J Korean Neurosurg Soc.
2014 Jan;55(1):5-11. 10.3340/jkns.2014.55.1.5.
Pseudoprogression and Pseudoresponse in the Management of High-Grade Glioma : Optimal Decision Timing According to the Response Assessment of the Neuro-Oncology Working Group
- Affiliations
-
- 1Department of Radiation Oncology, Seoul National University Bundang Hospital, Seongnam, Korea. inah228@snu.ac.kr
- 2Department of Neurosurgery, Seoul National University Bundang Hospital, Seongnam, Korea.
- 3Department of Radiology, Seoul National University Bundang Hospital, Seongnam, Korea.
- 4Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- KMID: 2191033
- DOI: http://doi.org/10.3340/jkns.2014.55.1.5
Abstract
OBJECTIVE
We evaluated pseudoprogression (PsPD) following radiation therapy combined with concurrent temozolomide (TMZ), and we assessed pseudoresponse following anti-angiogenic therapy for patients with recurrent disease using the Response Assessment of the Neuro-Oncology Working Group.
METHODS
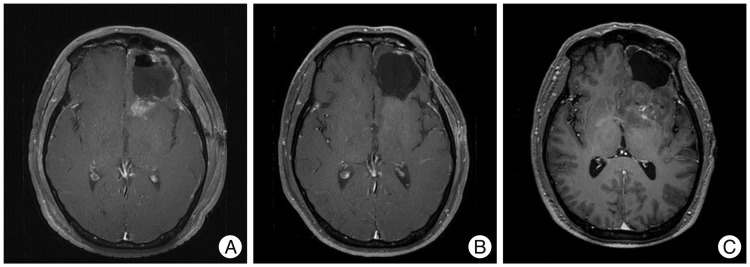
Patients who were pathologically confirmed as having high-grade glioma received radiotherapy with concurrent TMZ followed by adjuvant TMZ. Bevacizumab (Avastin) with CPT-11 were used as a salvage option for cases of radiologic progression. Magnetic resonance imaging (MRI) was routinely performed 1 month after concurrent radiochemotherapy (CRT) and every 3 months thereafter. For cases treated with the bevacizumab-containing regimen for progressive disease, MRI was performed every 2 months.
RESULTS
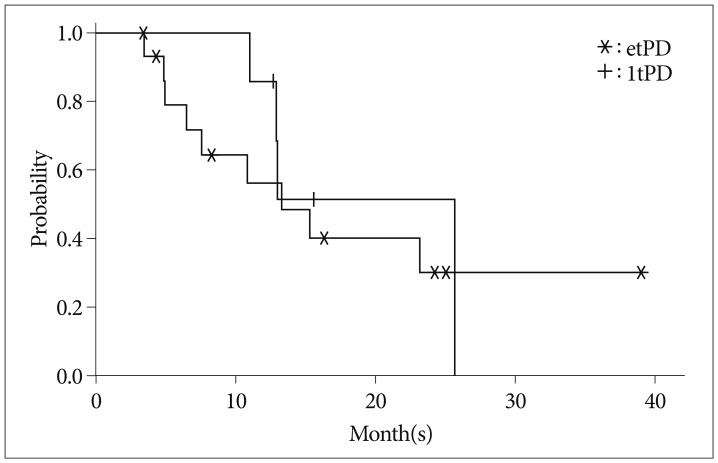
Of 55 patients, 21 (38%) showed radiologic progression within 4 weeks after CRT. Of these patients, 16 (29%) showed progression at second post-CRT MRI (etPD) and five (9%) showed improvement (PsPD). Seven of thirty-four initially non-progressed patients showed progression at the second post-CRT MRI (ltPD). No difference in survival was observed between the etPD and ltPD groups (p=0.595). Five (50%) of ten patients showed a radiological response after salvage bevacizumab therapy. Four of those patients exhibited rapid progression immediately after discontinuation of the drug (drug holiday).
CONCLUSION
Twelve weeks following treatment could be the optimal timing to determine PsPD or true progression. MRI with gadolinium enhancement alone is not sufficient to characterize tumor response or growth. Clinical correlation with adequate follow-up duration and histopathologic validation may be helpful in discriminating PsPD from true progression.
MeSH Terms
Figure
Reference
-
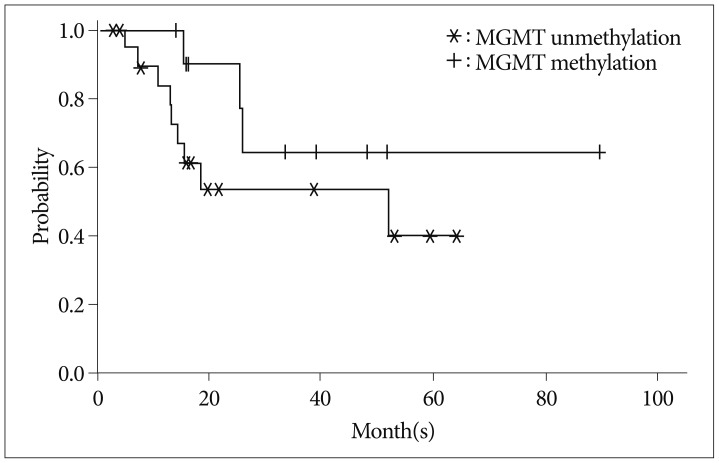
1. Brandes AA, Franceschi E, Tosoni A, Blatt V, Pession A, Tallini G. MGMT promoter methylation status can predict the incidence and outcome of pseudoprogression after concomitant radiochemotherapy in newly diagnosed glioblastoma patients. J Clin Oncol. 2008; 26:2192–2197. PMID: 18445844.
Article2. Caroline I, Rosenthal MA. Imaging modalities in high-grade gliomas : pseudoprogression, recurrence, or necrosis? J Clin Neurosci. 2012; 19:633–637. PMID: 22321359.
Article3. Chamberlain MC. Bevacizumab for the treatment of recurrent glioblastoma. Clin Med Insights Oncol. 2011; 5:117–129. PMID: 21603247.
Article4. Chaskis C, Neyns B, Michotte A, De Ridder M, Everaert H. Pseudoprogression after radiotherapy with concurrent temozolomide for high-grade glioma : clinical observations and working recommendations. Surg Neurol. 2009; 72:423–428. PMID: 19150114.
Article5. Easaw JC, Mason WP, Perry J, Laperrière N, Eisenstat DD, Del Maestro R, et al. Canadian recommendations for the treatment of recurrent or progressive glioblastoma multiforme. Curr Oncol. 2011; 18:e126–e136. PMID: 21655151.
Article6. Fauquette W, Amourette C, Dehouck MP, Diserbo M. Radiation-induced blood-brain barrier damages : an in vitro study. Brain Res. 2012; 1433:114–126. PMID: 22153623.7. Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE, et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009; 27:4733–4740. PMID: 19720927.
Article8. Gállego Pérez-Larraya J, Lahutte M, Petrirena G, Reyes-Botero G, González-Aguilar A, Houillier C, et al. Response assessment in recurrent glioblastoma treated with irinotecan-bevacizumab : comparative analysis of the Macdonald, RECIST, RANO, and RECIST + F criteria. Neuro Oncol. 2012; 14:667–673. PMID: 22492961.9. Joo JD, Chang JH, Kim JH, Hong YK, Kim YH, Kim CY. Temozolomide during and after radiotherapy for newly diagnosed glioblastomas : a prospective multicenter study of Korean patients. J Korean Neurosurg Soc. 2012; 52:92–97. PMID: 23091665.
Article10. Kang HC, Kim CY, Han JH, Choe GY, Kim JH, Kim JH, et al. Pseudoprogression in patients with malignant gliomas treated with concurrent temozolomide and radiotherapy : potential role of p53. J Neurooncol. 2011; 102:157–162. PMID: 20632071.
Article11. Kreisl TN, Kim L, Moore K, Duic P, Royce C, Stroud I, et al. Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol. 2009; 27:740–745. PMID: 19114704.
Article12. Lamborn KR, Yung WK, Chang SM, Wen PY, Cloughesy TF, DeAngelis LM, et al. Progression-free survival : an important end point in evaluating therapy for recurrent high-grade gliomas. Neuro Oncol. 2008; 10:162–170. PMID: 18356283.
Article13. Laymon CM, Oborski MJ, Lee VK, Davis DK, Wiener EC, Lieberman FS, et al. Combined imaging biomarkers for therapy evaluation in glioblastoma multiforme : correlating sodium MRI and F-18 FLT PET on a voxel-wise basis. Magn Reson Imaging. 2012; 30:1268–1278. PMID: 22819581.
Article14. Lutz K, Radbruch A, Wiestler B, Bäumer P, Wick W, Bendszus M. Neuroradiological response criteria for high-grade gliomas. Clin Neuroradiol. 2011; 21:199–205. PMID: 21681688.
Article15. Macdonald DR, Cascino TL, Schold SC Jr, Cairncross JG. Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol. 1990; 8:1277–1280. PMID: 2358840.
Article16. Mangla R, Singh G, Ziegelitz D, Milano MT, Korones DN, Zhong J, et al. Changes in relative cerebral blood volume 1 month after radiation-temozolomide therapy can help predict overall survival in patients with glioblastoma. Radiology. 2010; 256:575–584. PMID: 20529987.
Article17. Miyatake S, Furuse M, Kawabata S, Maruyama T, Kumabe T, Kuroiwa T, et al. Bevacizumab treatment of symptomatic pseudoprogression after boron neutron capture therapy for recurrent malignant gliomas. Report of 2 cases. Neuro Oncol. 2013; 15:650–655. PMID: 23460324.
Article18. Norden AD, Drappatz J, Muzikansky A, David K, Gerard M, McNamara MB, et al. An exploratory survival analysis of anti-angiogenic therapy for recurrent malignant glioma. J Neurooncol. 2009; 92:149–155. PMID: 19043778.
Article19. Pan E, Tsai JS, Mitchell SB. Retrospective study of venous thromboembolic and intracerebral hemorrhagic events in glioblastoma patients. Anticancer Res. 2009; 29:4309–4313. PMID: 19846992.20. Rosso L, Brock CS, Gallo JM, Saleem A, Price PM, Turkheimer FE, et al. A new model for prediction of drug distribution in tumor and normal tissues : pharmacokinetics of temozolomide in glioma patients. Cancer Res. 2009; 69:120–127. PMID: 19117994.
Article21. Sanghera P, Perry J, Sahgal A, Symons S, Aviv R, Morrison M, et al. Pseudoprogression following chemoradiotherapy for glioblastoma multiforme. Can J Neurol Sci. 2010; 37:36–42. PMID: 20169771.
Article22. Sanghera P, Rampling R, Haylock B, Jefferies S, McBain C, Rees JH, et al. The concepts, diagnosis and management of early imaging changes after therapy for glioblastomas. Clin Oncol (R Coll Radiol). 2012; 24:216–227. PMID: 21783349.
Article23. Scherrmann JM. Expression and function of multidrug resistance transporters at the blood-brain barriers. Expert Opin Drug Metab Toxicol. 2005; 1:233–246. PMID: 16922639.
Article24. Taal W, Brandsma D, de Bruin HG, Bromberg JE, Swaak-Kragten AT, Smitt PA, et al. Incidence of early pseudo-progression in a cohort of malignant glioma patients treated with chemoirradiation with temozolomide. Cancer. 2008; 113:405–410. PMID: 18484594.
Article25. Thompson EM, Frenkel EP, Neuwelt EA. The paradoxical effect of bevacizumab in the therapy of malignant gliomas. Neurology. 2011; 76:87–93. PMID: 21205697.
Article26. Vogelbaum MA, Jost S, Aghi MK, Heimberger AB, Sampson JH, Wen PY, et al. Application of novel response/progression measures for surgically delivered therapies for gliomas : Response Assessment in Neuro-Oncology (RANO) Working Group. Neurosurgery. 2012; 70:234–243. discussion 243-244. PMID: 21593697.27. Vredenburgh JJ, Desjardins A, Herndon JE 2nd, Dowell JM, Reardon DA, Quinn JA, et al. Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res. 2007; 13:1253–1259. PMID: 17317837.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Overview of CNS Gliomas in Childhood
- A Survey of the Neuro-Oncology Landscape
- The Use of MR Perfusion Imaging in the Evaluation of Tumor Progression in Gliomas
- Radiomics and Deep Learning from Research to Clinical Workflow: Neuro-Oncologic Imaging
- The Overview of Practical Guidelines for Gliomas by KSNO, NCCN, and EANO