lett. 1988; 41:323–32.
Article22. O'Connor KA, Hansen MK, Rachal Pugh C, et al. Further characterization of high mobility group box 1 (HMGB1) as a proinflammatory cytokine: central nervous system effects. Cytokine. 2003; 24:254–65.23. Ohno-Matsui K, Morita I, Tombran-Tink J, et al. Novel mechanism for age-related macular degeneration: an equilibrium shift between the angiogenesis factors VEGF and PEDF. J Cell Physiol. 2001; 189:323–33.
Article24. Nakajima-Iijima S, Hamada H, Reddy P, Kakunaga T. Molecular structure of the human cytoplasmic beta-actin gene: interspecies homology of sequences in the introns. Proc Natl Acad Sci U S A. 1985; 82:6133–37.
Article25. Laemmli UK, Beguin F, Gujer-Kellenberger G. A factor preventing the major head protein of bacteriophage T4 from random aggregation. J Mol Biol. 1970; 47:69–85.
Article26. Matsuda S, Gomi F, Oshima Y, et al. Vascular Endothelial Growth Factor Reduced and Connective Tissue Growth Factor Induced by Triamcinolone in ARPE19 Cells under Oxidative Stress. Invest Ophthalmol Vis Sci. 2005; 46:1062–8.
Article27. Song HS, Son MJ, Lee YM, et al. Oxygen tension regulates the maturation of the blood-brain barrier. Biochem Biophys Res Commun. 2002; 290:325–31.
Article28. Heidemann J, Ogawa H, Dwinell MB, et al. Angiogenic effects of interleukin-8 (CXCL8) in human intestinal microvascular endothelial cells are mediated by CXCR2. J Biol Chem. 2003; 278:8508–15.29. Smith CL, Birdsey GM, Anthony S, et al. Dimethylarginine dimethylaminohydrolase activity modulates ADMA levels, VEGF expression, and cell phenotype. Biochem Biophys Res Commun. 2003; 308:984–9.
Article30. Tombran-Tink J, Chader GG, Johnson LV. PEDF: a pigment epithelium-derived factor with potent neuronal differentiative activity. Exp Eye Res. 1991; 53:411–4.
Article31. Jung S, Kim HW, Lee JH, et al. Brain tumor invasion model system using organotypic brain-slice culture as an alternative to in vivo model. J Cancer Res Clin Oncol. 2002; 128:469–76.
Article32. Yancopoulos G, Klagsbrun M, Folkman J. Vasculogenesis, angiogenesis, and growth factor: ephrins enter the fray at the border. Cell. 1998; 93:661–4.33. Risau W. Mechanisms of angiogenesis. Nature. 1997; 386:671–4.
Article34. Dawson DW, Volpert OV, Gillis P, et al. Pigment epithelium-derived factor: A potent inhibitor of angiogenesis. Science. 1999; 285:245–48.
Article35. Bussolio F, Mantovani A, Persico G. Molecular mechanisms of blood vessel formation. Trends Biochem Sci. 1997; 22:251–6.36. Raymond L, Jacobson B. Isolation and identification of stimu-latory and inhibitory cell growth factors in bovine vitreous. Exp Eye Res. 1982; 34:267–86.
Article37. Schlingemann RO. Role of growth factor and the wound healing response in age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2004; 242:91–101.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of 5-Fluorouraci1 on the Activity of the Retinal Pigment Epithelium in Vitro
- Culture of Retinal Pigment Epithelial Cells on Collagen Membrane
- Growth Patterns of Human Retinal Pigment Epithelium in Vitro
- Histologic Study in Transplantation of Cultured Rabbit Retinal Pigment Epithelium
- Comparison of Inhibitory Effects of 5-Fluorouracil and Mitomycin C on Proliferation of Fibroblagts and Retinal Pigment Epithelial Cells